The seemingly simple question of urine temperature for drug testing delves into a surprisingly complex intersection of biological realities, laboratory science, and the technological advancements that ensure the integrity of these crucial assessments. Far from being a mere footnote, the temperature of a urine sample is a critical determinant in the validity of a drug test, directly impacting the reliability of the results and the trust placed in the testing process. This article will explore the technological underpinnings and practical considerations surrounding urine temperature in drug testing, focusing on the sophisticated methods employed to maintain sample integrity and prevent tampering.

The Science Behind Urine Temperature and Drug Testing
The primary concern regarding urine temperature in drug testing is its potential as an indicator of sample substitution or dilution. When a person is asked to provide a urine sample for a drug test, it is generally understood that the sample should be “freshly voided.” This biological reality is directly linked to temperature.
Biological Baseline: The Natural Temperature of Urine
Urine, as a bodily fluid, is expelled from the body at a temperature that closely mirrors core body temperature. For a healthy individual, this typically falls within the range of 90°F to 100°F (32°C to 38°C). This internal warmth is a direct consequence of metabolic processes occurring within the body. When urine is voided, it begins to cool almost immediately upon contact with the cooler ambient air. The rate of cooling depends on several factors, including the initial temperature, the volume of the sample, the ambient temperature, and the type of container it’s collected in.
The Significance of Temperature in Drug Testing Protocols
Laboratories and testing facilities adhere to strict protocols to ensure the accuracy and legitimacy of drug test results. One of the most fundamental checks is the measurement of the urine sample’s temperature. This is not an arbitrary requirement; it’s a scientifically grounded practice designed to detect potential manipulation.
- Detecting Substitution: If a donor is attempting to substitute their urine sample with a pre-collected or purchased sample (often referred to as “synthetic urine”), that substitute sample is unlikely to be at body temperature. Unless meticulously heated and maintained, it will likely be at room temperature or colder. A significantly low temperature reading is an immediate red flag, suggesting that the sample is not freshly voided and may be an attempt to circumvent the test.
- Identifying Dilution: While temperature doesn’t directly indicate dilution, it can be a secondary indicator. If a sample is too dilute, it might suggest the donor attempted to flush their system with excessive fluids prior to the test. While dilution primarily affects drug metabolite concentrations, an unnaturally cool temperature in conjunction with other indicators can reinforce suspicion.
- Ensuring Sample Integrity: Beyond detecting fraudulent attempts, maintaining an appropriate temperature range also serves to prevent degradation of certain analytes. While most drug metabolites are relatively stable, extreme temperature fluctuations or prolonged exposure to non-physiological temperatures could theoretically impact the accuracy of some testing methods, although this is less of a primary concern than sample substitution.
Technological Solutions for Temperature Verification
The need to accurately and reliably verify urine temperature has driven the development and integration of specific technologies within the drug testing process. These technologies are designed to be both efficient and robust, ensuring that the integrity of the sample can be assessed quickly and decisively.
The Role of Thermometers and Temperature Strips
The most common and straightforward technological application for temperature verification is the use of thermometers or, more frequently, integrated temperature strips.
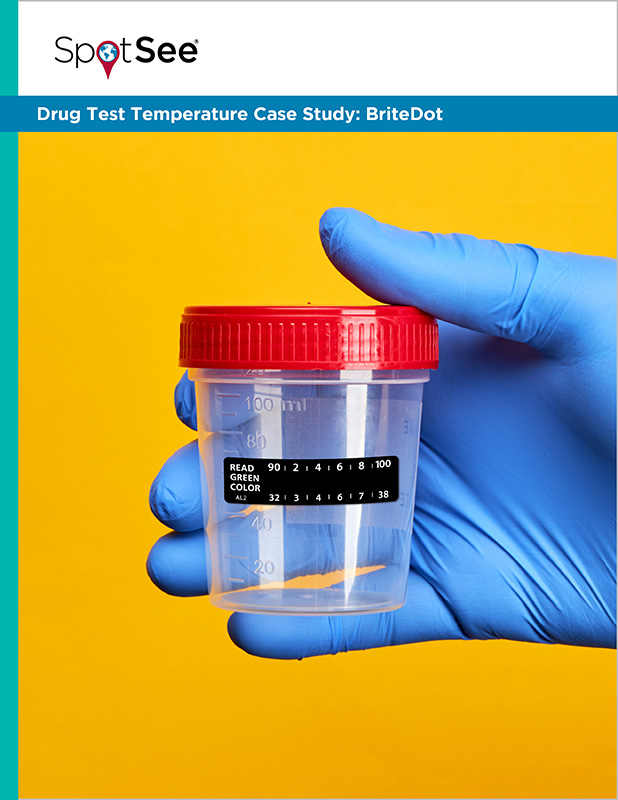
- Temperature Strips: Many collection cups used in drug testing come equipped with a built-in temperature strip. This strip contains a series of colored squares that change color at specific temperature intervals. When the urine sample is collected, the collector will observe the strip to determine the temperature range. A typical temperature strip will have a designated “acceptable range” indicated, often between 90°F and 100°F (32°C and 38°C). If the strip falls outside this range, the sample is immediately flagged.
- Digital Thermometers: In some advanced collection sites or for specific types of testing, digital thermometers might be used. These offer a more precise reading and can be easily calibrated, ensuring accuracy. However, their use requires more active handling by the collection personnel.
The primary technological advantage of these integrated strips is their simplicity and immediate feedback. They are designed for quick visual assessment, allowing the collection process to proceed efficiently while still capturing a crucial data point. The technology is essentially a passive indicator, relying on the principle of liquid crystal thermochromism, where specific chemical compounds change color in response to temperature.
The Importance of Timing: When Temperature is Measured
The timing of the temperature measurement is as critical as the measurement itself. Drug testing protocols mandate that the temperature of the urine sample be taken within a very specific timeframe after it has been voided.
- Immediate Measurement: The ideal scenario is for the temperature to be measured within minutes of the sample being provided. This is because urine cools rapidly. If a sample is collected and then sits for an extended period before its temperature is checked, it will naturally cool down, potentially falling outside the acceptable range even if it was initially at body temperature.
- Standard Operating Procedures (SOPs): Collection sites operate under strict SOPs that dictate the precise window for temperature assessment. This is typically within 4 minutes of collection. This short interval is designed to prevent a legitimate, freshly voided sample from cooling to an unacceptably low temperature due to ambient conditions, while still being early enough to detect a substitute sample that has not been properly heated.
- Documentation: The temperature reading, along with the time it was taken, is meticulously documented on the chain of custody form. This documentation is a vital part of the evidence trail, demonstrating that the testing protocols were followed correctly. Any deviation from these documented procedures can be grounds for challenging the validity of the test.
Beyond the Basic: Advanced Technological Safeguards
While temperature strips and immediate measurement are the most common technological interventions, the evolution of drug testing has seen the integration of more sophisticated methods to further enhance sample integrity and prevent tampering, especially in high-stakes scenarios.
Tamper-Evident Seals and Packaging
The technology of securing the sample extends beyond temperature verification. Tamper-evident features play a crucial role in ensuring that the sample remains uncontaminated and unaltered from the point of collection to its arrival at the laboratory.
- Sealed Containers: Urine collection cups are typically sealed with tamper-evident lids or caps. These are designed to show clear signs of tampering if the seal is broken or compromised. This technology provides a physical barrier and an immediate visual indicator that the sample has not been accessed or interfered with.
- Tamper-Evident Seals: Beyond the primary container, individual specimen bottles or transport bags often feature additional tamper-evident seals. These seals can be designed with specific holographic patterns, micro-text, or breakage mechanisms that make them difficult to counterfeit or remove without leaving obvious evidence. The technological design of these seals leverages advanced printing and material science to provide a high level of security.
Integrated Testing Devices and Electronic Monitoring
In some modern drug testing solutions, particularly those utilizing rapid on-site testing devices, temperature verification might be integrated into the device itself.
- Smart Collection Devices: Emerging technologies are exploring “smart” collection devices that can electronically record temperature at the point of collection and transmit this data wirelessly. This reduces reliance on manual documentation and minimizes the potential for human error or manipulation of records. These devices often incorporate sensors that continuously monitor temperature within a defined range.
- Direct Integration with Analyzers: For laboratory-based testing, sophisticated analytical instruments are designed to detect anomalies. While the primary focus is on drug metabolites, some systems can also flag samples with unusual physical characteristics, which might include temperature deviations not caught at the collection site, though this is less common as a primary detection method for temperature itself.
The underlying technology here is often based on microelectronics, sensor technology, and secure data transmission protocols. These advancements aim to create a more seamless and secure chain of custody, where every step of the sample handling process is digitally monitored and validated.

The Future of Temperature Integrity in Drug Testing
As technology continues to advance, we can expect even more sophisticated methods to emerge for ensuring the integrity of urine samples in drug testing. The focus will likely remain on preventing substitution and dilution, with temperature playing a key role as an initial, rapid indicator.
- Real-time Monitoring: Future technologies might involve devices capable of near real-time temperature monitoring and transmission, potentially even integrating with wearable technology or secure collection stations that digitally capture and timestamp all critical data points.
- Advanced Material Science: Innovations in tamper-evident materials and chemical indicators could lead to even more foolproof methods of detecting unauthorized access or manipulation.
- Biometric Integration: In highly sensitive testing environments, the integration of biometric verification at the point of collection, linked to the sample itself, could further fortify the integrity of the process, making temperature a secondary, albeit still vital, check.
In conclusion, the temperature of urine for a drug test is a critical parameter dictated by biological realities and meticulously monitored through various technological means. From simple temperature strips to potentially more advanced electronic monitoring systems, the overarching goal remains consistent: to ensure the accuracy, reliability, and trustworthiness of drug testing results by safeguarding the integrity of the biological sample. This seemingly basic measurement underpins the entire drug testing process, serving as a vital first line of defense against fraudulent practices and ultimately upholding the fairness and efficacy of drug screening programs across various sectors.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.