The ability to measure time across millennia is not merely a feat of historical inquiry; it is a triumph of high-precision technology. At the heart of this chronological mastery lies Carbon-14 (14C), a radioactive isotope that serves as Nature’s own digital clock. For technologists, engineers, and researchers, the “half-life of 14C” is more than a scientific constant—it is the foundational metric for a sophisticated array of hardware and software solutions that allow us to reconstruct the past with surgical precision.
Understanding the half-life of 14C requires an exploration of nuclear physics, high-end mass spectrometry, and the complex algorithms used to calibrate raw data into human history. As we delve into the mechanics of 14C, we find a field that has evolved from rudimentary Geiger counters to state-of-the-art Accelerator Mass Spectrometry (AMS), representing one of the most significant technological leaps in the last century.
Understanding the Core Mechanism: The Physics and Tech of Carbon-14
To appreciate the technology used to measure 14C, one must first understand the “data” itself. Carbon-14 is a rare, radioactive isotope of carbon created in the upper atmosphere. When cosmic rays collide with nitrogen atoms, they produce 14C, which eventually finds its way into all living organisms through the carbon cycle.
The Cosmogenic Production Cycle
The production of 14C is a continuous atmospheric process. Unlike Carbon-12, which is stable, 14C is unstable. As long as an organism is alive, it continuously replenishes its 14C supply by breathing or eating, maintaining an equilibrium with the atmospheric concentration. However, at the moment of death, this “intake” stops. The biological “clock” is set to zero, and the 14C begins to decay at a predictable rate.
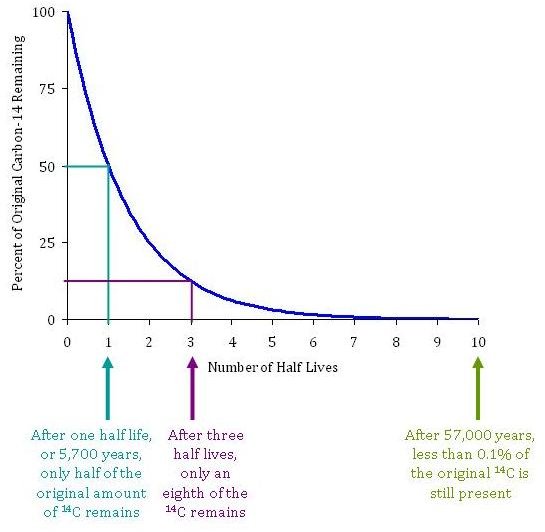
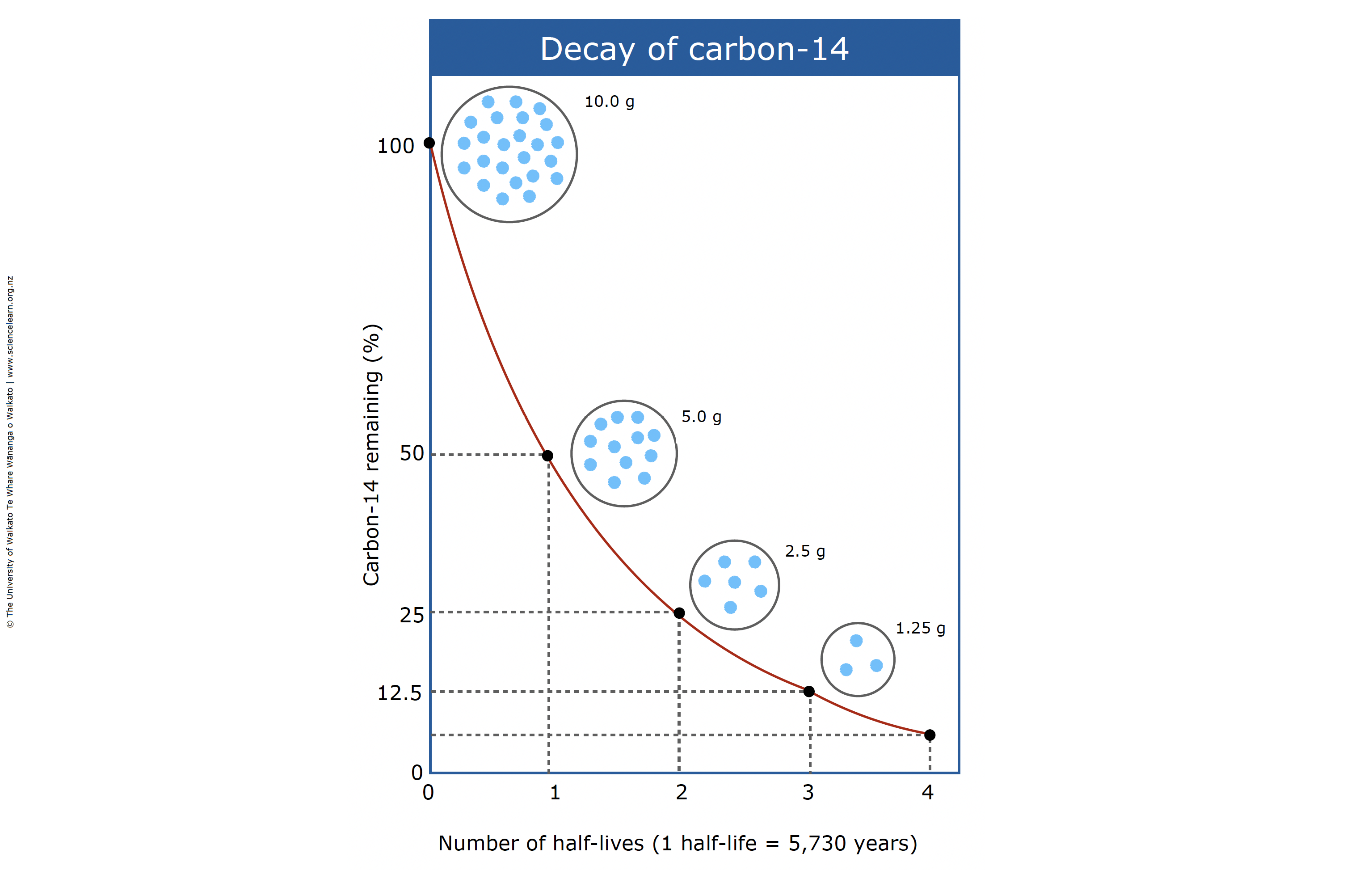
Defining the 5,730-Year Half-Life
The “half-life” is the duration required for half of the initial quantity of a radioactive substance to decay. For Carbon-14, the internationally accepted value—known as the Cambridge half-life—is approximately 5,730 years. From a technical perspective, this means that every 5,730 years, the “signal” from the 14C isotopes in a sample weakens by 50%.
This specific timeframe makes 14C an ideal “tech tool” for dating organic materials up to about 50,000 years old. Beyond that limit, the concentration of 14C becomes so infinitesimal that it drops below the “noise floor” of even the most sensitive modern sensors, requiring different isotopic technologies for older measurements.
Advanced Detection: From Geiger Counters to Accelerator Mass Spectrometry (AMS)
The evolution of 14C measurement hardware is a classic story of technological refinement. In the early days of radiocarbon dating, pioneered by Willard Libby in the late 1940s, the process was slow, required massive samples, and lacked the precision we expect today.
The Legacy of Beta Counting
The original technology for dating relied on “Gas Proportional Counting” or “Liquid Scintillation Counting.” These methods were essentially “listening” for the decay. Because 14C decays by emitting a beta particle, scientists would place a sample in a shielded chamber and wait for these emissions to occur.
The tech hurdle here was significant: because the half-life is so long (5,730 years), very few atoms decay in a short period. To get a statistically significant reading, researchers needed large chunks of precious artifacts—sometimes an entire wooden bowl or a large section of ancient fabric—to capture enough “pings” on their detectors.
AMS: The High-Tech Leap in Precision
The game-changer arrived with the development of Accelerator Mass Spectrometry (AMS). Instead of waiting for individual atoms to decay and emit a signal, AMS technology treats the sample like a stream of data to be sorted.
An AMS system is a massive particle accelerator. It ionizes the carbon atoms in a sample, accelerates them to incredibly high velocities using high-voltage gradients, and then passes them through a series of powerful magnets. Because 14C atoms have a different mass than 12C or 13C atoms, the magnets deflect them at different angles.
This allows the hardware to literally count every single 14C atom in the sample individually. The technological advantage is twofold:
- Sample Size: AMS requires only milligrams of material (the size of a grain of sand).
- Speed and Sensitivity: Measurements that once took weeks can now be completed in hours with a much higher signal-to-noise ratio.
Digital Calibration and the “IntCal” Software Revolution
Even with the most advanced AMS hardware, the raw data produced—the “radiocarbon age”—is not the same as the “calendar age.” This is where the intersection of technology and data science becomes critical.
Why Raw Data Isn’t Enough
The fundamental assumption of early 14C dating was that the concentration of 14C in the atmosphere has always been constant. We now know, through high-tech atmospheric modeling and dendrochronology (tree-ring dating), that this is false. Solar activity, changes in the Earth’s magnetic field, and human industrial activity have caused 14C levels to fluctuate over time.
Algorithm-Driven Calibration Curves
To correct these fluctuations, technologists have developed the “IntCal” (International Calibration) curves. These are massive databases that correlate radiocarbon years with actual calendar years. The most recent iteration, IntCal20, utilizes sophisticated Bayesian statistical modeling to integrate data from tree rings, lake sediments, and coral reefs.
Modern dating software allows researchers to input their AMS raw data, which the software then “runs” against the IntCal curve using Markov Chain Monte Carlo (MCMC) algorithms. This digital processing accounts for the “wiggles” in the carbon record, providing a probability distribution of the artifact’s true age. Without this software layer, the hardware’s precision would be historically meaningless.
Modern Applications: Tech Beyond Archaeology
While 14C is famous for dating ancient ruins, the technology has expanded into various modern tech sectors, including forensic science, environmental monitoring, and anti-fraud operations.
Detecting Art Forgery with Digital Isotopes
The “Bomb Pulse” is a fascinating technological byproduct of the Cold War. Atmospheric nuclear testing in the mid-20th century nearly doubled the amount of 14C in the atmosphere. This “spike” created a unique signature in any organic material grown after 1955.
High-tech forensic labs use this signature to identify art forgeries. If a painting claimed to be from the 17th century contains linseed oil or canvas fibers with “Bomb Pulse” levels of 14C, the technology instantly flags it as a modern creation. This same technique is used in the luxury wine industry to verify the vintage of world-class bottles.
Environmental Monitoring and Fossil Fuel Tracking
In the tech-driven fight against climate change, 14C serves as a vital sensor. Fossil fuels—coal, oil, and gas—are millions of years old. Because their age far exceeds the 50,000-year limit of 14C, they contain zero Carbon-14.
By using AMS to analyze the CO2 in the atmosphere near industrial hubs, tech firms can calculate exactly how much of that carbon comes from biological sources (which have 14C) versus fossil fuel combustion (which has none). This provides a precise, data-driven way to audit carbon emissions and verify the efficacy of “green” initiatives.
The Future of Dating Technology: Quantum and Laser Advancements
As we look toward the future, the technology of 14C measurement is moving away from massive, building-sized accelerators toward more portable and efficient systems.
Cavity Ring-Down Spectroscopy (CRDS)
One of the most exciting emerging technologies in this niche is Cavity Ring-Down Spectroscopy (CRDS). Unlike AMS, which uses magnets and acceleration, CRDS uses high-precision lasers. By tuning a laser to the specific “optical fingerprint” of 14C, researchers can measure the concentration of the isotope based on how the light is absorbed within a reflective cavity.
The potential for this tech is revolutionary. CRDS systems are significantly smaller and cheaper than AMS systems. In the near future, we may see “portable carbon dating” units that can be used in the field, providing real-time data to archaeologists and environmental scientists without the need to ship samples to a centralized particle accelerator facility.
Automation and AI Integration
Furthermore, the integration of Artificial Intelligence into calibration software is streamlining the process of data interpretation. AI models can now analyze thousands of sample results simultaneously, identifying patterns in historical carbon fluctuations that were previously invisible to human researchers. This allows for a more “automated” history, where the technology handles the heavy lifting of chronological reconstruction.
The half-life of 14C remains 5,730 years, but our ability to interact with that constant is evolving at the speed of modern tech. From the physical hardware of the accelerator to the Bayesian logic of the calibration software, the measurement of 14C stands as a testament to human ingenuity—a bridge between the ancient world and the digital future.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.