In the realm of quality management, the acronym CAPA stands for Corrective Action and Preventive Action. It’s a systematic, robust process designed to identify, investigate, and resolve issues that have occurred and to prevent them from happening again. While the term “quality” itself can be broad, encompassing everything from product defect rates to customer satisfaction scores, CAPA serves as a critical mechanism for driving continuous improvement within any organization striving for excellence.
At its core, CAPA is not merely a reactive measure to address problems; it’s a proactive strategy to enhance processes, products, and services. It’s a fundamental pillar of many quality management systems (QMS), including ISO 9001, and is particularly vital in highly regulated industries like pharmaceuticals, medical devices, and aerospace. However, the principles of CAPA are universally applicable and can significantly benefit businesses across all sectors seeking to maintain high standards, reduce waste, and foster customer loyalty.

Understanding CAPA is crucial for anyone involved in quality assurance, process improvement, or operational management. It’s about creating a cycle of learning and adaptation, ensuring that once a flaw is identified, it’s not only fixed but also understood to its root cause, thereby preventing its recurrence. This article will delve into the intricacies of CAPA, exploring its components, its significance, and its practical application in driving tangible improvements within a technological context.
The Core Components of CAPA
CAPA is more than just a two-word phrase; it represents a structured methodology with distinct phases and objectives. To truly grasp its essence, it’s important to break down the “Corrective Action” and “Preventive Action” into their individual components and understand how they work in tandem.
Understanding Corrective Action
Corrective action is the reactive part of the CAPA process. It’s what you do after a non-conformance, a defect, a customer complaint, or any other undesirable event has been identified. The primary goal of corrective action is to address the immediate problem and eliminate its cause. This involves a deep dive into what went wrong, why it happened, and how to fix it.
Identifying and Defining the Problem
The first step in corrective action is clearly and accurately identifying the problem. This might involve data from quality control tests, customer feedback, audit findings, or internal process monitoring. The problem needs to be precisely defined, including its scope, impact, and severity. Vague problem statements lead to ineffective solutions. For instance, “the software has bugs” is less effective than “users are experiencing frequent crashes when attempting to save large files in the application version 2.3.1 due to memory leaks.”
Investigating the Root Cause
This is arguably the most critical and challenging aspect of corrective action. Simply fixing the symptom won’t prevent future occurrences. Root cause analysis (RCA) is the systematic process of identifying the underlying reasons for the problem. Common RCA tools include:
- The 5 Whys: Repeatedly asking “why” until the fundamental cause is uncovered.
- Fishbone Diagram (Ishikawa Diagram): Categorizes potential causes into areas like People, Process, Equipment, Materials, Environment, and Management.
- Fault Tree Analysis (FTA): A top-down deductive failure analysis where a system’s undesired state is analyzed using Boolean logic to combine a series of lower-level events.
- Pareto Chart: Identifies the most significant factors that contribute to a problem, based on the Pareto principle (80/20 rule).
The aim of root cause analysis is to move beyond superficial explanations and uncover the fundamental systemic issues that allowed the problem to occur in the first place.
Developing and Implementing Solutions
Once the root cause(s) are identified, effective solutions must be developed and implemented. These solutions should directly address the root cause(s). This might involve modifying a process, updating software code, retraining personnel, changing equipment, or improving documentation. The implementation plan should include clear responsibilities, timelines, and necessary resources.
Verifying Effectiveness
After the corrective action has been implemented, its effectiveness must be verified. This means confirming that the problem has indeed been resolved and that the implemented solutions are working as intended. This verification should be objective and based on measurable data. If the corrective action proves to be ineffective, the CAPA process must be revisited.
Understanding Preventive Action
Preventive action, as the name suggests, is about looking ahead and proactively identifying potential issues before they manifest. While corrective action deals with what has happened, preventive action focuses on what could happen. It’s about leveraging insights gained from past issues (both your own and industry-wide) and potential future risks to implement changes that prevent problems from ever arising.
Identifying Potential Problems
This involves a forward-looking perspective. Organizations can identify potential problems through various means:
- Trend Analysis: Analyzing historical data for emerging patterns that might indicate future issues.
- Risk Assessments: Proactively evaluating processes, products, or systems for potential failure points.
- Industry Best Practices and Benchmarking: Learning from the experiences and challenges faced by other organizations.
- New Technology Adoption: Understanding potential risks and challenges associated with implementing new technologies.
- Customer Feedback and Market Research: Anticipating future customer needs and potential areas of dissatisfaction.
Analyzing Potential Causes
Similar to root cause analysis, preventive action requires understanding the potential causes of future problems. This might involve brainstorming, scenario planning, and predictive modeling. The goal is to anticipate the “why” behind a potential failure.
Developing and Implementing Preventive Measures
Based on the identified potential problems and their likely causes, preventive measures are developed and implemented. These are proactive steps designed to eliminate the possibility of the problem occurring. Examples in a tech context could include:
- Implementing more rigorous testing protocols for new software features.
- Investing in advanced cybersecurity training for IT staff.
- Designing systems with inherent redundancy to prevent single points of failure.
- Developing comprehensive user documentation to preempt common support queries.
- Automating quality checks at critical points in the development lifecycle.
Monitoring and Review
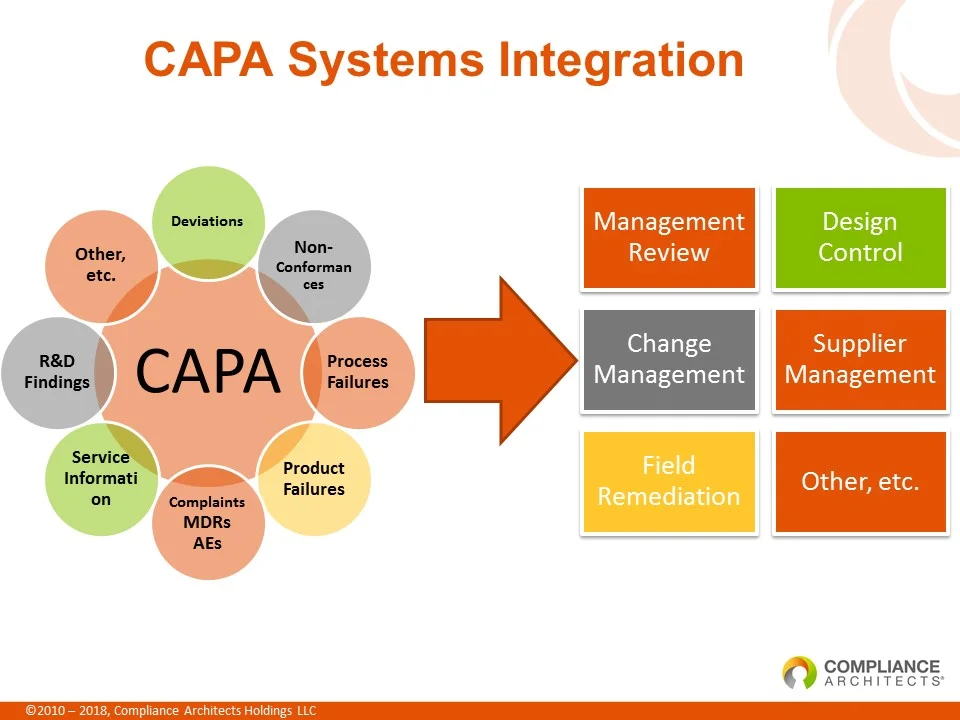
Preventive actions are not set-it-and-forget-it. They need to be continuously monitored to ensure they remain effective. The landscape of technology and business is constantly evolving, and what was a relevant preventive measure yesterday might be obsolete today. Regular review and updates are essential.
The Significance of CAPA in the Tech Landscape
In the fast-paced and ever-evolving tech industry, CAPA is not just a compliance requirement; it’s a strategic imperative for survival and growth. The nature of technology—rapid innovation, complex systems, and constant updates—presents unique challenges and opportunities that CAPA can effectively address.
Driving Continuous Improvement and Innovation
CAPA fosters a culture of continuous improvement, which is the lifeblood of the tech sector. By systematically addressing issues, companies learn from their mistakes and refine their processes, products, and services. This iterative process is directly linked to innovation. When you understand why a feature failed or a system crashed, you gain insights that can lead to better design, more robust architecture, and ultimately, more innovative solutions.
For example, if a software bug leads to data corruption, a thorough CAPA investigation might reveal that the database schema was not designed to handle certain data types or that error handling in a specific module was inadequate. The corrective action would fix the immediate issue, but the preventive action might involve re-evaluating database design principles for all future projects, implementing stricter code review processes for data-handling modules, or developing automated data integrity checks. This proactive approach ensures that future software releases are more robust and less prone to similar data-related problems, paving the way for more complex and reliable applications.
Enhancing Product Quality and Reliability
In technology, quality often translates to reliability. Users expect software to be stable, hardware to be durable, and services to be consistently available. CAPA plays a direct role in achieving these expectations. When a critical bug is identified in a widely used application, the CAPA process ensures that it’s not just patched but that the underlying cause is understood and prevented from recurring in future versions or related products.
Consider the development of a new AI model. If early testing reveals biases in its outputs that lead to unfair or discriminatory results, a CAPA process would be initiated. The corrective action would involve identifying the biased training data or algorithmic flaws and retraining the model with corrected data or adjusted algorithms. The preventive action, however, would be to establish robust protocols for data sourcing and bias detection throughout the AI development lifecycle, ensuring that future models are trained on diverse and unbiased datasets and undergo rigorous fairness testing before deployment. This commitment to quality and reliability builds user trust and reduces the reputational and financial damage associated with product failures.
Reducing Costs and Improving Efficiency
Unresolved issues in technology can lead to significant costs. These can include customer support expenses, lost sales due to system downtime, costs associated with product recalls or rework, and the expense of fixing critical bugs under pressure. CAPA, by preventing recurring issues, directly contributes to cost reduction and improved operational efficiency.
Imagine a scenario where a cloud service experiences frequent outages. Each outage incurs costs for downtime, customer service escalations, and potential SLA penalties. A CAPA process would investigate the root cause of these outages—perhaps it’s an aging infrastructure component, a lack of sufficient monitoring, or a flawed deployment process. The corrective actions would address the immediate failures. The preventive actions, however, would involve a strategic upgrade of infrastructure, implementation of advanced predictive monitoring tools, or a complete overhaul of the deployment pipeline. By proactively addressing the systemic causes, the company not only resolves the current problem but also significantly reduces the likelihood of future, costly outages, leading to substantial cost savings and improved operational efficiency.
Meeting Regulatory Compliance and Industry Standards
Many sectors within the technology industry, particularly those dealing with sensitive data or critical infrastructure (e.g., fintech, healthtech, cybersecurity), are subject to stringent regulatory requirements. CAPA is often a mandatory component of these regulations. For instance, the FDA’s Quality System Regulation (21 CFR Part 820) for medical devices, or ISO 27001 for information security management, explicitly require robust CAPA procedures.
For a cybersecurity firm developing security software, non-compliance with industry standards or regulations can have severe consequences, including hefty fines, loss of client trust, and even legal repercussions. A vulnerability discovered in their software that could be exploited by attackers would trigger a CAPA. The corrective action would involve patching the vulnerability immediately. The preventive action would be to implement a comprehensive security code review process, integrate static and dynamic analysis tools into the CI/CD pipeline, and conduct regular penetration testing exercises. This rigorous adherence to CAPA not only ensures compliance but also solidifies the company’s reputation as a secure and trustworthy provider in a highly sensitive market.
Implementing a Robust CAPA System in Tech
Establishing an effective CAPA system in a technology-focused organization requires a strategic approach, encompassing clear processes, the right tools, and a supportive culture. It’s not a one-size-fits-all solution but rather an adaptable framework.
Establishing Clear Procedures and Workflows
The foundation of any CAPA system is a well-defined set of procedures and workflows. These documents should clearly outline:
- How to identify and report issues: This includes defining what constitutes a reportable event and the channels through which these events can be reported (e.g., bug tracking systems, incident management platforms, feedback forms).
- The escalation process: Who is responsible for reviewing, prioritizing, and assigning CAPA investigations based on severity and impact.
- The investigation methodology: Specifying the tools and techniques to be used for root cause analysis and risk assessment.
- The approval process for actions: Defining who needs to sign off on proposed corrective and preventive actions.
- The verification and closure criteria: How the effectiveness of implemented actions will be measured and when a CAPA can be considered closed.
- Documentation requirements: What information needs to be recorded at each stage of the CAPA process for auditability and future reference.
These procedures should be accessible to all relevant personnel and regularly reviewed for updates.
Leveraging Technology and Tools
The tech industry, by its nature, can significantly benefit from technological solutions to manage its CAPA processes. Several types of tools can enhance efficiency and effectiveness:
- Quality Management System (QMS) Software: Dedicated QMS platforms often have built-in CAPA modules. These tools can automate workflows, manage documentation, track progress, and provide reporting and analytics. Examples include platforms like MasterControl, Greenlight Guru, or even integrated modules within enterprise resource planning (ERP) systems.
- Issue Tracking and Project Management Tools: Tools like Jira, Asana, or Trello can be adapted to manage CAPA workflows, especially for software development. They allow for the creation of specific CAPA tickets, assignment of tasks, tracking of progress, and attachment of relevant documentation.
- Data Analysis and Visualization Tools: For root cause analysis and trend identification, tools like Tableau, Power BI, or even advanced scripting with Python libraries (e.g., Pandas, Matplotlib) can be invaluable for sifting through large datasets and identifying patterns.
- Collaboration and Communication Platforms: Tools like Slack, Microsoft Teams, or Confluence facilitate seamless communication among teams involved in CAPA investigations, ensuring everyone is informed and can contribute effectively.
The key is to select tools that integrate well with existing systems and support the defined CAPA workflows, rather than creating additional silos.
![]()
Fostering a CAPA-Oriented Culture
Perhaps the most critical, yet often overlooked, aspect of implementing a successful CAPA system is cultivating the right organizational culture. This means moving beyond a purely compliance-driven mindset to one that embraces CAPA as a fundamental driver of improvement and innovation.
- Leadership Commitment: Senior leadership must champion CAPA, allocating resources and time for investigations and empowering teams to identify and address issues without fear of retribution.
- Cross-Functional Collaboration: CAPA issues often span multiple departments. Encouraging collaboration between engineering, quality assurance, product management, customer support, and operations is vital for comprehensive investigations and effective solutions.
- Training and Education: All employees, particularly those involved in product development, quality control, and customer service, should receive training on CAPA principles, their role in the process, and the tools available.
- Openness and Transparency: Creating an environment where employees feel comfortable reporting issues, even minor ones, is crucial. Transparency in the CAPA process, including sharing findings and lessons learned (appropriately), builds trust and reinforces the value of the system.
- Focus on Learning, Not Blame: The primary objective of CAPA is to learn from mistakes and improve. The culture should actively discourage a blame-oriented approach and instead focus on understanding systemic causes and implementing lasting solutions.
By integrating these elements, tech organizations can transform CAPA from a bureaucratic necessity into a powerful engine for continuous improvement, enhanced product quality, and sustained competitive advantage. The ability to effectively manage and learn from issues is a hallmark of mature and successful technology companies.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.