In the realm of chemistry, the concept of acidity and alkalinity is fundamental, dictating how substances interact and behave. Understanding the distinction between acidic and alkaline solutions is crucial for a wide array of applications, from industrial processes to biological functions. While often discussed in scientific contexts, the principles behind alkalinity have significant implications within the technology sector, particularly in areas concerning manufacturing, material science, and even in the development of advanced analytical tools. This article delves into the nature of alkaline solutions, their chemical underpinnings, and their relevant applications within the technology landscape.
The Chemical Foundation of Alkalinity
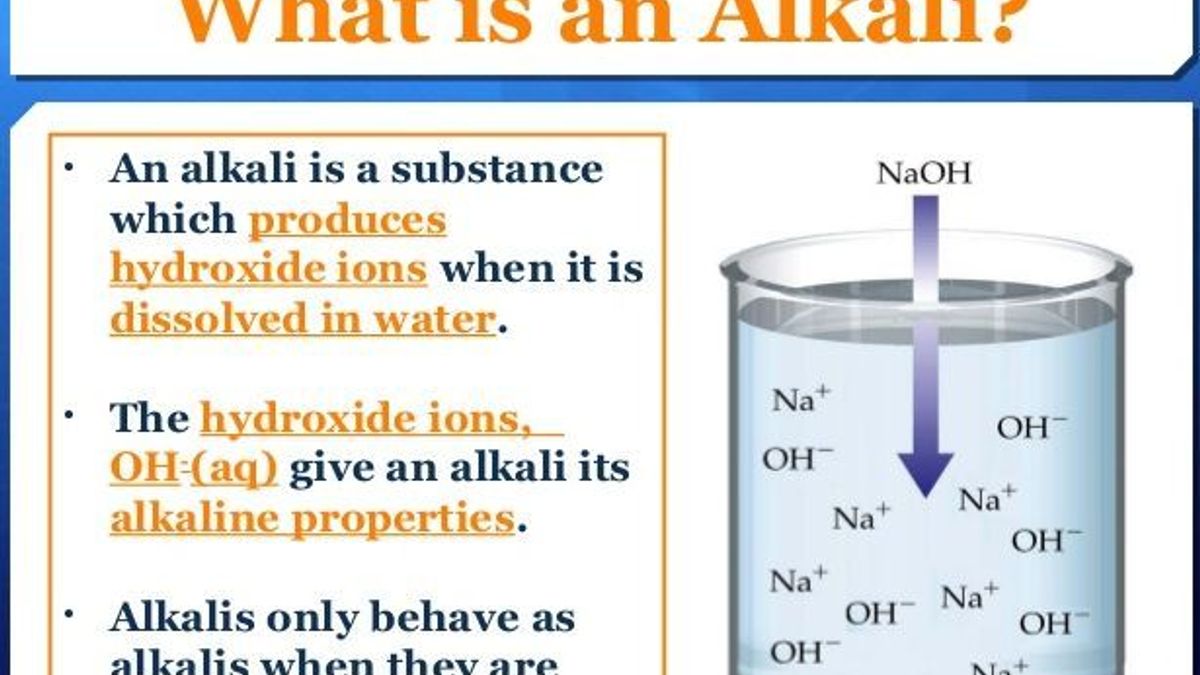
At its core, the alkalinity of a solution is determined by its concentration of hydroxide ions (OH⁻) relative to its concentration of hydrogen ions (H⁺). This relationship is quantified by the pH scale, a logarithmic measure ranging from 0 to 14. Solutions with a pH below 7 are considered acidic, those with a pH of exactly 7 are neutral, and solutions with a pH above 7 are alkaline, also commonly referred to as basic.
Defining pH and the pH Scale
The pH scale is a cornerstone of understanding acidity and alkalinity. Mathematically, pH is defined as the negative logarithm (base 10) of the hydrogen ion concentration: pH = -log₁₀[H⁺]. Conversely, pOH, which measures hydroxide ion concentration, is defined as pOH = -log₁₀[OH⁻]. In aqueous solutions at 25°C, the product of hydrogen and hydroxide ion concentrations is constant (Kw = [H⁺][OH⁻] = 1.0 x 10⁻¹⁴). This leads to a crucial relationship: pH + pOH = 14.
Therefore, when the concentration of hydrogen ions is low, the concentration of hydroxide ions must be high, resulting in a high pH value. This is the hallmark of an alkaline solution. Conversely, a high concentration of hydrogen ions signifies an acidic solution with a low pH. The scale is logarithmic, meaning that each unit change in pH represents a tenfold change in the hydrogen ion concentration. For instance, a solution with a pH of 9 is ten times more alkaline than a solution with a pH of 8, and one hundred times more alkaline than a solution with a pH of 7.
Common Alkaline Compounds and Their Properties
Alkaline solutions are typically formed by dissolving alkali metal hydroxides or alkaline earth metal hydroxides in water. These compounds readily dissociate, releasing hydroxide ions into the solution. Some of the most common and relevant alkaline compounds include:
- Sodium Hydroxide (NaOH): Also known as lye or caustic soda, sodium hydroxide is a strong alkali widely used in numerous industrial processes. Its high alkalinity makes it an effective cleaning agent and a crucial component in the manufacturing of soaps, detergents, and paper. In technology, it’s employed in etching processes for semiconductor fabrication.
- Potassium Hydroxide (KOH): Similar to sodium hydroxide, potassium hydroxide is another strong alkali. It finds applications in the production of soft soaps, alkaline batteries, and as an electrolyte in various electrochemical devices. Its use in certain battery technologies is directly relevant to the energy storage solutions underpinning much of modern technology.
- Ammonium Hydroxide (NH₄OH): This is a weaker alkali formed when ammonia gas dissolves in water. While not as strongly alkaline as the metal hydroxides, it still exhibits basic properties and is used in cleaning products, fertilizers, and in certain chemical synthesis processes. In technological contexts, it can be used in surface treatment and cleaning of sensitive electronic components.
- Calcium Hydroxide (Ca(OH)₂): Also known as slaked lime, calcium hydroxide is a moderately alkaline compound. It’s commonly used in water treatment to adjust pH and remove impurities, as well as in construction materials. Its role in water purification has indirect technological relevance, ensuring the quality of water used in manufacturing and research.
These compounds, when dissolved in water, significantly increase the concentration of hydroxide ions, thereby raising the pH and creating an alkaline environment. The strength of the alkali – whether it’s a strong or weak base – determines the extent of its dissociation and the resulting pH. Strong bases like NaOH and KOH dissociate almost completely, leading to highly alkaline solutions, while weak bases like NH₄OH only partially dissociate.
Alkaline Solutions in Technological Applications
The unique chemical properties of alkaline solutions – their ability to neutralize acids, act as solvents, and participate in redox reactions – make them indispensable in various technological sectors. From the intricate processes of microchip manufacturing to the development of advanced materials and energy storage systems, alkalinity plays a quiet yet critical role.
Microelectronics and Semiconductor Fabrication
One of the most significant areas where alkaline solutions are crucial is in the fabrication of microelectronic devices and semiconductors. The process of creating integrated circuits involves numerous steps that rely on precise chemical etching and cleaning.
-
Photolithography and Etching: In photolithography, a light-sensitive material (photoresist) is used to pattern semiconductor wafers. After exposing the photoresist to UV light through a mask, an alkaline developer solution is used to selectively remove the exposed or unexposed portions of the resist, depending on the type of photoresist used. This selective removal creates the intricate patterns that form the basis of circuits. Following the patterning of the photoresist, alkaline etching solutions are used to remove unwanted material from the wafer surface. For instance, buffered hydrofluoric acid (a mixture of hydrofluoric acid and a weak base like ammonium fluoride) is a common etchant for silicon dioxide layers, and its effectiveness is modulated by its pH. Alternatively, wet etching processes often utilize strong alkaline solutions like potassium hydroxide or tetramethylammonium hydroxide (TMAH) to etch silicon itself. TMAH is particularly favored for its selectivity and because it does not introduce mobile alkali metal ions (like sodium or potassium) that can degrade semiconductor performance.
-
Cleaning and Surface Preparation: The extremely high purity required for semiconductor manufacturing necessitates rigorous cleaning protocols at almost every stage. Alkaline solutions, particularly those based on sodium hydroxide or hydrogen peroxide mixtures (like SC-1 cleaning), are highly effective at removing organic residues, particles, and metallic contaminants from wafer surfaces. These cleaning steps are vital to prevent defects that could lead to device failure. The ability of alkaline solutions to saponify organic residues and complex metallic ions contributes to their effectiveness in achieving the pristine surfaces demanded by microelectronics.
Material Science and Advanced Manufacturing
Beyond semiconductors, alkaline solutions are integral to the development and processing of various advanced materials and in broader manufacturing contexts.
- Surface Treatment and Passivation: Alkaline solutions are used for cleaning and preparing metal surfaces before coating, plating, or painting. For example, alkaline degreasers effectively remove oils and greases from metal parts, ensuring good adhesion of subsequent layers. In some specialized applications, alkaline treatments are used for passivation, creating a protective oxide layer on metal surfaces to enhance corrosion resistance. This is relevant in the manufacturing of components for aerospace, automotive, and even consumer electronics where durability and aesthetics are paramount.

-
Nanomaterial Synthesis and Processing: The synthesis of many nanomaterials, such as nanoparticles and nanotubes, often involves alkaline conditions. For instance, the hydrothermal synthesis of certain metal oxide nanoparticles frequently utilizes alkaline solutions as the reaction medium. The pH influences the nucleation and growth kinetics of these nanomaterials, allowing for control over their size, shape, and crystalline structure. Furthermore, alkaline solutions can be used for dispersing and stabilizing nanoparticles in liquid media, preventing aggregation and facilitating their integration into composites and advanced functional materials. The ability to control the surface charge of nanoparticles via pH adjustment is a key factor in their colloidal stability and subsequent applications.
-
Composite Material Fabrication: In the production of certain composite materials, alkaline solutions can be used as part of the curing or processing stages. For example, in the manufacturing of some fiberglass composites, alkaline solutions might be used to treat the glass fibers, improving their adhesion to the polymer matrix.
Energy Storage and Electrochemistry
The development of more efficient and sustainable energy storage solutions is a critical technological frontier, and alkaline solutions play a direct role in several key technologies.
-
Alkaline Batteries: The classic alkaline battery, such as the AA or AAA battery, utilizes an electrolyte that is an alkaline solution, typically potassium hydroxide. In these batteries, the alkaline electrolyte facilitates the movement of ions between the anode and cathode, enabling the electrochemical reactions that produce electricity. While rechargeable lithium-ion batteries have become dominant, alkaline batteries remain prevalent due to their cost-effectiveness and reliability for low-drain devices. Research continues into advanced alkaline battery chemistries for specific applications.
-
Electrolysis and Hydrogen Production: Alkaline electrolysis is a well-established method for producing hydrogen gas from water. In this process, an alkaline electrolyte (often potassium hydroxide) is used to conduct ions between electrodes, splitting water molecules into hydrogen and oxygen. While other electrolysis methods exist, alkaline electrolysis is known for its robustness and relatively lower cost, making it a contender for large-scale hydrogen production, which is increasingly important for clean energy initiatives and industrial processes. The efficiency of this process is highly dependent on the concentration and purity of the alkaline electrolyte.
-
Supercapacitors: Some types of supercapacitors, which are electrochemical energy storage devices that store energy electrostatically, also employ alkaline electrolytes. These electrolytes offer high ionic conductivity, contributing to the rapid charge and discharge rates characteristic of supercapacitors, which are used in applications requiring quick bursts of power, such as regenerative braking systems in electric vehicles.
Safety and Handling of Alkaline Solutions
Despite their beneficial applications, alkaline solutions, particularly strong ones, can be corrosive and pose significant safety hazards if not handled properly. Understanding these risks and adhering to appropriate safety protocols is paramount in any technological setting where they are used.
Corrosivity and Tissue Damage
Strong alkaline solutions are highly corrosive to organic tissues. They can cause severe burns to the skin and eyes, and if ingested, can lead to serious internal damage to the digestive tract. The mechanism of alkaline burns differs from acidic burns; alkaline substances tend to saponify fats in tissues, leading to deep, penetrating, and often slow-healing wounds. This is because the damage can continue as long as the alkaline substance remains in contact with the tissue, as it penetrates deeper.
Protective Measures and Equipment
When working with alkaline solutions, it is essential to implement stringent safety measures:
- Personal Protective Equipment (PPE): This includes chemical-resistant gloves (e.g., nitrile or neoprene), safety goggles or a face shield to protect the eyes and face from splashes, and a lab coat or apron to protect clothing and skin.
- Ventilation: Many alkaline solutions can release fumes, especially when heated or mixed with other chemicals. Working in a well-ventilated area or using a fume hood is crucial to prevent inhalation of these vapors.
- Dilution Procedures: Always add alkaline substances slowly to water, never the other way around. Adding water to a concentrated alkali can generate significant heat, potentially causing boiling and splashing. The process of dilution itself is exothermic and requires careful management.
- Emergency Procedures: Knowing the location of eyewash stations and safety showers is vital. In case of skin contact, immediately rinse the affected area with copious amounts of water for at least 15-20 minutes. For eye contact, flush the eyes with water for an extended period and seek immediate medical attention.
Storage and Disposal
Proper storage and disposal of alkaline solutions are also critical for safety and environmental protection.
- Storage: Alkaline solutions should be stored in tightly sealed containers made of compatible materials (e.g., polyethylene or glass). They should be kept separate from acids and other incompatible chemicals to prevent dangerous reactions. Storage areas should be cool, dry, and well-ventilated.
- Disposal: Disposal of alkaline waste must comply with local environmental regulations. Typically, alkaline waste is neutralized with an acid (e.g., dilute acetic acid or hydrochloric acid) under controlled conditions to bring the pH close to neutral (between 6 and 9) before it can be discharged or further treated. The neutralization process generates heat and may produce salts, so it must be performed cautiously.

Conclusion: The Ubiquitous Role of Alkalinity in Technology
While the term “alkaline solution” might evoke images of chemistry labs, its practical implications extend deeply into the fabric of modern technology. From the microscopic precision required in semiconductor fabrication, where alkaline etchants and developers define the circuits that power our digital world, to the robust performance of everyday alkaline batteries and the emerging potential of alkaline electrolysis for clean energy, alkalinity is a pervasive and vital chemical principle.
The ability of alkaline solutions to selectively remove materials, clean surfaces to extraordinary purity levels, facilitate electrochemical reactions, and modify material properties makes them indispensable tools in research, development, and mass production. As technological innovation continues, the demand for precise chemical control will only grow, further cementing the importance of understanding and effectively utilizing alkaline solutions. However, this utility is intrinsically linked to a responsibility for safe handling, storage, and disposal, ensuring that the power of alkalinity is harnessed without compromising safety or environmental integrity. The ongoing exploration of new nanomaterials, advanced batteries, and sustainable energy technologies will undoubtedly continue to reveal novel and impactful applications for these fundamental chemical compounds.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.