The seemingly simple act of dissolving sugar in water is a foundational concept in chemistry, but beneath its everyday familiarity lies a complex interplay of molecular forces, energy transfer, and emergent properties. From a technological perspective, understanding this process is not merely an academic exercise; it underpins a vast array of applications, from food processing and pharmaceutical development to advanced materials science and environmental monitoring. Modern technology provides us with the tools to not only observe but also quantify and manipulate these interactions at an unprecedented level, revealing the sophisticated mechanisms at play.
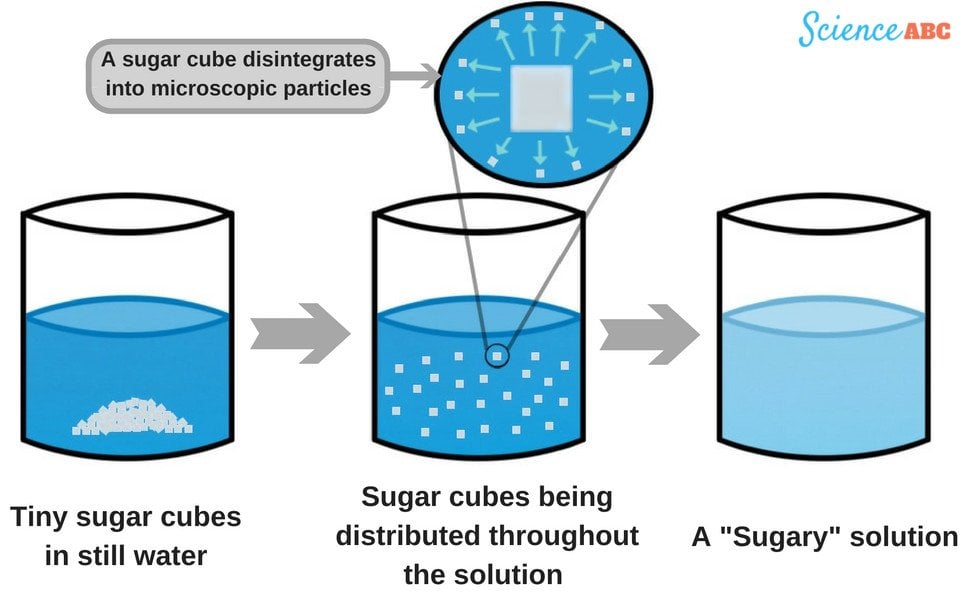
The Molecular Dance: Technology’s Role in Visualizing and Quantifying Interactions
At its core, dissolving sugar in water is a story of molecules. When crystalline sugar (typically sucrose) meets water, a transformative process begins. Technology plays a crucial role in allowing us to visualize, understand, and quantify these molecular interactions, moving beyond simple observation to in-depth analysis.
1. Microscopy and Spectroscopic Techniques: Peering into the Microcosm
The naked eye can only perceive the disappearance of sugar crystals. However, advanced imaging and spectroscopic technologies unveil the detailed events.
High-Resolution Microscopy for Molecular Visualization
Techniques like Atomic Force Microscopy (AFM) and Scanning Tunneling Microscopy (STM) allow scientists to visualize the surface of sugar crystals and water molecules with incredible precision. AFM, for instance, can map the topography of the sugar surface at the atomic level, revealing the crystalline lattice structure of sucrose. As water molecules begin to interact, these microscopes can capture subtle changes, showing how water molecules orient themselves around the sugar molecules. While direct visualization of individual water molecules interacting with sugar at the moment of dissolution is still challenging, these technologies provide powerful insights into the initial stages of interaction and the structural integrity of the crystal.
Spectroscopic Analysis of Chemical Bonds
Spectroscopy is instrumental in understanding the chemical changes and interactions occurring. Infrared (IR) spectroscopy, for example, can identify the vibrational modes of molecules. When sugar dissolves, the hydrogen bonds within the water network are disrupted, and new interactions form between water molecules and the hydroxyl (-OH) and glycosidic bonds of the sugar molecule. IR spectroscopy can detect shifts in these vibrational frequencies, providing evidence of the formation of new hydrogen bonds between water and sugar, and the breaking of existing ones within the sugar crystal and the water network. Raman spectroscopy offers complementary information, further confirming these molecular interactions and providing insights into the structural changes.
2. Computational Modeling and Simulation: Predicting and Understanding Molecular Behavior
Beyond direct observation, computational chemistry and advanced simulation software offer powerful tools for understanding the thermodynamics and kinetics of sugar dissolution.
Molecular Dynamics Simulations
Molecular dynamics (MD) simulations are a cornerstone of modern computational chemistry. These simulations use Newton’s laws of motion to track the positions and velocities of thousands to millions of atoms and molecules over time. By inputting the properties of water and sugar molecules, researchers can simulate the dissolution process at a near-atomic level. These simulations can show how water molecules surround and solvate the sugar molecules, gradually pulling them from the crystal lattice. They can also reveal the energy exchanges involved, such as the endothermic process of breaking sugar-sugar and water-water bonds and the exothermic process of forming sugar-water bonds. The balance of these energy changes determines whether dissolution is favorable.
Quantum Mechanical Calculations
For a deeper understanding of the specific bonding interactions, quantum mechanical (QM) calculations can be employed. These methods, based on the principles of quantum mechanics, provide highly accurate descriptions of electron distribution and bonding energies. QM calculations can predict the strength of hydrogen bonds between water and sugar, and the energy required to break the intermolecular forces holding the sugar crystal together. This theoretical underpinning complements experimental data, allowing for a more comprehensive understanding of the dissolution mechanism.
The Emergent Properties: Technology’s Applications in Exploiting Dissolution
The process of sugar dissolving in water isn’t just a chemical curiosity; it’s a phenomenon harnessed by numerous technological applications across various industries. Understanding the underlying principles through technological analysis allows for precise control and optimization.
1. Food and Beverage Industry: Precision Engineering of Taste and Texture
The food industry is a prime example of how the simple act of dissolving sugar is technologically managed and exploited.
Sweetener Formulation and Delivery Systems
The controlled dissolution of sugars and artificial sweeteners is critical for achieving desired taste profiles and shelf-life stability in beverages, confectionery, and baked goods. Technologies like spray drying and encapsulation are used to create powdered sweeteners that dissolve readily and uniformly, ensuring consistent flavor. Understanding the solubility of different sugars under varying conditions (temperature, pH) allows for the precise formulation of products. Rheology, the study of flow and deformation, is also crucial here. Technological tools allow food scientists to measure the viscosity of sugar solutions, which directly impacts mouthfeel and texture, enabling the creation of products with specific textural characteristics.

Crystallization Control in Food Production
While dissolution is the focus, understanding the reverse process, crystallization, is equally important. Technologies like controlled cooling and seeding are employed in industries like ice cream production to manage sugar crystallization, preventing a gritty texture. The principles of solubility and supersaturation, studied using sophisticated analytical equipment, are directly applied to prevent or encourage crystallization as needed.
2. Pharmaceutical and Biomedical Applications: Drug Delivery and Formulation
The ability of water to dissolve a wide range of substances, including sugars, is fundamental to pharmaceutical development.
Oral Drug Delivery Systems
Many active pharmaceutical ingredients (APIs) are formulated with excipients, including sugars, to improve palatability, stability, and bioavailability. The dissolution rate of these sugars impacts how quickly a tablet disintegrates and releases its medicinal components. Technologies like tablet coating, often involving polymer science and precise application techniques, are designed to control the rate at which water penetrates the tablet and dissolves the sugar excipients. Furthermore, understanding the osmotic properties of sugar solutions (driven by the concentration gradient created by dissolved sugar) is crucial for developing IV solutions and other parenteral drug formulations.
Biomaterials and Tissue Engineering
Biocompatible polymers and hydrogels, often derived from or incorporating sugar-like molecules (polysaccharides), are used in advanced biomedical applications. The ability of these materials to swell and release encapsulated substances is directly related to their interaction with aqueous environments. Technological advancements in materials science allow for the precise engineering of these hydrogels, tuning their solubility and swelling properties to act as controlled release systems for drugs or scaffolds for tissue regeneration.
The Science Behind the Sweetness: Technological Advancements in Understanding Solubility
Solubility is not a static property; it’s influenced by various factors that can be precisely measured and controlled using advanced technological tools. Understanding these influences allows for the optimization of processes in countless applications.
1. Thermochemistry and Calorimetry: Quantifying Energy Transfer
The dissolution of sugar is an endothermic process, meaning it absorbs heat from the surroundings. This energy balance is critical to understanding why sugar dissolves and how much energy is involved.
Isothermal Titration Calorimetry (ITC)
ITC is a powerful technique used to measure the heat absorbed or released during a chemical or physical process, such as dissolution. By precisely measuring the heat flow as sugar is added to water, scientists can quantify the enthalpy of dissolution. This thermodynamic data is vital for understanding the stability of sugar solutions and predicting their behavior under different conditions. It also helps in designing industrial processes where precise temperature control is essential.
Differential Scanning Calorimetry (DSC)
DSC measures the difference in heat flow into a sample and a reference as a function of temperature. While often used for phase transitions, DSC can also provide information about the thermal behavior of sugar solutions, including changes related to hydration and potential supersaturation. This helps in understanding the long-term stability of sugar-containing products and predicting their behavior during storage and processing.
2. Analytical Chemistry Techniques: Measuring Concentration and Purity
Accurate measurement of sugar concentration and purity in solutions is paramount for quality control and process optimization.
High-Performance Liquid Chromatography (HPLC)
HPLC is a widely used analytical technique that separates, identifies, and quantifies components of a mixture. In the context of sugar dissolution, HPLC can be used to verify the complete dissolution of crystalline sugar and to identify and quantify any other dissolved impurities. It’s also essential for analyzing complex mixtures of sugars, such as those found in food products or biological samples.
Refractometry and Polarimetry
These optical techniques are simple yet effective for measuring the concentration of dissolved sugars. Refractometers measure the refractive index of a solution, which changes with the concentration of dissolved solutes. Polarimeters measure the optical rotation of plane-polarized light by chiral molecules like sucrose. Both are widely used in the food and beverage industry for rapid quality control and process monitoring, allowing for real-time adjustments to ensure consistent product quality.
In conclusion, the simple act of sugar dissolving in water is a profound illustration of molecular interactions. Through the lens of technology, we can appreciate the sophisticated tools and techniques that allow us to not only observe but also deeply understand and harness this fundamental chemical process. From the atomic-level visualizations of microscopy to the predictive power of computational simulations and the precise measurements of analytical instruments, technology transforms our understanding of dissolution, enabling innovation and driving progress across a vast spectrum of industries. The “what happens when sugar dissolves in water” is a question whose answer, thanks to technology, continues to reveal ever more intricate and impactful insights.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.