In the pursuit of technological advancement, the fundamental properties of matter often dictate the limits of what we can build. From the transistors in a smartphone to the cooling systems in a quantum computer, the behavior of electrons is the primary currency of the digital age. One of the most critical metrics in understanding this behavior is ionization energy—the amount of energy required to remove an electron from an isolated atom.
When we ask the question, “What element has the largest ionization energy?” the answer is Helium. While this might seem like a simple trivia fact for a chemistry student, in the realms of advanced hardware engineering, semiconductor manufacturing, and aerospace technology, Helium’s extreme atomic stability is a cornerstone of innovation. This article explores why Helium holds this title and how its unique energetic profile drives the most sophisticated tech industries on the planet today.
Understanding Ionization Energy in the Tech Landscape
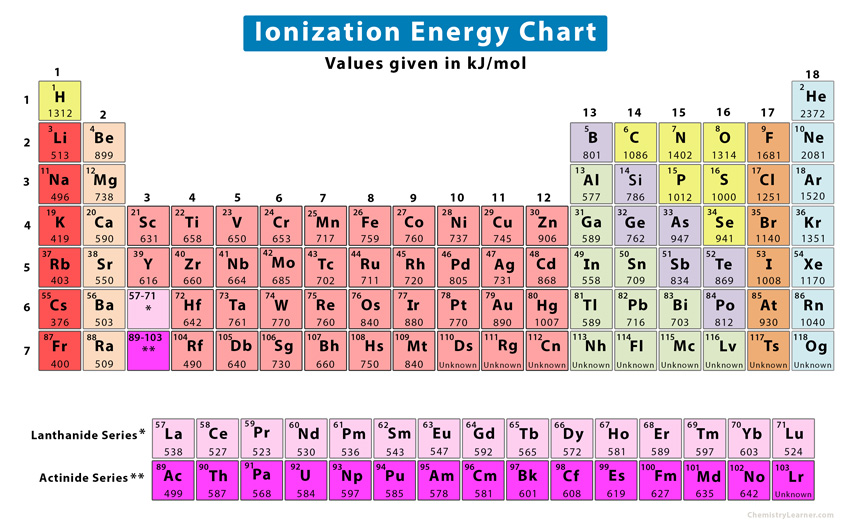
To appreciate why Helium is a technological marvel, we must first understand the physics behind its “ionization energy.” Ionization energy is a measure of an atom’s “grip” on its outermost electrons. The tighter the grip, the more energy is required to strip an electron away, making the element more chemically inert and stable.
Defining the Barrier: What is Ionization Energy?
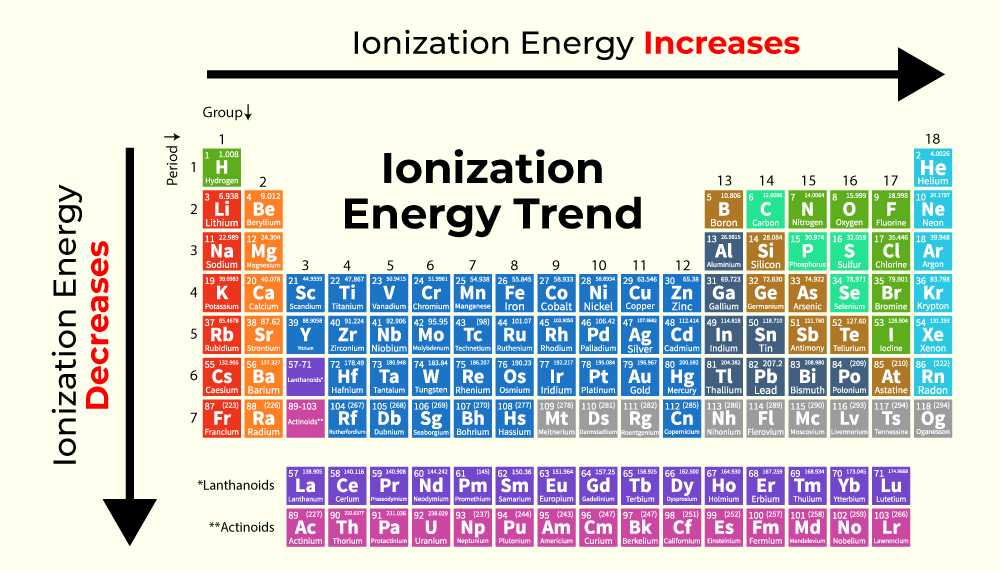
In a vacuum, removing an electron from an atom requires overcoming the electromagnetic pull of the nucleus. This process is essential in creating plasma, managing electrical conductivity, and designing sensors. For most elements, ionization happens relatively easily. However, as we move across the periodic table from left to right, the “effective nuclear charge” increases—meaning the nucleus pulls harder on the electrons. Simultaneously, as we move from bottom to top, the distance between the nucleus and the outer electrons decreases.
Why Helium Takes the Crown
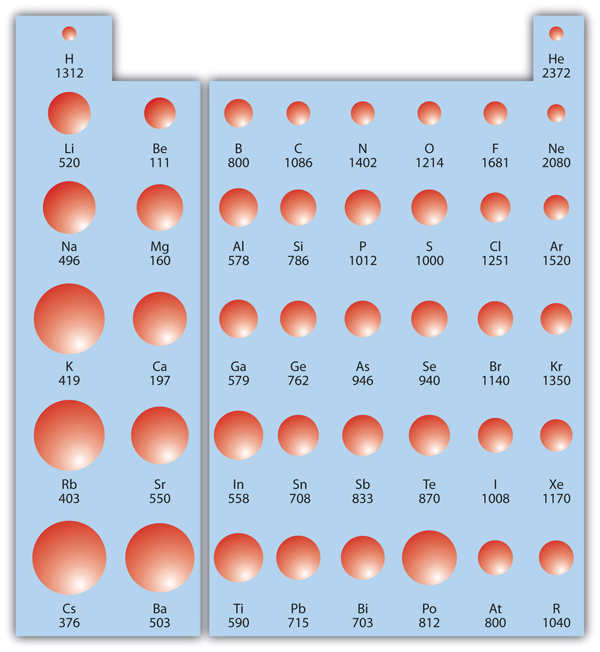
Helium sits at the intersection of these two trends: it is at the very top and the very right of the periodic table (excluding the theoretical properties of some heavier, synthetic elements). With a full 1s shell and two protons exerting a massive relative pull on those electrons, Helium requires 24.59 electron volts (eV) to ionize. This is significantly higher than any other element. For context, Francium, at the other end of the spectrum, requires only 4.07 eV. This massive energy barrier makes Helium the ultimate “non-reactive” tool in a technologist’s arsenal.
Semiconductors and the Physics of Atomic Retention
The semiconductor industry is perhaps the greatest beneficiary of high-ionization energy materials. In the quest to pack billions of transistors onto a single silicon chip, the environment in which these chips are manufactured must be perfectly controlled.
The Role of Noble Gases in Microchip Fabrication
In the process of photolithography—where light is used to etch circuit patterns onto silicon wafers—the presence of reactive gases can lead to oxidation or contamination, rendering the chip useless. Because Helium has the highest ionization energy, it is virtually impossible for it to react with the silicon or the metallic layers under standard manufacturing conditions.
Helium is frequently used as a carrier gas in chemical vapor deposition (CVD) and as a cooling medium during the etching process. Its ability to remain stable while transferring heat away from the wafer allows for the creation of 3nm and 2nm nodes. Without the stability afforded by Helium’s atomic structure, the thermal management of high-precision lithography would be significantly more volatile.
Overcoming Energy Barriers in Transistor Design
Beyond manufacturing, the concept of ionization energy informs the materials science of the transistors themselves. As we reach the physical limits of silicon, engineers are looking at “wide bandgap” semiconductors like Gallium Nitride (GaN) and Silicon Carbide (SiC). The goal is to mimic the high-energy barriers found in noble gases to prevent “leakage current.” By understanding how Helium maintains its electrons under duress, researchers can develop insulating layers that prevent electrons from jumping where they don’t belong, leading to faster and more power-efficient AI processors.
Industrial Applications: From Cryogenics to Fiber Optics
The high ionization energy of Helium is intrinsically linked to its low boiling point and high thermal conductivity. These physical traits make it indispensable in several high-tech sectors that require extreme environments.
Helium as a Mission-Critical Tech Resource
In the world of medical technology and particle physics, Helium is the only element capable of cooling superconducting magnets to temperatures near absolute zero. MRI machines and the Large Hadron Collider rely on liquid Helium to maintain a state of zero electrical resistance. The reason Helium is used is its refusal to interact with the system; its high ionization energy ensures that even under intense electromagnetic fields, the Helium atoms do not become ionized or conductive, which would lead to a catastrophic “quench” or failure of the cooling system.
High-Energy Plasma and Advanced Manufacturing
In the field of aerospace and high-precision welding, Helium-shielded plasma arcs are used to join exotic metals like titanium and aluminum. Because it takes so much energy to ionize Helium, the plasma created in a Helium environment is much hotter and more stable than that created with Argon or Nitrogen. This allows for deeper penetration and cleaner welds in the fuselages of modern spacecraft and high-performance jet engines. The “energy barrier” of the Helium atom essentially acts as a stabilizer for the thermal energy being channeled into the metal.
The Future of Materials Science: Beyond Traditional Ionization
As we look toward the next decade of technology, the principles of atomic stability and ionization energy are being applied to the frontiers of quantum computing and sustainable energy.
Quantum Computing and Atomic Coherence
Quantum computers require an environment of absolute “quiet” to prevent decoherence—the loss of quantum information. Many quantum systems use trapped ions or superconducting circuits. Helium, specifically the isotope Helium-3, is used in dilution refrigerators to reach millikelvin temperatures. The high ionization energy of Helium ensures that it remains a neutral, non-interfering background medium. If the cooling agent were easily ionized, the resulting stray electrons would interfere with the qubits, leading to computational errors.
Developing Next-Generation Superconductors
The tech world is currently obsessed with finding a room-temperature superconductor. To achieve this, scientists are studying how high-pressure environments can force elements to mimic the electronic configuration of Helium. By compressing hydrogen-rich compounds, researchers aim to create a state where electrons are held in a rigid, stable lattice, similar to the stability found in the Helium atom. Understanding the “largest ionization energy” isn’t just about Helium; it’s about a blueprint for stability that we try to replicate in synthetic materials.
Conclusion: The Quiet Giant of the Periodic Table
While Helium is often associated with party balloons and squeaky voices, its status as the element with the largest ionization energy makes it a silent titan of the technology industry. This single physical property—the refusal to give up an electron—enables the creation of the world’s fastest microchips, the most powerful medical imagers, and the most precise aerospace components.
As technology continues to shrink toward the atomic scale, the fundamental “rules” of the periodic table become the boundary lines for innovation. Helium’s extreme energy barrier provides a benchmark for stability, a tool for manufacturing, and a medium for exploration. In a world where we are constantly trying to move electrons faster and more efficiently, the element that holds onto them the tightest remains, paradoxically, our most valuable asset. Helium doesn’t just represent a point on a chart; it represents the upper limit of atomic resilience, a quality that will continue to fuel the tech trends of tomorrow.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.