In the modern healthcare landscape, the intersection of pharmacology and technology has transformed how we understand drug interactions and patient outcomes. Cyclobenzaprine, a widely prescribed skeletal muscle relaxant primarily used for the short-term treatment of muscle spasms associated with acute, painful musculoskeletal conditions, serves as a primary case study for this digital evolution. While the medication is effective, its side effect profile—ranging from mild sedation to complex neurological impacts—presents a unique challenge that modern technology is now equipped to monitor, predict, and mitigate.
As we move deeper into the era of HealthTech, the question is no longer just “what are side effects of cyclobenzaprine,” but rather, “how can digital tools help us manage these side effects more effectively?” Through the use of artificial intelligence (AI), wearable sensors, and big data analytics, the tech industry is redefining the safety parameters of traditional medicine.
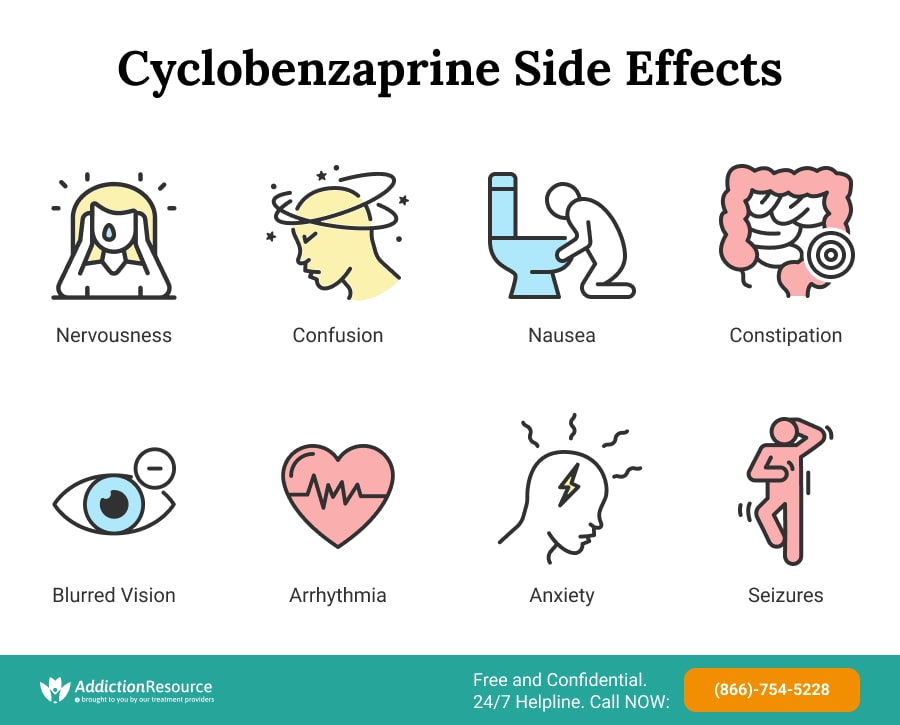
Predictive Analytics: AI’s Role in Mapping Adverse Reactions
The pharmaceutical industry has undergone a massive digital transformation, moving away from reactive monitoring toward proactive, predictive modeling. Artificial intelligence and machine learning (ML) algorithms are now being utilized to map the side effects of cyclobenzaprine before they even manifest in a clinical setting.
Molecular Modeling and Digital Twins
One of the most groundbreaking advancements in tech-driven pharmacology is the creation of “Digital Twins.” A digital twin is a virtual representation of a biological system—in this case, the human nervous system—that allows researchers to simulate how cyclobenzaprine molecules interact with various receptors.
Cyclobenzaprine is structurally related to first-generation tricyclic antidepressants. By using high-performance computing (HPC), tech companies can simulate the drug’s binding affinity to cholinergic and serotonergic receptors. This digital modeling helps predict side effects like xerostomia (dry mouth) and tachycardia (increased heart rate) with high precision. By simulating these interactions digitally, tech developers can provide clinicians with data-backed predictions on which patients are most likely to experience severe adverse reactions based on their unique biological markers.
Data Mining Clinical Trials and Real-World Evidence
Beyond simulations, the tech sector utilizes natural language processing (NLP) to mine vast databases of clinical trial results and electronic health records (EHR). By analyzing millions of data points, AI can identify patterns that a human researcher might miss. For instance, tech platforms can correlate the frequency of “brain fog” or cognitive impairment—common side effects of cyclobenzaprine—with specific age groups or co-medications. This data-driven approach allows for the creation of sophisticated risk-assessment software that alerts physicians in real-time if a patient’s profile suggests a high risk of side effects.
Wearable Tech and Real-Time Bio-Monitoring
While predictive modeling happens in the lab, wearable technology provides the frontline defense against the side effects of cyclobenzaprine in the real world. Cyclobenzaprine’s primary side effect is central nervous system (CNS) depression, which manifests as drowsiness, dizziness, and decreased motor coordination.
Monitoring Sedation via Smartwatches and Keystroke Dynamics
The ubiquitous nature of smartwatches and smartphones has opened new avenues for monitoring the sedative effects of muscle relaxants. Tech firms are developing “digital biomarkers” to track the onset of drowsiness. For example, some apps utilize the smartphone’s gyroscope and accelerometer to detect subtle changes in gait or posture that indicate dizziness—a common side effect of cyclobenzaprine.
Furthermore, keystroke dynamics—the rhythm and timing of typing—can serve as a sensitive measure of cognitive impairment. If a patient’s typing speed slows or error rate increases after taking a dose of cyclobenzaprine, machine learning algorithms can flag this as a level of sedation that makes driving or operating machinery dangerous. This real-time feedback loop represents a significant leap in digital safety.
Bio-Sensors for Detecting Ataxia and Motor Impairment
For patients recovering from severe musculoskeletal injuries, wearable bio-sensors can track muscle activity (EMG) and limb coordination. Since cyclobenzaprine acts on the brain stem to reduce tonic somatic motor activity, excessive dosing can lead to ataxia or significant muscle weakness. Advanced wearable patches now transmit data directly to a cloud-based platform where software analyzes the movement patterns. If the software detects a high risk of falls or impaired motor control, it can send an automated notification to both the patient and their healthcare provider, mitigating the risk of accidental injury.
Digital Health Platforms and Patient Safety Software

The ecosystem of digital health extends beyond hardware. Software solutions, ranging from telemedicine platforms to interactive pharmacopeias, play a critical role in educating patients about the side effects of cyclobenzaprine and ensuring medication adherence.
Telemedicine and Remote Patient Monitoring (RPM)
Telemedicine has become a staple of modern healthcare, but its true power lies in Remote Patient Monitoring (RPM). Through integrated RPM software, patients prescribed cyclobenzaprine can log their daily symptoms through a secure interface. These platforms often use “symptom checkers” powered by decision-tree logic to categorize side effects.
If a patient logs “blurred vision” or “urinary retention”—side effects that require medical attention—the software can automatically escalate the case to a human practitioner. This tech-enabled oversight ensures that the side effects of cyclobenzaprine are managed within a closed-loop system, reducing the likelihood of emergency room visits and improving the overall user experience of the healthcare system.
Interactive Pharmacopeias and Interaction Checkers
The risk of side effects is significantly heightened when cyclobenzaprine is mixed with other substances, such as alcohol or CNS depressants. Modern drug-interaction APIs (Application Programming Interfaces) are now integrated into most pharmacy software and consumer health apps. These digital tools allow users to scan a medication bottle and receive an instant, tech-driven report on potential side effects.
By using advanced database management systems, these apps cross-reference the chemical structure of cyclobenzaprine against thousands of other compounds. This digital vigilance prevents dangerous interactions, such as serotonin syndrome when combined with certain antidepressants, by providing the user with clear, actionable warnings via their mobile device.
The Future of Personalized Medicine: Genomics and Blockchain
Looking toward the future, the tech industry is pushing the boundaries of how we handle drug safety through the lenses of personalized medicine and secure data management.
Pharmacogenomics: Tech-Driven Custom Dosing
The “one size fits all” approach to medication is being replaced by pharmacogenomics, a field heavily reliant on DNA sequencing technology and bioinformatics. Different people metabolize cyclobenzaprine at different rates based on the CYP450 enzyme system in the liver.
Tech platforms now exist that allow patients to upload their genetic data (from services like 23andMe or clinical labs) to receive a personalized risk profile. If a patient is a “poor metabolizer” of the drug, the software can predict that they will experience much more intense and prolonged side effects, such as extreme fatigue or dry mouth. This tech-driven personalization allows for precision dosing, where the software suggests the optimal dose to maximize relief while minimizing the adverse effects.
Blockchain for Supply Chain Integrity and Counterfeit Prevention
Side effects can sometimes be exacerbated by poor drug quality or counterfeit medications. Blockchain technology is increasingly being used in the pharmaceutical supply chain to ensure the integrity of drugs like cyclobenzaprine. By creating an immutable ledger of every transaction and shipment, blockchain ensures that the medication the patient receives is exactly what the manufacturer produced.
This technological layer of security prevents the introduction of impurities or incorrect dosages that could lead to unexpected or severe side effects. For the consumer, this means that the “side effects” they experience are limited to the drug itself and are not the result of manufacturing errors or digital tampering within the supply chain.

Conclusion: The Synergy of Tech and Pharmacology
Understanding the side effects of cyclobenzaprine is no longer a purely medical endeavor; it is a technological challenge. Through the integration of AI-driven predictive modeling, real-time wearable monitoring, and personalized genomic software, the tech industry is providing the tools necessary to make muscle relaxant therapy safer than ever before.
As digital health continues to evolve, the synergy between software and medicine will only deepen. By leveraging these tech trends, we can transform the patient experience—moving from a state of uncertainty regarding drug side effects to a state of data-driven confidence. The future of pharmacological safety is digital, and the tech tools we build today are the key to a healthier, more informed tomorrow.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.