In the complex intersection of federal regulation and financial markets, few classifications carry as much weight as the Controlled Substances Act (CSA). For the savvy investor, entrepreneur, or financial analyst, understanding the nuances of drug scheduling is not merely a matter of legal compliance—it is a matter of calculating market valuations, tax liabilities, and capital flow. Specifically, the classification of “Schedule III drugs” has recently emerged as a focal point for institutional investors and venture capitalists.
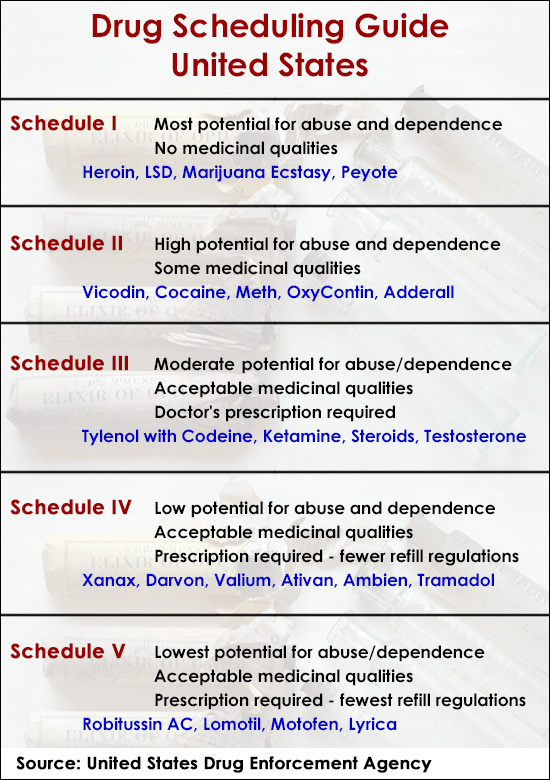
When a substance is categorized under Schedule III, it is defined as having a moderate to low potential for physical and psychological dependence. While the medical community focuses on the therapeutic implications, the “Money” sector focuses on the economic unlocking that occurs when a substance moves from the highly restrictive Schedule I or II into the more commercially viable Schedule III. This shift represents a fundamental change in business finance, tax treatment, and investment risk profiles.
Understanding Schedule III from a Financial Regulatory Framework
From a financial perspective, the classification of a drug dictates the size of its addressable market and the cost of bringing it to that market. Schedule III drugs—which include substances like ketamine, anabolic steroids, and certain codeine mixtures—occupy a “sweet spot” in the regulatory landscape. They are strictly regulated enough to maintain high barriers to entry (protecting established players), yet accessible enough to allow for traditional commercial banking and standardized pharmaceutical distribution.
The Legal Definition and Market Stability
For a business to operate within the Schedule III space, it must adhere to stringent DEA registration and record-keeping requirements. To an investor, these requirements function as a form of “regulatory moat.” Unlike the unregulated supplement market, the Schedule III market is defined by high compliance costs that prevent “fly-by-night” operations from diluting market share. This creates a more stable environment for long-term capital allocation. Businesses dealing in these substances often have predictable cash flows because their products are integrated into the formal healthcare system, backed by insurance reimbursement and institutional procurement.
Section 280E: The Massive Tax Implication for Businesses
The most significant financial driver behind the interest in Schedule III is Internal Revenue Code Section 280E. Historically, businesses involved in the “trafficking” of Schedule I or II substances are prohibited from deducting ordinary business expenses—such as rent, payroll, and marketing—from their federal taxes. They are taxed on their gross profit rather than their net income, leading to effective tax rates that can exceed 70% or 80%.
Moving a substance to Schedule III effectively renders Section 280E inapplicable. This transition is a massive financial catalyst; it can instantly transform a cash-strapped, struggling enterprise into a highly profitable one without changing a single operational detail. For private equity firms and retail investors, the removal of the 280E tax burden is the single most important metric when valuing companies in the pharmaceutical and emerging cannabis sectors.
Investment Opportunities in the Shift to Schedule III
The transition of substances into the Schedule III category often signals a “green light” for institutional capital that was previously sidelined by risk management policies. When a drug is categorized as Schedule III, it simplifies the clinical trial process and reduces the cost of Research and Development (R&D), making the underlying companies more attractive for mergers and acquisitions (M&A).
Biotech and Pharmaceutical R&D Funding
Schedule III status facilitates a more streamlined pathway for FDA approval. For biotech investors, this means a shorter “time to liquidity.” Because Schedule III substances are recognized as having an accepted medical use, the bureaucratic hurdles for conducting human trials are significantly lower than those for Schedule I substances. This allows venture capital firms to model their ROI with higher precision. We are currently seeing a surge in funding for companies developing synthetic versions of Schedule III compounds, as the intellectual property (IP) protections combined with the easier regulatory path offer a lucrative risk-reward ratio.
Publicly Traded Companies and Market Volatility
The announcement or even the rumor of a drug being rescheduled to Schedule III often leads to significant “alpha” in the stock market. Small-cap pharmaceutical companies and ancillary service providers frequently see double-digit growth in their share prices upon news of rescheduling. However, the sophisticated investor must look beyond the initial hype. The real value lies in the “uplisting” potential. Many companies operating in the grey areas of the law (such as those in the cannabis industry) cannot list on major exchanges like the NYSE or NASDAQ due to federal restrictions. A shift to Schedule III removes many of these barriers, allowing for greater liquidity and the entry of “big money” institutional players like BlackRock or Vanguard.
The Cannabis Pivot: A Multi-Billion Dollar Financial Shift
Perhaps the most timely discussion regarding Schedule III drugs involves the United States Department of Health and Human Services (HHS) recommendation to move marijuana from Schedule I to Schedule III. This move is not just a social shift; it is a seismic event in the world of business finance.
Institutional Capital Inflow and Banking Access
Currently, many state-legal cannabis businesses are forced to operate in cash or through high-fee, “workaround” banking solutions. This creates massive operational inefficiencies and security risks. Reclassification to Schedule III would signal to major commercial banks (such as JPMorgan Chase or Bank of America) that they can provide traditional commercial loans, lines of credit, and merchant processing without fear of federal money laundering charges. For the business owner, this means a lower “Cost of Capital.” Instead of relying on predatory high-interest private loans, they can access the same financial tools available to any other legitimate industry.
Valuation Metrics for Emerging Markets
When analyzing the financial health of companies poised to benefit from Schedule III rescheduling, analysts shift their focus from “Price-to-Sales” to “EBITDA-driven” valuations. In a pre-Schedule III environment, “Sales” is the only reliable metric because “Profit” is cannibalized by the 280E tax code. Post-rescheduling, the “Earnings” portion of the “Price-to-Earnings” (P/E) ratio becomes the dominant factor. Investors are currently looking for “undervalued” companies with strong top-line revenue that are currently reporting net losses due to tax inefficiencies, betting that a Schedule III move will instantly flip those losses into significant gains.
Risk Management and Long-Term Financial Outlook
While the financial prospects of Schedule III drugs are enticing, professional wealth management requires a sober assessment of the risks. Regulation is a double-edged sword; while it provides a framework for growth, it also imposes a heavy burden of oversight.
Regulatory Compliance Costs
The move to Schedule III does not mean a “free-for-all.” In fact, it brings businesses under the direct supervision of the DEA and the FDA in a way they may not be prepared for. Companies must invest heavily in compliance software, specialized legal counsel, and rigorous supply chain auditing. From an investment standpoint, the “winners” in this niche will not necessarily be the companies with the best product, but the companies with the most robust administrative infrastructure. When performing due diligence, investors must scrutinize a company’s compliance department as closely as its sales figures.

Diversifying Portfolios in Controlled Substance Sectors
For the individual investor looking to capitalize on the Schedule III trend, diversification is essential. Because the rescheduling process is inherently political and subject to the whims of changing administrations, “regulatory risk” is the highest threat to the portfolio. A sound financial strategy involves balancing direct equity in pharmaceutical producers with investments in “ancillary” players—companies that provide the software, security, packaging, and real estate necessary for the Schedule III economy to function. These “picks and shovels” plays often offer a more stable return on investment, as they benefit from the overall growth of the sector regardless of which specific drug manufacturer dominates the market.
In conclusion, the classification of Schedule III drugs is a cornerstone of modern financial planning within the healthcare and pharmaceutical sectors. Whether it is the massive tax relief provided by the bypassing of Section 280E, the opening of institutional banking channels, or the surge in M&A activity within the biotech space, Schedule III status is a powerful driver of valuation. For those who understand the financial mechanics behind the law, the transition of substances into this category represents one of the most significant wealth-generation opportunities of the decade.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.