In the realm of automotive technology, few components are as misunderstood or as vital as the catalytic converter—often colloquially and incorrectly referred to as a “cadillac converter.” While the name might evoke the luxury of a specific vehicle brand, the device itself is a masterwork of chemical engineering and material science that serves as the primary defense against vehicle pollution. Since its mandatory integration into the automotive landscape in the 1970s, the catalytic converter has evolved from a simple mechanical filter into a sophisticated tech-driven system that works in tandem with modern onboard computers to ensure internal combustion engines operate as cleanly as possible.
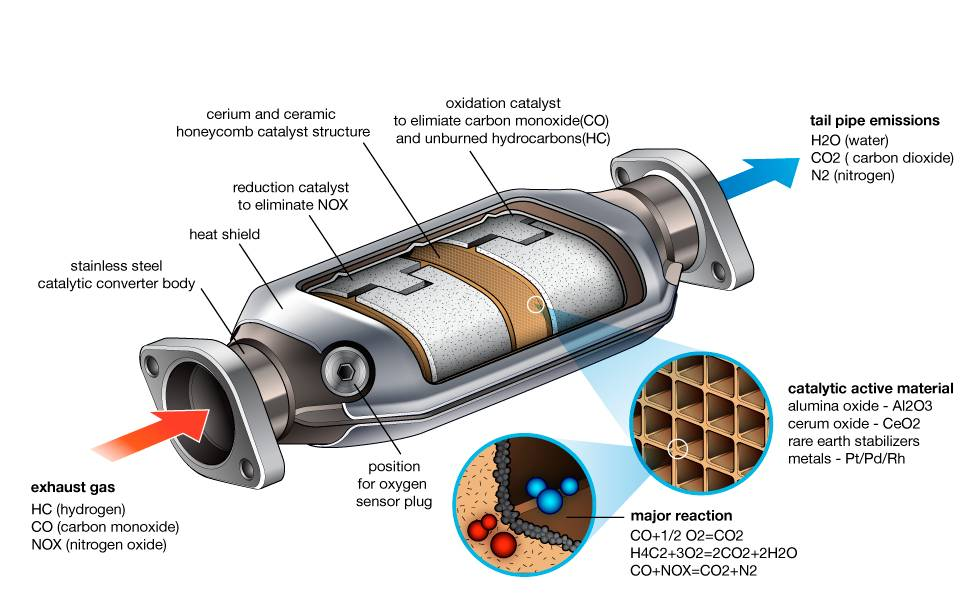
To understand what this component does for a car is to dive into a world of high-temperature chemistry, rare earth metals, and real-time data processing. This article explores the technical intricacies of the catalytic converter, the materials that make its reactions possible, and how it integrates with contemporary vehicle software to maintain performance and environmental standards.
The Chemical Engine: How Catalytic Converters Redefine Vehicle Emissions
At its core, a catalytic converter is an after-treatment system. It does not change how the engine creates power; rather, it changes the chemical composition of the “waste” generated by the combustion process. To the untrained eye, it looks like a simple metal canister integrated into the exhaust system, but internally, it is a high-tech laboratory.
The Core Structure: Honeycombs and Precious Metals
The internal architecture of a modern converter is designed for maximum surface area. Most units utilize a ceramic monolith with a honeycomb structure. This design ensures that as exhaust gases flow through the device, they come into maximum contact with the “washcoat.” This washcoat is a layer of precious metals—primarily platinum, palladium, and rhodium.
These metals act as the “catalyst.” In chemistry, a catalyst is a substance that initiates or accelerates a chemical reaction without being consumed itself. In the context of automotive tech, these metals allow toxic gases to transform into less harmful substances at lower temperatures than would otherwise be required. The honeycomb structure is a marvel of spatial efficiency, providing thousands of tiny channels that force the gas to interact with the catalyst-coated walls.
Redox Reactions: The Science of Cleaning Exhaust
Modern vehicles typically use a “three-way” catalytic converter, named because it addresses the three main pollutants found in exhaust: nitrogen oxides (NOx), carbon monoxide (CO), and unburnt hydrocarbons (HC). The process happens in two distinct stages:
- Reduction: This is the first stage of the converter’s tech cycle. It uses platinum and rhodium to reduce nitrogen oxides. When a NOx molecule contacts the catalyst, the metal atoms pull the nitrogen atom out of the molecule, holding it and releasing the oxygen. The nitrogen atoms then bond with other nitrogen atoms to form N2 (nitrogen gas), which makes up about 78% of the air we breathe.
- Oxidation: In the second stage, platinum and palladium are used to oxidize the remaining carbon monoxide and hydrocarbons. Oxygen is introduced to turn CO into CO2 (carbon dioxide) and to turn unburnt fuel into water vapor and CO2.
Through these precise chemical reactions, the “tech” inside the converter effectively neutralizes the most hazardous byproducts of the internal combustion engine.
Evolutionary Milestones in Automotive Tech
The journey of the catalytic converter is a testament to the rapid advancement of automotive technology. When it was first introduced, it was a relatively “dumb” component—a passive filter that worked until it eventually clogged or burned out. Today, it is part of a highly reactive, software-controlled ecosystem.
From the 1970s to Today: Digital Sensors and Feedback Loops
The biggest technological leap in emission control was the transition from passive converters to “closed-loop” systems. Early versions often struggled because they could not adapt to changing engine conditions. If an engine ran too “rich” (too much fuel), the converter would overheat. If it ran too “lean” (too much air), the reduction process would fail.
Modern tech solved this through the implementation of Oxygen (O2) sensors. These sensors are located both before and after the catalytic converter. They act as data collection points, feeding real-time information to the car’s computer. This feedback loop allows the vehicle to adjust the fuel-air mixture thousands of times per minute, ensuring the converter is always operating within its “sweet spot” of efficiency.
Integration with Electronic Control Units (ECUs)
The catalytic converter is no longer an isolated part; it is a critical input for the Electronic Control Unit (ECU). The ECU uses the data from the converter’s sensors to monitor engine health. If the software detects that the conversion efficiency has dropped below a certain threshold, it triggers a “Check Engine” light (MIL). This digital oversight ensures that the hardware is functioning correctly and alerts the driver to potential tech failures long before they result in a breakdown.

The Material Science Challenge: Rare Earth Elements and Sustainability
The “tech” of the catalytic converter is inseparable from the materials that power it. The reliance on platinum-group metals (PGMs) creates a unique challenge in the automotive industry. These metals are chosen not for their aesthetic value, but for their specific electronic configurations that allow for the “adsorption” of gas molecules.
Platinum, Palladium, and Rhodium: The Tech Specs
Each of these metals serves a specific technological function. Platinum is highly versatile, acting as both an oxidation and reduction catalyst. Palladium is exceptionally efficient at handling high-heat oxidation, making it ideal for modern, high-performance engines. Rhodium is the most effective at reducing nitrogen oxides, which are the primary cause of smog.
The engineering challenge lies in the “loading” of these metals. Automotive engineers must determine the exact milligrams of each metal required to meet emission standards over the 100,000 to 150,000-mile lifespan of a vehicle. This requires advanced modeling software to predict how the metals will degrade over time when exposed to sulfur, lead, and high-temperature thermal cycling.
Smart Alternatives and Synthetic Innovations
As the tech industry moves toward more sustainable solutions, researchers are looking for ways to reduce the “loading” of these expensive metals. New innovations include the use of nanotechnology to create smaller, more reactive clusters of catalyst particles. By increasing the effective surface area at a molecular level, engineers can achieve the same level of emission scrubbing with a fraction of the precious metal content. This shift represents a significant trend in green tech: doing more with less through atomic-level precision.
Diagnostic Tech and Maintenance Optimization
For the modern tech-savvy car owner, the catalytic converter is a focal point for diagnostics. Because the converter is the “end of the line” for the engine’s combustion process, its performance is often the best indicator of overall engine health.
Utilizing OBD-II Scanners for Performance Tracking
The On-Board Diagnostics II (OBD-II) system is the primary tool for interacting with the catalytic converter’s tech. By using a diagnostic scanner, a technician or enthusiast can view “Live Data” streams. This includes “Fuel Trims”—the adjustments the ECU makes to the fuel mixture—and O2 sensor voltages.
In a healthy system, the upstream O2 sensor should fluctuate rapidly, while the downstream sensor (the one after the converter) should remain relatively steady. If the downstream sensor begins to mimic the upstream sensor, the diagnostic software knows the “catalytic tech” has failed. This level of granular data allows for precision maintenance, moving away from the “guess and check” mechanics of the past.
Predictive Maintenance and Software-Led Diagnostics
Emerging automotive software is moving toward predictive maintenance. By analyzing the rate of degradation in the converter’s efficiency over time, advanced algorithms can predict when a part is likely to fail. This tech allows fleet managers and individual owners to address minor engine issues—such as a misfire or a faulty fuel injector—before they “poison” the expensive catalytic converter, saving the overall health of the vehicle’s tech ecosystem.
The Future of Emission Control in a Hybrid and Electric World
As the automotive industry pivots toward electrification, the role of the catalytic converter is shifting. While Battery Electric Vehicles (BEVs) do not require them, the vast majority of vehicles produced globally over the next decade will be Hybrids (HEVs) or Plug-in Hybrids (PHEVs).
Transitioning from Internal Combustion to Green Energy
In a hybrid vehicle, the catalytic converter faces a new technological hurdle: the “cold start” problem. Because the internal combustion engine in a hybrid cycles on and off, the catalytic converter often cools down below its “light-off” temperature (the temperature at which the chemical reactions begin).
To solve this, automotive engineers are developing electrically heated catalytic converters. These units use a small amount of battery power to pre-heat the catalyst before the engine even starts. This integration of electrical engineering with chemical filtration represents the next frontier in exhaust tech, ensuring that even in a hybrid configuration, emissions are kept to a digital minimum.
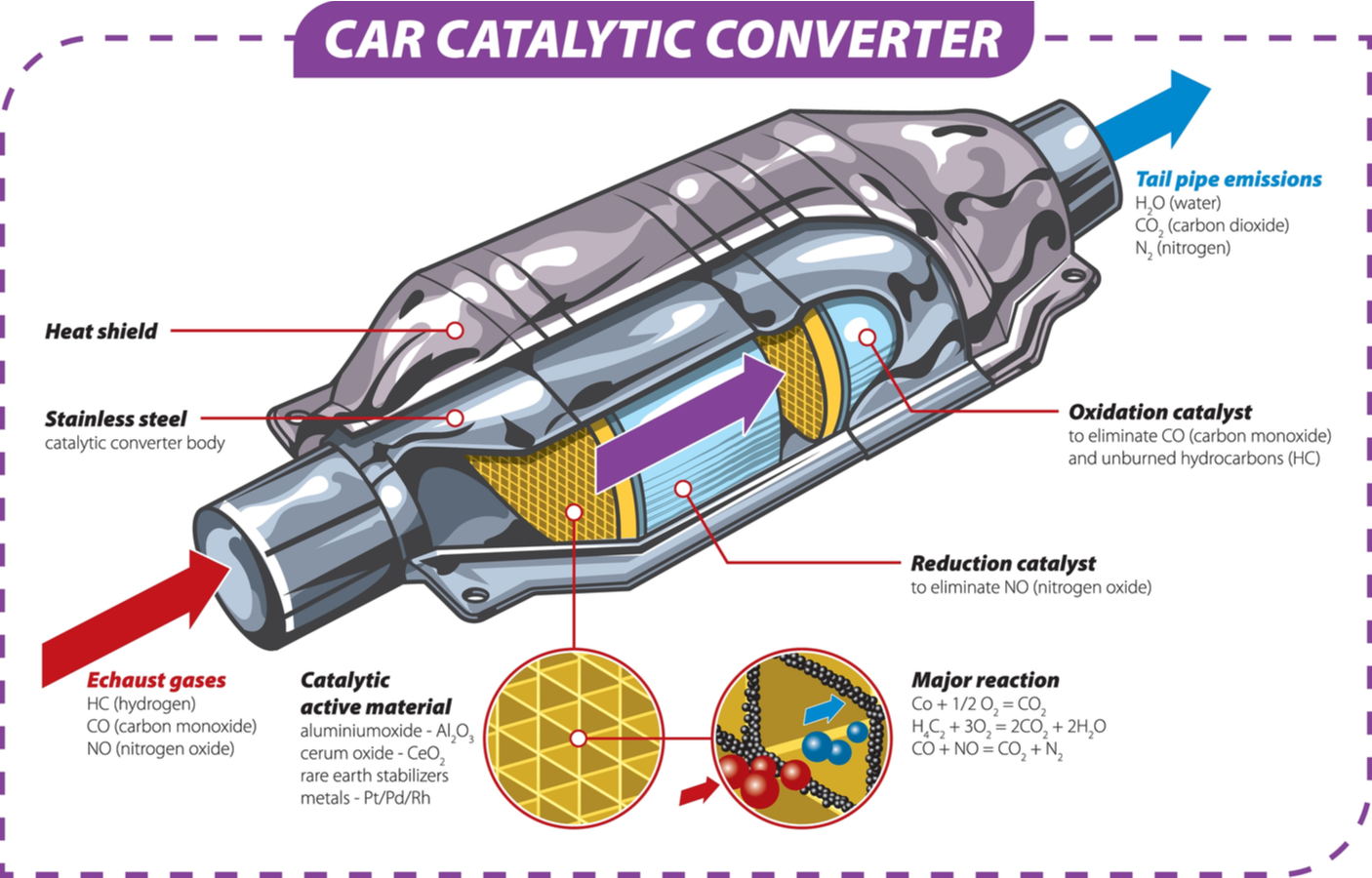
Conclusion
The “cadillac converter”—properly known as the catalytic converter—is much more than a component; it is the pinnacle of automotive environmental technology. From the complex honeycomb structures and precious metal washcoats to the sophisticated O2 sensor feedback loops and ECU integration, it represents a massive investment in precision engineering. As we move forward, the technology within these devices will continue to refine, proving that even as we transition to new energy sources, the innovations born from the internal combustion era remain foundational to our technological progress. Understanding this part is essential for anyone interested in the intersection of hardware, software, and the science of the modern automobile.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.