Mitosis, the fundamental process of cell division, lies at the very heart of life on Earth. From the simplest single-celled organisms to complex multicellular beings, this intricate biological dance ensures growth, repair, and reproduction. While seemingly a pure biology topic, understanding “what type of cells does mitosis create” is not merely academic; it is a critical cornerstone for innovation across a myriad of technological fields, driving progress in medicine, biotechnology, computational science, and even artificial intelligence. This exploration delves into how technology both deciphers and harnesses the power of mitotic cell creation, transforming our ability to heal, grow, and understand life itself.
The Core Biological Engine Driving Biotechnological Progress
At its essence, mitosis is the cellular mechanism for producing two genetically identical daughter cells from a single parent cell. This precise duplication is responsible for the proliferation of somatic cells, which constitute virtually all the non-reproductive cells in an organism. From a technological standpoint, this foundational process represents an exquisitely engineered system of self-replication and repair that biotechnologists strive to understand, mimic, and control.
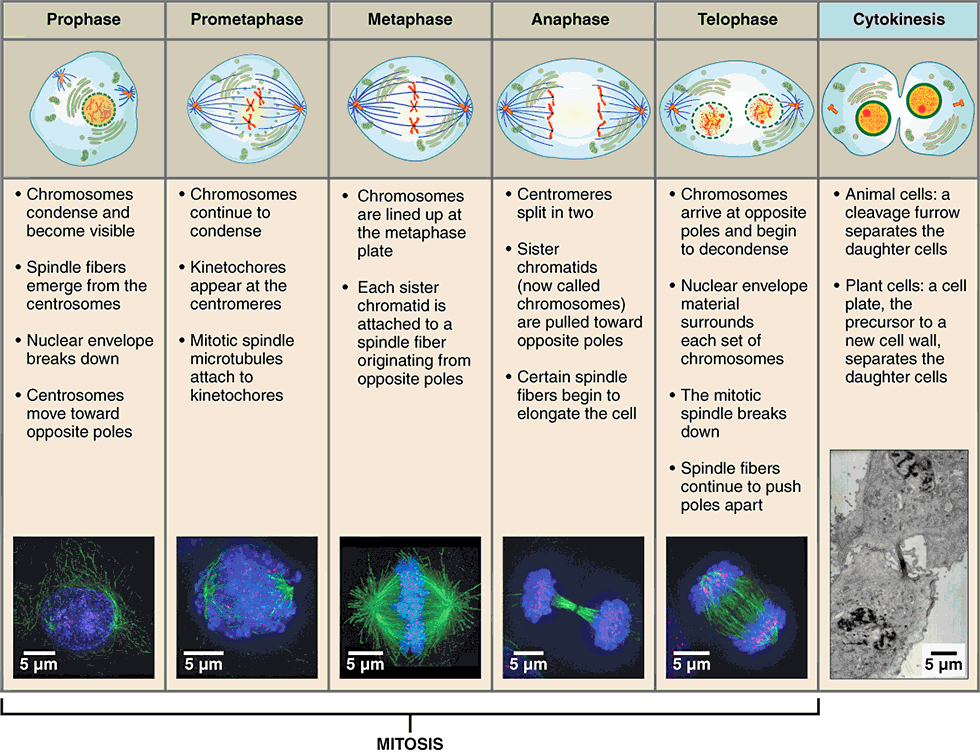
Mitosis Explained: The Blueprint for Cellular Duplication
The mitotic cell cycle is a tightly regulated series of events, including DNA replication (during interphase) followed by nuclear division (mitosis) and cytoplasmic division (cytokinesis). Each stage—prophase, metaphase, anaphase, and telophase—involves complex molecular machinery and precise choreography, ensuring that each daughter cell receives a complete and identical set of chromosomes. The flawless execution of this process is paramount for healthy tissue function and development. Any deviation can lead to severe consequences, such as developmental disorders or uncontrolled cell proliferation characteristic of cancer. For technology, this precision offers a blueprint for building robust biological systems and identifying vulnerabilities for therapeutic intervention. Modern microscopy, advanced imaging techniques, and genomic sequencing technologies are constantly refining our understanding of these intricate stages, offering unprecedented detail into the molecular mechanisms at play.
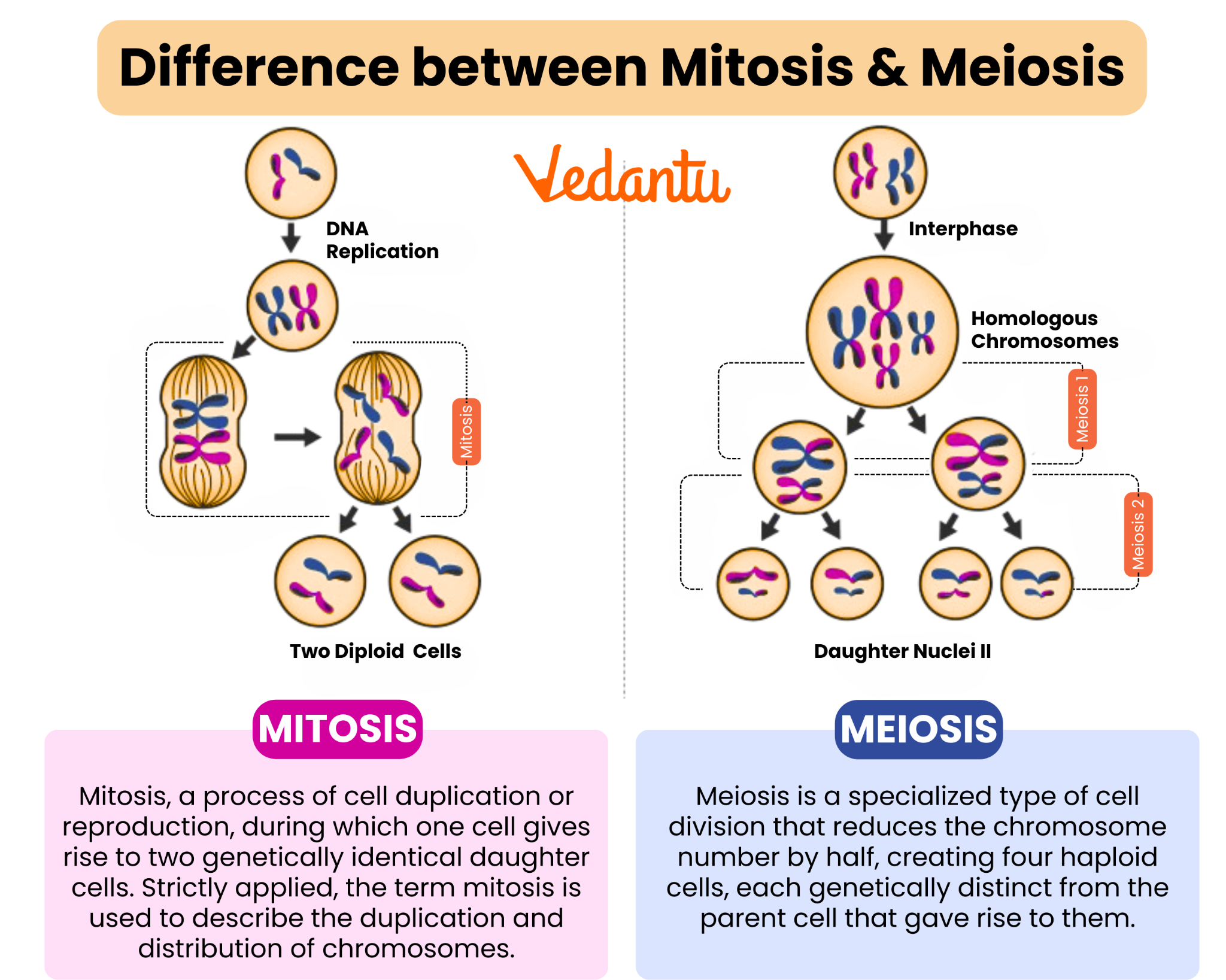
The Product of Mitosis: Somatic Cells and Their Diverse Roles
When we ask “what type of cells does mitosis create,” the primary answer is somatic cells. These are the body cells that make up tissues, organs, and systems—skin cells, muscle cells, nerve cells, blood cells (except for germline stem cells), bone cells, and countless others. These cells are diploid, meaning they contain two complete sets of chromosomes, one from each parent. The creation of these identical somatic cells is crucial for:
- Growth: An organism grows by increasing the number of its cells through mitosis.
- Repair and Regeneration: Damaged or worn-out cells are replaced by new ones generated through mitosis, maintaining tissue integrity.
- Asexual Reproduction: In some organisms, mitosis is the primary mode of reproduction.
The technological implications here are profound. If we can precisely control the mitotic creation of specific somatic cell types, we unlock pathways for regenerative medicine, drug screening, and even the creation of artificial tissues. Biotechnology platforms are built upon this very principle, aiming to direct cellular replication for therapeutic gain.
Leveraging Mitosis in Regenerative Medicine and Tissue Engineering
The ability of mitosis to generate vast quantities of specific cell types from a single starting cell is the cornerstone of modern regenerative medicine and tissue engineering. These fields are dedicated to repairing or replacing damaged tissues and organs, offering hope for countless debilitating diseases and injuries.
Cultivating Tissues and Organs: Mitosis in Vitro
The promise of growing functional human tissues and even whole organs in a lab environment relies heavily on controlling mitosis. Stem cells, with their remarkable capacity for self-renewal (through mitosis) and differentiation into various specialized cell types, are central to this endeavor. Technologies like 3D bioprinting leverage computer-aided design and specialized bio-inks containing living cells to precisely deposit cells layer by layer, mimicking the natural tissue architecture. These bioprinted constructs rely on the embedded cells undergoing mitosis to proliferate, mature, and integrate, ultimately forming functional tissues such as cartilage, skin grafts, or even miniature organoids that serve as disease models for drug discovery. High-throughput screening platforms are then used to test thousands of compounds on these lab-grown tissues, vastly accelerating pharmaceutical development.
Gene Editing and Cellular Therapies: Directing Mitotic Outcomes
Advanced gene editing tools, notably CRISPR-Cas9, allow scientists to make precise modifications to the DNA within cells. These modifications can correct genetic defects, enhance cellular function, or program cells to perform new tasks. When these edited cells undergo mitosis, the corrected or enhanced genetic information is faithfully passed on to all daughter cells. This has revolutionized cellular therapies, such as CAR-T cell therapy, where a patient’s own immune cells are genetically modified to better target cancer cells, then expanded through controlled mitosis before being reinfused into the patient. Furthermore, researchers are exploring how to precisely regulate the cell cycle and mitotic division through genetic manipulation to halt uncontrolled proliferation in cancer or stimulate regeneration in damaged organs.
Computational Biology and AI: Unveiling Mitotic Mechanisms

The complexity of mitosis, involving thousands of interacting proteins and signaling pathways, presents a perfect challenge for computational biology and artificial intelligence. These technologies are not just observing mitosis; they are helping us to understand its intricacies, predict its behavior, and even simulate its processes.
Predictive Modeling of Cell Division: From Disease Progression to Drug Efficacy
Computational models, ranging from agent-based simulations to complex network analyses, are being developed to map the entire mitotic process. These models integrate vast datasets from genomics, proteomics, and live-cell imaging to predict how mutations or drug treatments might affect cell division, cellular fate, and tissue dynamics. For instance, in cancer research, predictive models can simulate how different chemotherapy agents interfere with mitotic checkpoints, helping to optimize drug combinations and dosage. These simulations significantly reduce the need for extensive, time-consuming, and costly wet-lab experiments, accelerating drug discovery and personalized medicine strategies.
AI-Driven Image Analysis: Accelerating Mitosis Research and Diagnostics
AI, particularly deep learning algorithms, has transformed how we analyze microscopic images of dividing cells. Traditionally, identifying and classifying cells in different mitotic stages was a labor-intensive and subjective task performed by human experts. AI-powered image analysis platforms can now automatically detect mitotic cells, track their movement, quantify cell division rates, and even identify subtle morphological abnormalities indicative of disease, often with greater accuracy and speed than humans. This capability is invaluable in drug screening, diagnostics (e.g., assessing tumor aggressiveness), and fundamental research into cell cycle regulation, enabling high-throughput analysis that was previously unimaginable.
Pharmaceutical Innovation and Targeted Therapies
The precise regulation of mitosis is fundamental to healthy cellular function, making its dysregulation a critical factor in numerous diseases, most notably cancer. Technological advancements in pharmacology and drug development are heavily focused on understanding and manipulating mitotic pathways.
Understanding Uncontrolled Mitosis: The Battle Against Cancer
Cancer is fundamentally a disease of uncontrolled cell proliferation, where cells bypass the normal regulatory checkpoints of the cell cycle and divide relentlessly through mitosis. Many chemotherapeutic agents work by directly or indirectly targeting components of the mitotic machinery, aiming to halt cell division in rapidly proliferating cancer cells. Technologies such as high-throughput drug screening platforms allow pharmaceutical companies to test millions of compounds for their ability to interfere with mitotic proteins or disrupt the cell cycle in cancer cells. This involves advanced robotics, automated microscopy, and bioinformatics to identify promising drug candidates that selectively inhibit cancerous mitosis while sparing healthy cells, thus reducing side effects.
Developing Precision Medicine: Tailoring Treatments Based on Cellular Replication
The advent of precision medicine relies on understanding the specific genetic and molecular characteristics of a patient’s disease, including aberrations in mitotic regulation. Next-generation sequencing (NGS) technologies, alongside advanced proteomic analyses, allow clinicians to identify specific mutations in genes that control the cell cycle (e.g., oncogenes and tumor suppressor genes). This technological capability enables the development of targeted therapies that specifically inhibit the mutated mitotic pathways in cancer cells, rather than indiscriminately destroying all dividing cells. For example, drugs that inhibit specific kinases involved in mitotic progression (e.g., aurora kinase inhibitors) are being developed and tested, representing a sophisticated technological approach to selectively disrupt the creation of cancerous cells.
Ethical Considerations and Future Horizons in Mitotic Technologies
As our technological prowess in understanding and manipulating mitosis grows, so too do the ethical considerations and the vast potential for future breakthroughs. The ability to control cell creation at will opens doors to unprecedented biological engineering.
Navigating the Bioethical Landscape of Artificial Cellular Creation
The manipulation of mitosis, especially in the context of stem cells, gene editing, and reproductive technologies, raises significant ethical questions. The creation of human embryos for research (though not directly mitosis-driven, the subsequent cellular expansion is), germline editing that affects future generations, and the potential for “designer babies” necessitate careful societal and ethical deliberation. Technology provides the tools, but humanity must guide their responsible application, ensuring that the pursuit of scientific advancement aligns with moral and societal values. International guidelines and regulatory frameworks are constantly evolving to address these complex challenges.
The Promise of Cellular Programming and Enhanced Lifespans
Looking ahead, the technological mastery of mitosis promises revolutionary advancements. Imagine programmable cells that can precisely sense and respond to their environment, initiating mitosis only when repair is needed, or ceasing division when a tumor begins to form. Technologies like synthetic biology aim to engineer entirely new biological systems or re-program existing ones to achieve novel functions. Further understanding of cellular aging and the Hayflick limit (the finite number of times a normal human somatic cell population can divide before senescence) could lead to interventions that extend healthy human lifespans, by modulating mitotic frequency or protecting telomeres. While these horizons are distant, the ongoing technological advancements in microscopy, genomics, bioinformatics, and automation are steadily bringing us closer to a future where we can precisely control what type of cells mitosis creates, and for what purpose, ushering in an era of unprecedented biological engineering.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.