Titanium, a metal renowned for its exceptional strength-to-weight ratio, corrosion resistance, and biocompatibility, plays a critical role in numerous advanced technological applications. Its unique properties, however, are intrinsically linked to its physical characteristics, chief among them its melting point. Understanding the precise temperature at which titanium transitions from a solid to a liquid state is not merely an academic curiosity; it is fundamental to its successful utilization in fields ranging from aerospace engineering to medical implants. This article delves into the science behind titanium’s melting point, exploring the factors that influence it and the technological implications of this crucial parameter.
The Intrinsic Properties of Titanium and Its Melting Point
The melting point of a metal is a direct consequence of the strength of the metallic bonds that hold its atoms together in a solid lattice structure. In titanium, these bonds are particularly robust, contributing to its high melting temperature.
Understanding Metallic Bonding in Titanium
Titanium (Ti) is a transition metal found in Group 4 of the periodic table. Its atomic structure features a nucleus with 22 protons, surrounded by electrons arranged in specific orbital shells. The outermost electrons, known as valence electrons, are not tightly bound to individual atoms but rather form a “sea” of delocalized electrons that can move freely throughout the metallic crystal lattice. These shared electrons create strong electrostatic attractions between the positively charged atomic nuclei and the negatively charged electron sea, forming the metallic bond.
The strength of this metallic bond is a key determinant of a material’s melting point. A stronger bond requires more thermal energy to overcome the forces holding the atoms in their fixed positions, thus resulting in a higher melting point. Titanium’s electronic configuration and the way its atoms pack in the solid state contribute to exceptionally strong metallic bonds compared to many other common metals. This inherent characteristic is what sets titanium apart and underpins its utility in demanding environments.
The Standard Melting Point of Pure Titanium
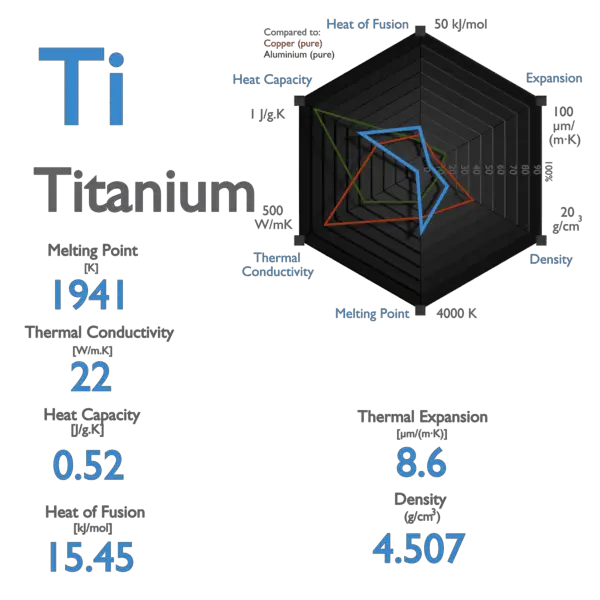
Pure titanium typically melts at approximately 1668 degrees Celsius (3032 degrees Fahrenheit). This high melting point is a significant advantage in applications where materials are exposed to extreme heat. For comparison, aluminum melts at around 660°C, steel at roughly 1370°C, and copper at about 1085°C. Titanium’s elevated melting point allows it to retain its structural integrity and mechanical properties in conditions that would cause many other metals to soften, deform, or even liquefy.
This value, however, is for pure titanium. In practical applications, titanium is rarely used in its pure form. It is often alloyed with other elements to enhance specific properties, and these alloying elements can subtly influence the melting point.
Factors Influencing Titanium’s Melting Point
While the melting point of pure titanium is a well-established value, several factors can affect this temperature in real-world scenarios:
Alloying Elements and Their Impact
Titanium alloys are engineered to optimize performance for specific applications. Common alloying elements include aluminum (Al), vanadium (V), molybdenum (Mo), iron (Fe), and tin (Sn). The addition of these elements can either slightly increase or decrease the melting point of titanium.
- Aluminum: Often added to increase strength and high-temperature creep resistance. Aluminum generally has a slight hardening effect on titanium, and its presence in alloys can sometimes lead to a minor increase in the overall melting range.
- Vanadium: Another common alloying element that enhances strength and toughness. Vanadium can influence the phase transformations within the titanium alloy, which in turn can affect the melting behavior.
- Molybdenum and Iron: These elements are often added to improve strength and hardness. They can lower the melting point of titanium alloys, sometimes quite significantly depending on their concentration.
- Eutectic Systems: In some alloy compositions, interactions between titanium and alloying elements can form eutectic systems. A eutectic point is the composition at which a mixture of substances has a single melting point, which is lower than that of any of the components. This means that certain titanium alloys can have a lower melting point than pure titanium.
The presence of impurities can also affect the melting point. Even small amounts of contaminants like oxygen, nitrogen, or carbon can form interstitial solid solutions or carbides, which can alter the lattice structure and the bonding strength, leading to a change in the melting point.
Pressure and Its Role
While the influence of pressure on the melting point of solids is generally less pronounced than that of temperature, it still plays a role. For most substances, including metals, an increase in pressure leads to a slight increase in the melting point. This is because increased pressure favors the denser solid phase over the less dense liquid phase. However, for practical engineering applications involving titanium, the effects of pressure on the melting point are usually negligible compared to the influence of alloying elements and the base temperature.
Technological Applications Driven by Titanium’s High Melting Point
Titanium’s impressive melting point, combined with its other desirable attributes, makes it an indispensable material in a wide array of high-performance technological sectors. The ability of titanium to withstand extreme temperatures without compromising its structural integrity is a primary driver for its adoption.
Aerospace and Defense
The aerospace industry is a major consumer of titanium and its alloys. The high operating temperatures experienced by aircraft and spacecraft components, such as engine parts, airframes, and exhaust systems, necessitate materials that can perform reliably under thermal stress.
- Jet Engines: Titanium alloys are extensively used in the cold sections of jet engines, including compressor blades, disks, and housings. While the hottest parts of the engine utilize superalloys with even higher temperature resistance, titanium’s strength and light weight at moderate-to-high temperatures are crucial for efficiency and performance. Its melting point ensures that these components do not succumb to the heat generated during combustion and high-speed flight.
- Airframes and Structural Components: The use of titanium alloys in airframes contributes to weight reduction, leading to improved fuel efficiency and increased payload capacity. Components like landing gear, wing spars, and fuselage sections benefit from titanium’s strength-to-weight ratio, which is maintained at temperatures encountered during flight.
- Missile and Rocketry: The high temperatures generated by rocket propulsion systems and atmospheric re-entry demand materials with excellent thermal stability. Titanium alloys are employed in various parts of missiles and rockets due to their ability to withstand these extreme conditions without melting or degrading.
Medical Implants and Biocompatibility

Titanium’s inertness and excellent biocompatibility, meaning it does not elicit an adverse immune response from the body, are as critical as its thermal properties in medical applications. However, its high melting point also contributes to its suitability for implants that may experience thermal fluctuations.
- Orthopedic Implants: Hip and knee replacement components, bone screws, and plates are often made from titanium alloys. These implants are designed to withstand the mechanical stresses of daily activity and the thermal cycles that occur within the body. The melting point ensures the material’s integrity throughout its service life.
- Dental Implants: Titanium’s osseointegration (ability to fuse with bone) and corrosion resistance make it the standard material for dental implants. The implant must remain stable and functional within the oral cavity, a challenging environment that titanium’s inherent properties, including its melting point, help it to endure.
- Surgical Instruments: High-quality surgical tools benefit from titanium’s strength, durability, and resistance to sterilization processes, which often involve heat.
Chemical Processing and Industrial Equipment
The chemical industry frequently deals with corrosive substances and high-temperature processes, making titanium an ideal choice for certain applications.
- Heat Exchangers: Titanium’s exceptional corrosion resistance, especially in aggressive environments like saltwater or acidic solutions, makes it suitable for heat exchangers used in chemical plants and desalination facilities. Its high melting point ensures longevity under thermal loads.
- Reactors and Vessels: In specialized chemical reactors where high temperatures and corrosive agents are present, titanium can be used for components that require both resistance to degradation and structural integrity.
- Offshore Oil and Gas: Titanium alloys are employed in offshore oil and gas exploration and production, particularly in components exposed to seawater and high pressures. The material’s resistance to corrosion and its ability to maintain strength at elevated temperatures are crucial.
Processing and Manufacturing Titanium: Navigating the Melting Point
The high melting point of titanium presents unique challenges and considerations for its processing and manufacturing. Techniques that are commonplace for lower-melting-point metals often require significant adaptation or are entirely unsuitable for titanium.
Melting and Casting Titanium
The sheer temperature required to melt titanium necessitates specialized equipment and precise control.
- Vacuum Arc Remelting (VAR): This is a common process for refining titanium ingots. An electric arc is struck between a consumable titanium electrode and a water-cooled crucible in a vacuum or inert atmosphere. The melting and solidification are carefully controlled to produce sound ingots free from porosity and segregation. The high vacuum prevents oxidation, which is a significant concern at titanium’s melting temperature.
- Electron Beam Melting (EBM): Similar to VAR, EBM uses a beam of electrons in a vacuum to melt titanium. This process offers excellent control over melting rates and purity, making it suitable for high-performance applications. The intense energy of the electron beam can efficiently melt titanium, but requires sophisticated vacuum systems and power control.
- Casting: While direct casting of titanium is possible, it is often challenging due to its reactivity with mold materials at high temperatures. Investment casting in ceramic molds, often under vacuum or inert gas, is a prevalent method for producing complex titanium parts. The molten titanium’s tendency to absorb gases from the atmosphere and react with mold materials means that casting processes must be meticulously controlled.
Welding and Joining Titanium
Welding titanium requires careful consideration of its reactivity with atmospheric gases, which can lead to embrittlement at elevated temperatures.
- Inert Gas Shielding: Titanium welding is almost exclusively performed using TIG (Tungsten Inert Gas) or MIG (Metal Inert Gas) welding processes under a blanket of inert gases like argon or helium. This shields the molten weld pool and the heat-affected zone from atmospheric oxygen and nitrogen, preventing contamination.
- Full Containment: In some critical applications, the entire welding setup may be enclosed in a special inert gas chamber to ensure complete protection from the atmosphere. This level of control is vital for maintaining the ductility and strength of the welded joint.
Machining and Forming Titanium
Titanium’s high strength and toughness, even at room temperature, can make machining and forming more difficult and energy-intensive compared to softer metals.
- Cutting Tools: Specialized cutting tools made from carbide or ceramic materials are often required, along with high-speed machining techniques and appropriate coolants to manage the heat generated.
- Forming: Hot forming processes are frequently employed to improve the ductility of titanium, making it easier to shape. However, even during hot forming, controlling the temperature to prevent oxidation and maintain desired material properties is crucial.
The Future of Titanium and its Melting Point in Technological Advancements
As technology continues to push the boundaries of performance, the demand for materials with exceptional properties, including high melting points, will only increase. Titanium, with its unique combination of characteristics, is poised to play an even more significant role in future innovations.
Advanced Alloys and Composites
Ongoing research and development are focused on creating new titanium alloys with enhanced properties, potentially including slightly modified melting behavior that can be tailored for specific extreme environments. Furthermore, the integration of titanium with advanced composite materials is opening up new possibilities for lightweight, high-strength structures that can withstand even greater thermal challenges.
- Ceramic Matrix Composites (CMCs) with Titanium Inserts: While CMCs offer superior high-temperature performance, they can be brittle. Integrating titanium components in strategic locations can provide toughness and improve the overall structural integrity of CMC components, especially in areas that might experience localized thermal excursions or mechanical impact.
- Titanium Foams and Lattice Structures: The development of advanced manufacturing techniques like additive manufacturing (3D printing) allows for the creation of complex titanium geometries, such as foams and lattice structures. These can offer improved thermal management properties and reduced weight, further expanding titanium’s applications in areas requiring high-temperature resistance and efficient heat dissipation.

Extreme Environments and New Frontiers
As humanity explores more extreme environments, from deep space to the depths of the ocean, materials that can reliably perform under intense conditions become paramount.
- Space Exploration: Components for spacecraft that are exposed to the vacuum of space, solar radiation, and the extreme temperature fluctuations of orbit require materials with excellent thermal stability and resistance to degradation. Titanium alloys are already essential, and future missions may demand even more specialized titanium-based solutions.
- Hypersonic Flight: The development of hypersonic vehicles, which travel at speeds exceeding Mach 5, generates immense frictional heat. Titanium alloys are critical for the leading edges and airframe components of these vehicles, and understanding their behavior at the high temperatures encountered is vital for safe and effective design. The melting point of titanium, while high, is a critical parameter to consider when designing for the sustained thermal loads of hypersonic flight.
In conclusion, the melting point of titanium—approximately 1668°C (3032°F) for pure titanium—is a fundamental property that underpins its remarkable utility across a diverse range of high-technology sectors. While alloying elements and manufacturing processes introduce nuances, the inherent thermal stability of titanium, stemming from its robust metallic bonding, makes it an indispensable material for applications where performance under extreme temperatures is non-negotiable. As technological ambitions continue to soar, titanium’s role as a cornerstone material for innovation is set to become even more pronounced.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.