In the intricate landscape of modern medicine, certain procedures stand out not just for their life-saving potential, but for the sheer ingenuity of the technology that underpins them. Ventriculoperitoneal (VP) shunting is one such procedure, a cornerstone in neurosurgery for managing hydrocephalus. Far from a simple medical intervention, VP shunting is a testament to precision biomedical engineering, material science, and a deep understanding of human physiology. This article delves into the “what” and “how” of VP shunting, exploring the technological marvels that allow a complex fluid dynamics problem within the human brain to be effectively managed by an implanted device.

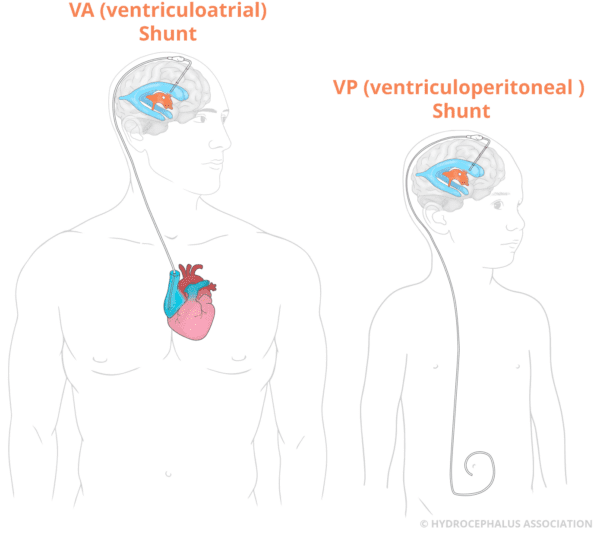
At its heart, VP shunting is the surgical implantation of a shunt system to divert excess cerebrospinal fluid (CSF) from the brain’s ventricles to another part of the body, typically the peritoneal cavity, where it can be safely absorbed. This elegant solution prevents the dangerous buildup of pressure within the skull, a condition known as hydrocephalus, which can lead to severe neurological damage or even death if left untreated. But to truly appreciate VP shunting, one must look beyond the surgical act and into the sophisticated technology that makes it possible.
The Core Mechanism: How VP Shunts Work
Understanding VP shunting begins with grasping the problem it solves and the ingenious components designed to address it. It’s a prime example of engineering principles applied directly to biological systems.
Anatomy and Physiology of Hydrocephalus: The Engineering Problem
The human brain and spinal cord are bathed in cerebrospinal fluid (CSF), a clear, watery substance that provides buoyancy, protection, and nutrient transport. CSF is continuously produced within the brain’s ventricles, circulates through a complex pathway, and is then absorbed into the bloodstream. A delicate balance exists between CSF production and absorption. Hydrocephalus occurs when this balance is disrupted, leading to an excessive accumulation of CSF and subsequent increase in intracranial pressure. This disruption can stem from overproduction, impaired absorption, or, most commonly, an obstruction in the CSF pathways.
From an engineering perspective, hydrocephalus presents a fluid dynamics challenge: how to regulate pressure within a closed, semi-rigid system (the skull) when its internal fluid management system is compromised. The solution devised by medical technology is to create an artificial bypass, effectively restoring a healthy pressure gradient.
Components of a VP Shunt System: Precision Devices
A VP shunt system is not a single device but an integrated assembly of technologically advanced components, each meticulously designed for its specific role and to operate harmoniously within the delicate environment of the human body.
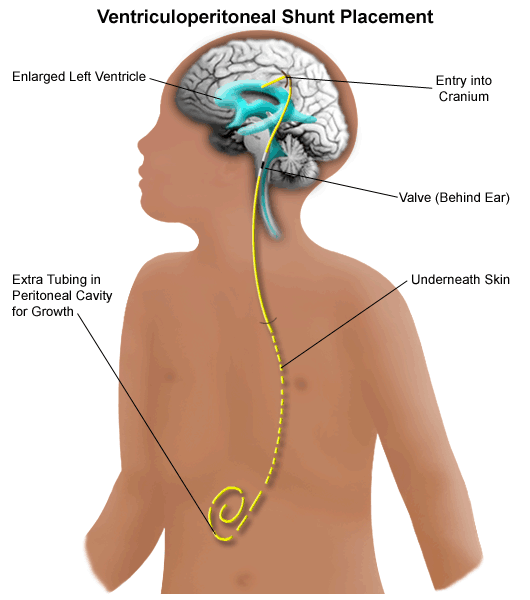
- Ventricular Catheter: This thin, flexible tube, typically made of high-grade silicone, is inserted into one of the brain’s ventricles. Its distal end often features multiple small holes to maximize CSF collection while minimizing the risk of clogging by brain tissue or debris. The material choice is crucial: silicone offers biocompatibility, flexibility, and durability, resisting degradation within the body’s environment for many years.
- Valve Mechanism: This is the “brain” of the shunt system, a pressure-regulating device that controls the flow of CSF. Valves are perhaps the most technologically advanced component. Early versions were fixed-pressure valves, allowing CSF to drain only when intracranial pressure exceeded a preset level. Modern valves, however, represent significant strides in engineering:
- Programmable Valves: These allow neurosurgeons to non-invasively adjust the pressure setting after implantation using an external magnetic device. This crucial innovation enables personalized pressure management, adapting to the patient’s evolving needs without requiring additional surgery. The internal mechanism involves intricate magnetic or mechanical components that resist external magnetic interference while allowing precise adjustment.
- Anti-Siphon Devices (ASDs): Often integrated with valves or as separate components, ASDs prevent over-drainage of CSF, particularly when a patient stands up. Gravity can create a siphoning effect, leading to a sudden drop in intracranial pressure. ASDs incorporate clever mechanical designs (e.g., diaphragms, springs) to counteract this gravitational pull, maintaining a more stable CSF pressure regardless of body position.
- Peritoneal Catheter: This long, flexible tube connects the valve to the peritoneal cavity in the abdomen. Its design ensures smooth CSF flow and typically has multiple openings along its length to facilitate absorption by the peritoneum, which acts as a vast biological filter. Like the ventricular catheter, it’s made of biocompatible silicone, engineered to withstand the dynamic environment of the abdominal cavity.
- Reservoir (Optional but Common): Often integrated into the valve housing, a reservoir is a small dome that allows the surgeon to directly access the CSF pathway. It can be used to sample CSF, inject medication, or perform a manual flush of the shunt to check for patency. Its design requires a self-sealing membrane that can withstand repeated punctures.
The Surgical Procedure: Implantation and Integration
The implantation of a VP shunt is a highly technical surgical procedure that connects these components to form a functional, integrated system. A small incision is made in the scalp, a burr hole is created in the skull, and the ventricular catheter is carefully guided into a ventricle. The valve is typically placed under the skin behind the ear or on the top of the head. The peritoneal catheter is then tunneled subcutaneously down the neck and chest wall, finally inserted into the abdominal cavity. The entire system lies beneath the skin, invisible externally save for a slight bump where the valve is located. This subcutaneous placement is crucial for minimizing infection risk and ensuring long-term patient comfort.
Technological Evolution of Shunt Systems
The journey of VP shunt technology is a compelling narrative of continuous innovation driven by clinical need and advancements in materials science, micro-mechanics, and digital control systems.
From Basic Drains to Sophisticated Valves
Early shunt systems, pioneered in the 1950s, were relatively simple one-way valves. While revolutionary, they were prone to complications such as fixed pressure settings that didn’t adapt to patient needs, leading to frequent revisions. The 1960s saw the introduction of the first effective shunts, primarily the Hakim valve, marking a significant leap. These initial designs laid the groundwork, but their limitations spurred engineers and clinicians to seek more refined solutions.
Programmable Shunts and Advanced Materials
The advent of programmable valves in the 1990s was a paradigm shift. These devices moved shunt technology from a “one-size-fits-all” approach to personalized medicine. The ability to non-invasively adjust the pressure setting using an external magnet meant that complications related to over- or under-drainage could often be managed without further surgery. This required sophisticated internal mechanisms resistant to everyday magnetic fields but responsive to a specific external programmer.
Concurrently, advancements in materials science have been critical. Shunts now use highly biocompatible silicone elastomers that minimize inflammatory responses and have improved resistance to calcification or degradation. Research into coatings has led to devices with surfaces designed to resist infection (e.g., antibiotic-impregnated shunts) or reduce the adherence of biological debris, thereby mitigating the risk of blockages. MRI compatibility became another crucial design consideration, ensuring that patients with shunts could safely undergo essential diagnostic imaging.
Telemetry and Monitoring Integration: The Smart Shunt Era
The frontier of shunt technology is increasingly moving towards “smart” systems. While still largely in research and development, the integration of sensors and telemetry offers the promise of real-time monitoring of intracranial pressure (ICP) and shunt function. Imagine a shunt that can wirelessly transmit data on CSF flow or pressure directly to a clinician, allowing for proactive adjustments or early detection of potential malfunctions. Such systems would leverage miniaturized sensors, low-power wireless communication protocols, and sophisticated data analytics to provide an unprecedented level of personalized care. This represents a significant convergence of medical device technology with digital health and IoT (Internet of Things) principles.
Challenges and Innovations in Shunt Technology
Despite their effectiveness, VP shunts are not without challenges. These difficulties, however, continually drive innovation, pushing the boundaries of what medical technology can achieve.
Common Complications and Engineering Solutions
The most common complications associated with VP shunts include infection, obstruction (blockage), and shunt malfunction (over-drainage or under-drainage). Each of these problems presents a specific engineering challenge:
- Infection: Implanted devices are susceptible to bacterial colonization. Innovations here include antibiotic-impregnated shunt catheters and new surface coatings designed to resist microbial adhesion. Future research explores anti-biofilm technologies and materials that actively repel bacteria.
- Obstruction: Blockages can occur from tissue ingrowth, blood clots, or proteinaceous debris. Engineers are working on new catheter designs with improved flow characteristics, materials that reduce protein adhesion, and even neuro-endoscopic techniques that can clear blockages with minimally invasive tools.
- Malfunction (Over/Under-drainage): This is often a function of valve setting or changes in a patient’s physiological state. Programmable valves significantly reduced this issue, but ongoing research focuses on more dynamic, responsive valve systems that can automatically adjust to subtle changes in ICP or patient posture, using internal micro-sensors and feedback loops.
Miniaturization and Precision Engineering
The trend in all medical devices is towards miniaturization and less invasive approaches. For shunts, this means developing smaller, more discrete components that reduce the surgical footprint and enhance patient comfort. Precision engineering allows for the creation of intricate valve mechanisms that are both robust and highly accurate, operating within tolerances measured in microns. Advanced manufacturing techniques like micro-molding and precision machining are critical in this endeavor.
AI and Diagnostic Support: The Future Horizon
Looking ahead, artificial intelligence (AI) is poised to play a transformative role. AI algorithms could analyze vast datasets of patient ICP, CSF flow, and clinical symptoms to predict shunt malfunctions before they become critical. They could also help optimize programmable shunt settings for individual patients, moving towards a truly personalized “smart” shunt that learns and adapts. Diagnostic tools enhanced with AI could provide earlier and more accurate identification of hydrocephalus subtypes, guiding the choice of shunt technology or alternative treatments.
The Future Landscape of Neurological Shunting Technology
The evolution of VP shunting is far from over. The coming decades promise even more sophisticated, integrated, and personalized solutions, marrying advanced engineering with biological insights.
Bio-integrated Systems and Personalized Medicine
The ultimate goal for many medical technologies is seamless integration with the human body. For shunts, this could mean bio-resorbable materials for temporary shunts, or components that can adapt and even “heal” within the body. Personalized medicine will see shunts designed not just for a patient’s specific pressure needs, but potentially for their unique anatomical structures, perhaps even using 3D printing to create custom-fit components. The convergence of bio-sensing, advanced materials, and AI will enable systems that are highly responsive and tailored to each individual’s physiological dynamics.
Non-Invasive Alternatives and Adjunctive Technologies
While VP shunting remains a vital intervention, technological advancements are also driving alternatives. Endoscopic Third Ventriculostomy (ETV), a minimally invasive neurosurgical procedure, uses specialized endoscopes to create a bypass within the brain, negating the need for an implanted shunt in some cases. Further research into pharmacological treatments or even targeted therapies using nanotechnology could reduce the reliance on mechanical shunts for certain types of hydrocephalus, although these are currently still distant prospects. However, technology will continue to play a role in diagnostic precision, helping identify which patients are best suited for shunting versus other interventions.
Conclusion
VP shunting stands as a powerful testament to the impact of biomedical engineering on human health. What began as a rudimentary solution to a complex medical problem has evolved into a highly sophisticated technological system, capable of restoring a semblance of normal life to millions affected by hydrocephalus. From the carefully chosen biocompatible materials to the intricate pressure-regulating valves and the emerging promise of smart, connected systems, every aspect of VP shunting is a product of relentless innovation. As technology continues its rapid advancement, the future promises even more effective, personalized, and seamlessly integrated solutions, further solidifying VP shunting’s place as a triumph of medical technology and a beacon of hope for neurological patients worldwide.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.