The Economic Footprint of Pharmaceutical Research and Dosage Determination
The question of “what is the maximum dose of flecainide” might seem purely medical at first glance, but beneath the clinical definition lies a vast economic landscape shaped by extensive research and development (R&D), regulatory processes, and market dynamics. For pharmaceutical companies, determining optimal dosages, including the maximum safe and effective threshold for drugs like flecainide, represents a monumental financial investment. This process is not merely scientific; it is a business strategy, an allocation of substantial capital aimed at securing market authorization and patient access, ultimately driving revenue and shareholder value. The journey from molecular compound to a precisely defined dosage involves billions of dollars in expenditure, influencing everything from stock valuations to national healthcare budgets.

Investment in Clinical Trials and Regulatory Approval
Establishing the maximum dose of any medication, flecainide included, is a critical phase within a multi-stage clinical trial process. This rigorous testing, which spans preclinical studies, Phase I (safety and dosage), Phase II (efficacy and safety), and Phase III (large-scale efficacy and monitoring), demands immense financial resources. Pharmaceutical firms invest heavily in recruiting patients, compensating clinical investigators, managing data, and ensuring compliance with stringent ethical and regulatory standards across multiple jurisdictions. Each trial phase carries its own significant cost, with Phase III trials often running into hundreds of millions of dollars due to their scale and duration.
Furthermore, securing regulatory approval from bodies like the FDA in the United States or the EMA in Europe is an exhaustive and expensive endeavor. Companies must compile comprehensive data sets, including extensive dose-response analyses, demonstrating the drug’s safety and efficacy within a defined dosage range. This submission process alone involves substantial fees and the employment of highly skilled regulatory affairs professionals. The success or failure of a drug to gain approval, often hinging on the clarity and safety profile of its dosage recommendations, directly impacts a company’s financial future, affecting investor confidence, stock prices, and future R&D budgets. The “maximum dose” isn’t just a number; it’s the culmination of years of financial commitment and scientific validation, dictating the market entry and lifespan of a product.
The Cost Structure Behind Drug Development
The full cost of bringing a new drug to market, inclusive of the intricate work involved in determining its maximum dose, is staggering. Estimates vary, but many studies place the average cost at over $1 billion, with some reaching upwards of $2.5 billion when factoring in the capitalized cost of failures. This includes costs associated with basic research, discovery, preclinical testing, clinical trials, manufacturing development, and regulatory submission. For a drug like flecainide, developed decades ago but still subject to ongoing post-market surveillance and potential re-evaluation of its profile, these costs represent a long-term capital commitment.
The economic model of pharmaceutical development is heavily reliant on patent protection, which grants a period of market exclusivity to recoup these enormous R&D costs. The “maximum dose” becomes a key specification within the patent and subsequent intellectual property, ensuring that the company’s investment in defining its optimal use is protected. This cost structure fundamentally shapes the pricing strategies for pharmaceutical products, influencing the accessibility and affordability of treatments globally. From a business finance perspective, understanding the investment required to define critical parameters like maximum dose is crucial for assessing a company’s asset valuation, future earnings potential, and risk profile. It is a direct link between scientific endeavor and financial performance.
Understanding the Financial Impact on Healthcare Systems and Patients
Beyond the pharmaceutical industry’s investment, the established dosage guidelines for medications like flecainide, including its maximum dose, have profound financial implications for healthcare systems, insurers, and individual patients. These impacts manifest in direct costs such as medication pricing and insurance coverage, as well as indirect costs related to managing chronic health conditions.
Direct Costs: Medication Pricing and Insurance Dynamics
The price of a medication is influenced by a multitude of factors, not least of which is the R&D investment discussed previously. For flecainide, available as a generic, its pricing structure has evolved considerably from its proprietary origins. However, for any medication, the cost per dose, and consequently the cost of a maximum dose regimen, directly affects patient out-of-pocket expenses, co-pays, and deductibles. Patients requiring a maximum dose often face higher cumulative medication costs over time due to the increased quantity of drug prescribed.
Insurance providers play a pivotal role in mediating these costs. Formularies, which list covered medications, often categorize drugs into tiers, with varying co-payment levels. The inclusion of a drug like flecainide, and the typical costs associated with its prescribed dosages, influences insurance premiums and the financial health of the insurer. Maximum dose prescriptions can trigger specific review processes by insurance companies to ensure medical necessity, adding administrative overhead for both providers and patients. From a personal finance perspective, understanding how one’s insurance plan covers potentially high-dose, long-term medications is critical for financial planning, potentially preventing unexpected medical debt. Employers offering health benefits also grapple with these costs, as rising pharmaceutical expenses directly impact their business finance and employee compensation packages.
Indirect Costs: Managing Chronic Conditions
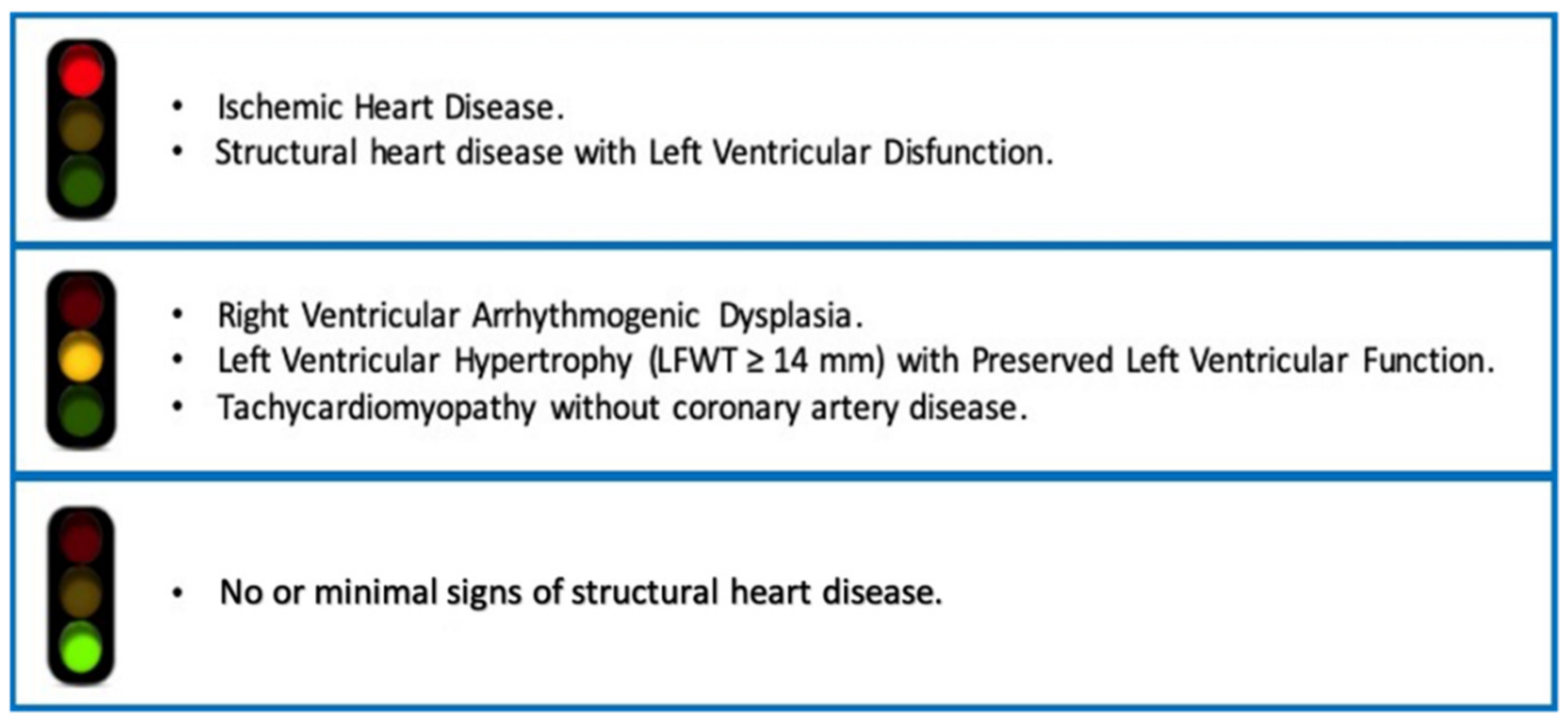
Conditions necessitating flecainide, such as certain cardiac arrhythmias, often require long-term management, extending beyond just medication costs. The maximum dose might indicate a more severe or resistant form of the condition, potentially leading to increased healthcare utilization, including more frequent specialist visits, diagnostic tests, or even hospitalizations. These indirect costs—missed workdays, caregiver burden, transportation to appointments, and the financial strain of lifestyle adjustments—can accumulate significantly, impacting a patient’s overall financial well-being.
For healthcare systems, managing populations with chronic conditions like arrhythmias represents a substantial budgetary challenge. The cost of complex care pathways, emergency interventions, and ongoing monitoring associated with conditions managed by drugs like flecainide, especially at higher dosages, contributes significantly to national healthcare expenditures. These costs influence policy decisions regarding public health funding, resource allocation, and initiatives aimed at improving chronic disease management, all of which fall under the umbrella of business finance for governmental and private healthcare providers. The “maximum dose” in this context becomes a marker of potential financial burden on individuals and the broader healthcare economy, necessitating comprehensive financial planning and support structures.
Flecainide’s Role in the Pharmaceutical Market: A Financial Perspective
The existence and utilization of flecainide, including its dosage spectrum, contribute to a specific segment of the pharmaceutical market. Understanding its position from a financial perspective involves analyzing market valuation, competitive dynamics, and the investment opportunities it presents, particularly for companies operating in the cardiovascular therapeutic area.
Market Valuation and Competitive Landscape
Flecainide, while an established antiarrhythmic, operates within a competitive market segment that includes other antiarrhythmic drugs and alternative treatment modalities. Its market valuation, as part of a generic drug portfolio, is influenced by factors such as global demand for arrhythmia treatments, patent expirations of newer competing drugs, and the overall trajectory of cardiovascular disease prevalence. Companies that manufacture generic flecainide generate revenue from its sale, contributing to their overall market capitalization and attractiveness to investors.
The “maximum dose” contributes to a drug’s market share by defining its therapeutic window and suitability for various patient profiles. A drug with a well-defined and safe maximum dose can instill confidence in prescribers, potentially leading to broader adoption within its indication. Conversely, a drug with a narrow therapeutic window or significant safety concerns at higher doses might face market limitations. From a business finance standpoint, understanding the competitive landscape and a drug’s specific role within it is crucial for strategic planning, pricing decisions, and projecting future sales volumes.
Investment Opportunities in Antiarrhythmic Drug Producers
For investors interested in the pharmaceutical sector, companies involved in the production and distribution of antiarrhythmic drugs like flecainide represent specific investment opportunities. These can range from large multinational pharmaceutical corporations with diverse portfolios to smaller generic drug manufacturers. Investment analysis often considers a company’s pipeline of new drugs, its market share in key therapeutic areas, its R&D efficiency (including the cost-effectiveness of dosage determination), and its ability to manage regulatory changes.
The stability of demand for essential medications like flecainide can offer a degree of resilience in investment portfolios, particularly during economic downturns, as healthcare needs often remain consistent. However, investors also weigh risks such as increased generic competition, potential changes in clinical guidelines that might affect prescribed dosages, or the introduction of superior alternative therapies. Analyzing the financial health and strategic direction of companies involved in this therapeutic area requires a deep dive into their product portfolios, manufacturing capabilities, and their long-term growth prospects, all influenced by the scientific and financial rigor applied to defining critical product specifications like a drug’s maximum dose.
Financial Tools and Strategies for Managing Medication Costs
Given the potential financial implications associated with medication regimens, particularly those involving maximum doses for chronic conditions, various financial tools and strategies become essential for patients and healthcare providers alike. These tools aim to mitigate the economic burden and ensure sustainable access to necessary treatments.
Personal Financial Planning for Healthcare Expenses
For individuals, proactive personal financial planning is paramount. This includes establishing emergency funds to cover unexpected medical costs, carefully selecting health insurance plans that align with anticipated healthcare needs (e.g., strong prescription drug coverage for chronic conditions), and utilizing Health Savings Accounts (HSAs) or Flexible Spending Accounts (FSAs) to pay for qualified medical expenses with pre-tax dollars. Understanding one’s specific insurance benefits, including co-pays, deductibles, and out-of-pocket maximums for prescription drugs, can help budget for medication costs, especially when a maximum dose is prescribed. Many pharmaceutical companies also offer patient assistance programs that can reduce the cost of medications for eligible individuals, a financial tool worth exploring.

Business Finance and Healthcare Cost Management
From the perspective of healthcare systems and employers, managing medication costs is a significant component of business finance. Strategies include negotiating favorable pricing with pharmaceutical manufacturers and distributors, implementing formulary management to promote cost-effective generic alternatives (like generic flecainide), and investing in population health management programs that focus on preventative care and chronic disease management to reduce the need for high-cost interventions. For self-insured employers, data analytics on employee health claims can inform benefit design and wellness initiatives aimed at controlling pharmaceutical expenditures. These financial strategies are crucial for maintaining the solvency of healthcare organizations and ensuring the sustainability of employee health benefits. The maximum dose of a widely used drug like flecainide, therefore, is not just a clinical detail but a vital data point that informs actuarial calculations, budget allocations, and long-term financial forecasting across the healthcare ecosystem.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.