In the rapidly evolving landscape of green technology and industrial engineering, fundamental chemical constants are no longer relegated to high school textbooks. Instead, they serve as the foundational “source code” for modern hardware and software innovations. Among these, the value of $Kw$—the self-ionization constant of water—stands as a critical metric. While a chemist might view $Kw$ as a simple equilibrium constant, a technology professional sees it as a vital parameter for optimizing hydrogen fuel cells, enhancing semiconductor manufacturing, and refining the algorithms used in environmental monitoring software.

Understanding what $K_w$ is in chemistry and why its value ($1.0 times 10^{-14}$ at 25°C) matters is essential for anyone working at the intersection of “Hard Tech” and sustainable energy. This article explores how this specific chemical value drives technological trends, powers digital simulations, and secures the future of high-precision industrial gadgets.
The Fundamental Constant of Clean-Tech Innovation
At its core, $Kw$ represents the equilibrium constant for the auto-ionization of water. In any aqueous solution, water molecules are constantly dissociating into hydronium ions ($H3O^+$) and hydroxide ions ($OH^-$). The mathematical product of these ion concentrations is the $K_w$ value. In the tech sector, this isn’t just a number; it is a hardware constraint that dictates how sensors are calibrated and how energy systems are designed.
Defining $K_w$ for the Engineering Mindset
For the tech-oriented professional, $Kw$ can be viewed as the “noise floor” of aqueous chemistry. At standard laboratory temperature (25°C), $Kw$ is exactly $1.0 times 10^{-14}$. This value determines the neutral point of the pH scale (pH 7). However, in industrial tech applications—such as high-pressure steam turbines or deep-sea sensors—temperatures vary wildly. Because the dissociation of water is endothermic, the $K_w$ value increases as temperature rises.
Engineers building climate-tech hardware must program these thermal variances into their logic controllers. If a digital sensor fails to account for the shift in $K_w$ at 50°C (where it rises to approximately $5.47 times 10^{-14}$), the resulting pH readings will be inaccurate, potentially leading to catastrophic hardware corrosion or system failure.
Why Temperature Variance is a Critical Software Variable
In the world of Software as a Service (SaaS) for industrial management, $Kw$ is a critical variable in predictive modeling. Modern “Digital Twin” technology—which creates a virtual replica of a physical asset—relies on precise chemical algorithms. Software developers building tools for water treatment plants or chemical refineries must hard-code the temperature dependence of $Kw$ using the van ‘t Hoff equation.
This ensures that the software can predict how the “value” of water’s ionic product will change under stress. Without this technical integration, AI-driven automation systems would be unable to maintain the delicate chemical balance required for high-efficiency operations, proving that even a microscopic constant has massive digital implications.
$K_w$ and the Evolution of Green Hydrogen Technology
One of the most significant technology trends of the decade is the shift toward Green Hydrogen. This process relies on electrolysis, where electricity is used to split water into oxygen and hydrogen gas. Here, the $K_w$ value is the technical gatekeeper of efficiency.
Precision Electrolysis: The Hardware Challenges
The efficiency of an electrolyzer is directly tied to the ionic conductivity of the medium. Since $Kw$ dictates the concentration of ions available in pure water, it sets the baseline for the electrical resistance the hardware must overcome. Tech companies specializing in Proton Exchange Membrane (PEM) electrolyzers are constantly researching ways to manipulate the environment around the $Kw$ equilibrium to lower energy consumption.
By understanding the $Kw$ value, engineers can design better catalysts and membranes that operate more effectively at specific ionic strengths. The goal is to maximize the “value” of every kilowatt-hour of renewable energy fed into the system, making $Kw$ a silent partner in the quest for 100% efficient energy conversion.
Digital Twins: Modeling Ionic Equilibria in Real-Time
In the realm of AI and simulation tools, modeling the behavior of $Kw$ within an electrolyzer stack allows for real-time optimization. Advanced software suites now use machine learning to analyze how fluctuations in $Kw$ (due to localized heating within the cell) affect the longevity of the hardware.
By feeding real-time data from IoT (Internet of Things) sensors into these models, tech firms can predict when a membrane is likely to degrade. This “predictive maintenance” is a cornerstone of modern industrial tech, turning a basic chemistry value into a tool for reducing operational downtime and increasing the ROI of green energy gadgets.
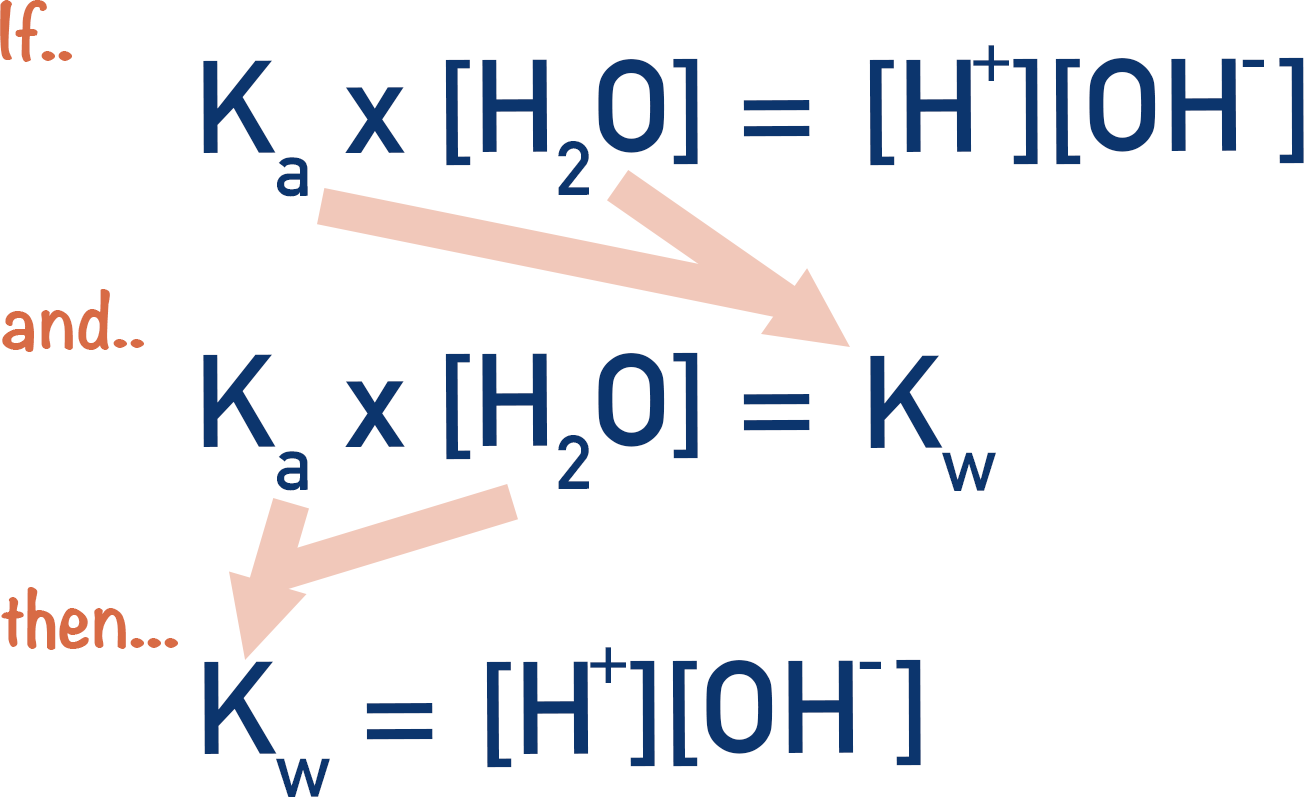
Industrial Tech: Ultrapure Water in Semiconductor Fabrication
The tech world’s reliance on $Kw$ extends into the very heart of our digital lives: the semiconductor. To manufacture the microchips that power our smartphones and AI tools, “Ultrapure Water” (UPW) is required. In this niche, the $Kw$ value is the ultimate benchmark for quality control.
The Tech of Water Purification
In semiconductor fabrication, even the slightest impurity can ruin a silicon wafer. Ultrapure water must be stripped of all ions except for those naturally occurring due to the $Kw$ constant. The theoretical maximum resistivity of water—18.2 MΩ·cm at 25°C—is determined entirely by the $Kw$ value.
Hardware engineers designing UPW systems use the $Kw$ constant to calibrate the resistivity meters that monitor water quality. If the measured resistivity deviates from the $Kw$-calculated ideal, the automated system immediately triggers a lockdown of the production line. In this context, $K_w$ is not just a value; it is a digital security guard for the global supply chain of technology.
Monitoring Systems and Sensor Accuracy
The gadgets used to monitor $Kw$-related metrics have seen a massive technological leap. We have moved from manual titration to solid-state ionic sensors that integrate directly with cloud-based dashboards. These sensors use sophisticated algorithms to compensate for the “temperature coefficient” of $Kw$.
For tech professionals, the “value” of $Kw$ here is its predictability. Because we know exactly how $Kw$ behaves under different physical conditions, we can build highly reliable automated systems that require minimal human intervention. This reliability is what allows massive chip-making “fabs” to operate 24/7 with extreme precision.
The Future of Chemical Tech: AI and Predictive Molecular Modeling
As we look toward the future of technology, the intersection of chemistry and AI is becoming increasingly blurred. The $K_w$ value is now being used to train neural networks that can discover new materials and carbon-capture technologies.
Machine Learning in pH and $K_w$ Calculations
New AI tools are being developed to simulate complex chemical environments, such as the interior of a lithium-ion battery or the carbon-rich waters of a carbon-sequestration plant. In these simulations, $Kw$ is a fundamental constant in the training data. By teaching AI how $Kw$ interacts with other variables, researchers can use “In-Silico” (software-based) testing to bypass years of expensive laboratory work.
This tech-forward approach to chemistry is accelerating the development of new gadgets, from high-capacity batteries to wearable medical sensors that monitor the ionic balance of human sweat. The “value” of $K_w$ in this digital era is its role as a stable anchor in a sea of complex, simulated data.
Quantum Computing’s Role in Solving Ionic Puzzles
While classical computers struggle with the nuances of molecular bonding and ionic dissociation at the quantum level, the next generation of gadgets—Quantum Computers—is set to revolutionize this space. Quantum algorithms are uniquely suited to modeling the behavior of water molecules as they dissociate according to the $K_w$ constant.
As tech giants like IBM and Google advance quantum hardware, we expect to see a breakthrough in our understanding of $K_w$ at extreme states (such as supercritical water). This could lead to entirely new classes of technology, including more efficient cooling systems for data centers and advanced desalination techniques that could solve the global water crisis.

Conclusion: The Technical Value of a Chemical Constant
What is $K_w$ in chemistry value? To a student, it is $1.0 times 10^{-14}$. To the tech industry, it is a vital metric that ensures our semiconductors are flawless, our green hydrogen is efficient, and our AI simulations are accurate. In the world of Technology Trends, Software, and Gadgets, the most profound innovations often stem from the most basic truths of the physical world.
By mastering the technical implications of $Kw$, engineers and software developers are not just observing a chemical reaction; they are leveraging a fundamental law of nature to build a smarter, cleaner, and more efficient technological future. Whether it is through the lens of a digital twin or the hardware of a semiconductor fab, $Kw$ remains a high-value constant in the ever-expanding universe of Tech.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.