Homologous recombination (HR) is a fundamental biological process that plays a critical role in maintaining genomic stability and ensuring the accurate transmission of genetic information across generations. At its core, HR is a type of genetic recombination that occurs between sequences of DNA that are highly similar, or homologous. This similarity is typically found between two identical chromosomes during meiosis, or between sister chromatids following DNA replication. While the concept of “recombination” might evoke images of shuffling decks of cards to create new combinations of traits, homologous recombination is a far more precise and essential molecular mechanism. It’s not just about creating genetic diversity; it’s about repairing damaged DNA and ensuring the integrity of our blueprints for life.
In the realm of technology, particularly as it intersects with biology and medicine, understanding homologous recombination has profound implications. Advances in molecular biology, genetics, and bioinformatics are increasingly leveraging and manipulating this natural process for a variety of applications. From developing new gene therapies to engineering crops with improved traits, the ability to understand, control, and even induce homologous recombination is a cornerstone of modern biotechnology. This article will delve into the fundamental mechanisms of homologous recombination, its critical biological roles, and its burgeoning applications within the technological landscape.
The Molecular Machinery of Homologous Recombination
Homologous recombination is a complex, multi-step process orchestrated by a suite of specialized proteins. These proteins work in a coordinated fashion to identify homologous DNA sequences, initiate DNA breakage, strand invasion, and ultimately, the accurate exchange of genetic material. The efficiency and fidelity of this process are paramount, as errors can lead to significant genomic instability and disease.
Initiation: Creating the Double-Strand Break
The initiation of homologous recombination typically begins with the generation of a DNA double-strand break (DSB). This break can arise spontaneously due to various cellular stresses or can be deliberately induced by cellular machinery for the purpose of recombination.
DNA Double-Strand Break Formation
In the context of DNA repair, DSBs are often recognized as signals of significant DNA damage that require immediate attention. Proteins like the MRN complex (MRE11-RAD50-NBS1) are among the first responders, binding to the broken ends and initiating a cascade of downstream events. These proteins play a crucial role in sensing the break, signaling to other repair proteins, and preparing the DNA ends for further processing.
Resection: Generating Single-Stranded Tails
Once the DSB is recognized, the DNA ends must be processed to create single-stranded DNA (ssDNA) tails. This process, known as resection, is carried out by exonucleases. The goal is to degrade the 5′ ends of the broken DNA, leaving behind 3′ ssDNA overhangs. These 3′ overhangs are critical because they are the “invasion-competent” ends that will search for and bind to a homologous DNA template. Enzymes like CtIP and the MRN complex are involved in initiating this resection, which is then extended by other nucleases like Exo1 or BLM helicase. The length and structure of these ssDNA tails are tightly regulated to ensure efficient strand invasion.
Strand Invasion and Template Search
The heart of homologous recombination lies in the invasion of a homologous DNA duplex by the 3′ ssDNA tail. This step is mediated by specialized proteins that load onto the ssDNA and facilitate its search for and binding to an undamaged homologous DNA molecule.
Loading of Recombination Proteins
The key protein complex responsible for loading onto the ssDNA tails is Rad51 (in eukaryotes). Rad51, along with accessory proteins like BRCA2, assembles into a nucleoprotein filament on the ssDNA. This filament is crucial for both protecting the ssDNA from degradation and for mediating the search for homology. BRCA2, for instance, is a critical tumor suppressor that plays a vital role in delivering Rad51 to the ssDNA and promoting the formation of stable nucleoprotein filaments. Defects in BRCA2 are strongly linked to an increased risk of breast and ovarian cancers, highlighting the importance of HR in preventing tumorigenesis.
Homology Search and Strand Invasion
The Rad51-coated ssDNA filament then scans a homologous DNA duplex, searching for a region of complementary sequence. Once a region of sufficient homology is found, the filament mediates “strand invasion.” This involves the displacement of one strand of the duplex DNA by the invading ssDNA, forming a displacement loop (D-loop). This D-loop structure is a critical intermediate, establishing the physical connection between the broken DNA molecule and its intact homologous template. The search for homology is a highly specific process, ensuring that recombination occurs between the correct DNA sequences.
DNA Synthesis and Resolution
Following strand invasion, DNA synthesis occurs, using the homologous template to fill in the gaps and repair the broken DNA. The subsequent steps involve the resolution of complex DNA structures and the ligation of DNA strands to complete the recombination event.
DNA Synthesis and Holliday Junction Formation
Once the strand invasion is established, DNA polymerase can begin to synthesize new DNA, extending the invading strand and using the homologous template as a guide. This process can lead to the formation of a structure known as a Holliday junction. A Holliday junction is a cross-shaped structure where two homologous DNA molecules are linked by the reciprocal exchange of strands. These junctions can be formed through various pathways, such as the Double Holliday Junction (dHJ) model or the Synthesis-Dependent Strand Annealing (SDSA) model. The dHJ model involves the formation of two Holliday junctions, which are then resolved to complete the exchange. The SDSA model is a more direct pathway that avoids the formation of stable double Holliday junctions.
Resolution of Holliday Junctions and DNA Ligation
The final stages of homologous recombination involve the resolution of Holliday junctions and the ligation of DNA breaks. Specialized enzymes called resolvases cleave the Holliday junctions in specific orientations, leading to the formation of either crossover products (where flanking genetic material is exchanged) or non-crossover products (where the genetic information is exchanged within the recombination site but flanking markers remain in their original positions). Following resolution, DNA ligases seal any remaining nicks in the DNA backbone, restoring the integrity of the chromosome. The precise resolution of Holliday junctions is critical, as incorrect resolution can lead to chromosomal rearrangements and mutations.
Biological Significance: Maintaining Genome Integrity
Homologous recombination is not merely an esoteric biochemical reaction; it is a vital process that underpins the health and stability of all living organisms. Its roles extend from ensuring accurate chromosome segregation during cell division to facilitating the repair of catastrophic DNA damage.
DNA Repair: The Primary Guardian of the Genome
The most prominent role of homologous recombination is in the repair of DNA double-strand breaks. While other DNA repair pathways exist, HR is considered the most accurate and error-free mechanism for repairing DSBs, particularly in diploid organisms.
Repairing Double-Strand Breaks
When a DSB occurs, the cell has multiple repair options. Non-homologous end joining (NHEJ) is a faster but often error-prone pathway that directly ligates the broken ends. In contrast, homologous recombination requires a homologous template, typically the sister chromatid, to accurately reconstruct the lost DNA sequence. This reliance on a template makes HR a high-fidelity repair mechanism, minimizing the introduction of mutations. This is particularly important in actively dividing cells, where unrepaired or misrepaired DSBs can lead to cell death or the development of cancer.
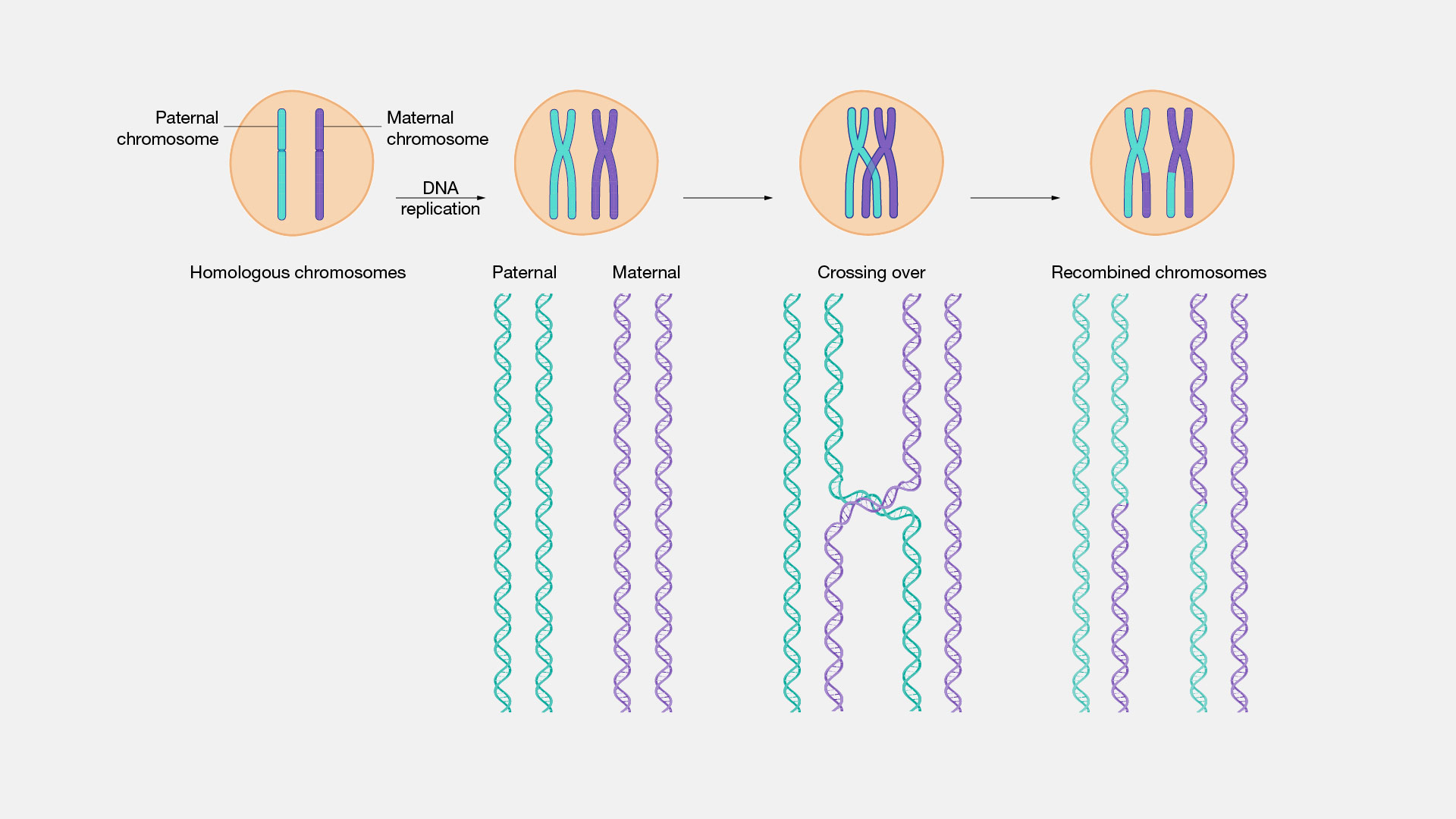
Protecting the Germline
The integrity of the germline is paramount for the survival of a species. Homologous recombination plays a crucial role in safeguarding the genetic material that will be passed on to the next generation. During meiosis, the specialized cell division that produces sperm and egg cells, HR is essential for ensuring proper chromosome pairing and segregation.
Meiosis and Genetic Diversity
Meiosis is a two-stage cell division process that halves the chromosome number. Homologous recombination, specifically crossing over, is a hallmark of prophase I of meiosis. It’s during this phase that homologous chromosomes pair up tightly and exchange segments of genetic material.
Crossing Over: The Engine of Genetic Variation
The process of crossing over, mediated by homologous recombination, is the primary source of genetic variation within sexually reproducing populations. By shuffling alleles between homologous chromosomes, HR creates new combinations of genes that can be passed on to offspring. This genetic shuffling is essential for adaptation and evolution, allowing species to respond to changing environmental pressures. Without HR, offspring would inherit chromosomes virtually identical to those of their parents, significantly limiting evolutionary potential.
Ensuring Accurate Chromosome Segregation
Beyond generating diversity, the physical linkages formed by the reciprocal exchanges (chiasmata) between homologous chromosomes during meiotic recombination are essential for their proper alignment and segregation during meiosis I. Without these connections, homologous chromosomes might not align correctly, leading to aneuploidy (an abnormal number of chromosomes) in the gametes, which can result in developmental disorders or infertility.
Technological Applications: Harnessing the Power of Recombination
The intricate mechanisms and critical roles of homologous recombination have not gone unnoticed by the scientific and technological communities. Researchers and engineers are increasingly devising innovative ways to leverage this natural process for a wide range of applications, from gene editing to drug discovery.
Gene Editing Technologies: Precision Engineering of the Genome
The advent of powerful gene editing tools has revolutionized our ability to manipulate DNA. Homologous recombination is a key component of some of the most prominent gene editing technologies.
CRISPR-Cas9 and Homology-Directed Repair (HDR)
While CRISPR-Cas9 is primarily known for its ability to introduce double-strand breaks at specific genomic locations, the subsequent repair of these breaks can be guided by homologous recombination. When a DNA template with homology to the targeted break site is provided along with the CRISPR-Cas9 system, cells can utilize homology-directed repair (HDR) to precisely incorporate the sequence from the template into the genome. This allows for the correction of mutations, the insertion of new genes, or the precise modification of existing ones. This targeted genetic engineering has immense potential for treating genetic diseases.
Other Gene Editing Approaches
Beyond CRISPR-Cas9, other gene editing technologies also rely on or are influenced by homologous recombination. For instance, zinc-finger nucleases (ZFNs) and transcription activator-like effector nucleases (TALENs) also create DSBs that can be repaired via HDR, enabling similar precise genomic modifications. The ability to control and direct these repairs opens up vast possibilities in synthetic biology and personalized medicine.
Therapeutic Development: Fighting Disease at the Genetic Level
The ability to repair or modify genes associated with diseases offers a paradigm shift in therapeutic development. Homologous recombination is central to many of these cutting-edge approaches.
Gene Therapy
Gene therapy aims to treat diseases by introducing, removing, or altering genetic material within a patient’s cells. Homologous recombination, particularly through HDR, is a crucial mechanism for precisely integrating therapeutic genes into the genome at specific loci. This allows for stable and long-term expression of the corrective gene, offering potential cures for a range of inherited disorders, such as cystic fibrosis, sickle cell anemia, and certain types of cancer. The precision afforded by HR ensures that therapeutic genes are inserted in a controlled manner, minimizing off-target effects.
Cancer Treatment and Prevention
Given HR’s role in DNA repair and its link to cancer development (e.g., BRCA mutations), it is a significant target for cancer research and treatment. Understanding how cancer cells exploit or evade HR pathways can lead to novel therapeutic strategies. Conversely, enhancing HR in healthy cells could offer preventative measures against certain cancers. Furthermore, many chemotherapies work by inducing DNA damage, and the cell’s response, including HR, can influence treatment efficacy. Research into targeting the HR pathway in cancer cells themselves, making them more susceptible to DNA damage and death, is a very active area of investigation.
Biotechnology and Agriculture: Enhancing Traits and Understanding Organisms
The principles of homologous recombination are also being applied to improve crops, develop new biofuels, and gain deeper insights into the fundamental biology of various organisms.
Genetically Modified Organisms (GMOs)
In agriculture, homologous recombination plays a role in the development of genetically modified organisms (GMOs). While transformation methods can vary, precise gene insertion, often facilitated by HR, allows for the introduction of desirable traits such as pest resistance, herbicide tolerance, or improved nutritional content. This has the potential to increase crop yields, reduce pesticide use, and enhance food security.
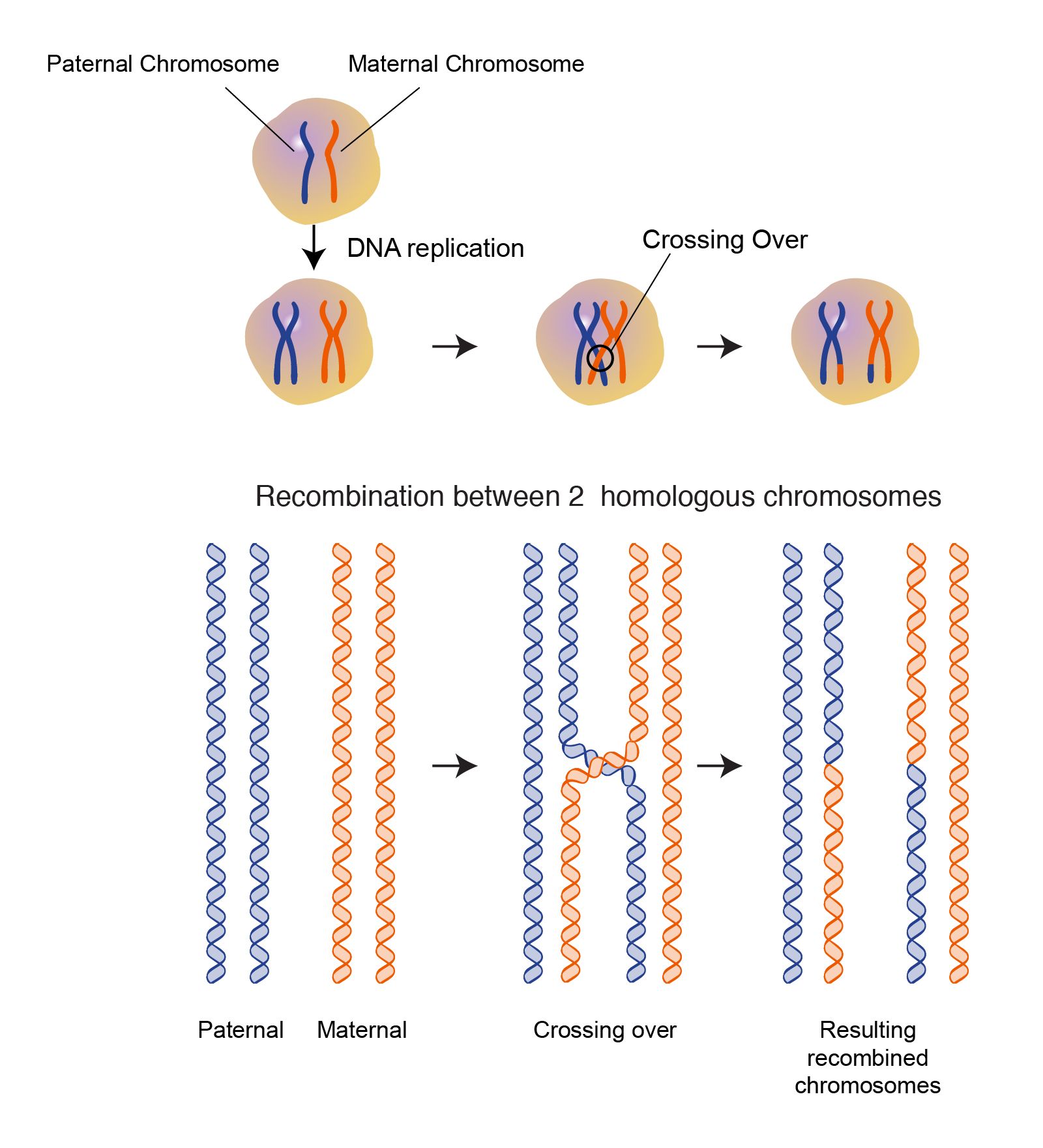
Synthetic Biology and Research Tools
Synthetic biology aims to design and construct new biological parts, devices, and systems, or to redesign existing natural biological systems for useful purposes. Homologous recombination is a foundational tool in this field, enabling the precise assembly of complex genetic circuits and the engineering of novel metabolic pathways. It is also indispensable for creating knockout and knock-in animal models used in research to study gene function and disease mechanisms. The ability to make specific, targeted genetic changes is crucial for building and testing engineered biological systems.
In conclusion, homologous recombination is far more than a simple biological process; it is a sophisticated molecular machinery essential for life as we know it. Its intricate dance of proteins ensures the faithful replication and repair of our DNA, drives evolutionary diversity, and serves as a powerful engine for technological innovation. As our understanding of HR deepens, its applications in medicine, agriculture, and beyond will undoubtedly continue to expand, shaping the future of biology and technology in profound ways.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.