In the vast and interconnected landscape of scientific inquiry, understanding the fundamental building blocks of matter is paramount. From the microscopic realm of atoms to the macroscopic world of engineered materials, chemical compounds form the bedrock upon which much of our technological progress is built. Among these compounds, a particularly important and ubiquitous class is the binary ionic compound. These substances, formed from the electrostatic attraction between oppositely charged ions derived from two different elements, are not merely academic curiosities. Instead, they are integral to a multitude of technological applications, influencing everything from the batteries powering our smartphones to the advanced ceramics used in aerospace. This exploration delves into the nature of binary ionic compounds, their formation, nomenclature, and their indispensable role in shaping the technological innovations that define our modern era.

The Essence of Ionic Bonding: A Technological Foundation
At its core, the formation of a binary ionic compound is a testament to the fundamental principles of atomic structure and electron behavior. These compounds arise from the interaction between elements with significantly different electronegativities – a measure of an atom’s ability to attract electrons. Typically, this involves a metal, which readily loses electrons to achieve a stable electron configuration, and a nonmetal, which readily gains electrons for the same reason. This electron transfer process results in the formation of ions: positively charged cations (from the metal) and negatively charged anions (from the nonmetal). The strong electrostatic attraction, known as ionic bonding, that holds these oppositely charged ions together in a crystal lattice is the defining characteristic of an ionic compound.
Electron Transfer: The Genesis of Ions
The journey to forming a binary ionic compound begins with the electron configurations of the participating elements. Elements in Group 1 (alkali metals) and Group 2 (alkaline earth metals) of the periodic table have one or two valence electrons, respectively, which are relatively easy to remove. For instance, sodium (Na), a Group 1 metal, has one valence electron. To achieve a stable octet (eight electrons in its outermost shell), it readily loses this electron to form a sodium ion, Na$^{+}$. Similarly, magnesium (Mg), a Group 2 metal, loses its two valence electrons to form a magnesium ion, Mg$^{2+}$.
On the other side of the spectrum are nonmetals, particularly those in Group 16 (chalcogens) and Group 17 (halogens). These elements are electron-deficient and have a strong affinity for electrons. Chlorine (Cl), a halogen, needs one electron to complete its octet and thus readily gains an electron to form a chloride ion, Cl$^{-}$. Oxygen (O), a chalcogen, requires two electrons and forms an oxide ion, O$^{2-}$. The driving force for this electron transfer is the attainment of a more stable, lower-energy electron configuration for both the cation and the anion. This fundamental chemical process is the initial step in creating the materials that enable countless technological advancements. The predictable nature of this electron transfer, based on periodic trends, allows chemists and material scientists to design and synthesize compounds with specific properties, a crucial aspect of technological development.
The Crystal Lattice: A Symphony of Electrostatic Forces
Once ions are formed, they do not exist in isolation. Instead, they arrange themselves in a highly ordered, three-dimensional structure known as a crystal lattice. In this lattice, each cation is surrounded by anions, and each anion is surrounded by cations, maximizing the attractive forces between oppositely charged ions and minimizing the repulsive forces between like-charged ions. This arrangement is driven by the minimization of potential energy, resulting in a stable and rigid structure. The specific arrangement of ions in the lattice determines the macroscopic properties of the ionic compound, such as its melting point, hardness, and electrical conductivity. The strong electrostatic forces within the lattice require a significant amount of energy to overcome, which is why ionic compounds typically have very high melting and boiling points. This inherent stability and resilience are highly desirable in many technological applications where materials must withstand extreme conditions.
Types of Binary Ionic Compounds: Tailoring Properties for Innovation
Binary ionic compounds are broadly classified based on the types of elements they contain. This classification is not merely an academic exercise; it provides a framework for understanding and predicting the properties of these compounds, allowing for their targeted application in diverse technological fields. The distinction between those formed by Group 1 or 2 metals and those formed by transition metals, for example, has significant implications for material design.
Type I Binary Ionic Compounds: Simple Cation Charges
Type I binary ionic compounds are formed between a metal that typically exhibits only one common ionic charge and a nonmetal. The most common examples involve metals from Group 1 (e.g., Li, Na, K) and Group 2 (e.g., Mg, Ca, Sr), as well as some metals from Group 13 like aluminum (Al). These metals have predictable charges due to their position in the periodic table. For instance, Group 1 metals consistently form +1 cations, and Group 2 metals consistently form +2 cations. Aluminum typically forms a +3 cation. The nonmetal component is usually from Groups 16 or 17, forming anions with charges like -2 or -1, respectively. The predictable nature of these ionic charges simplifies the determination of the chemical formula and the naming of these compounds. This predictability is invaluable in industrial settings where consistency and ease of identification are crucial for efficient production and quality control in technological manufacturing.
Type II Binary Ionic Compounds: Variable Cation Charges
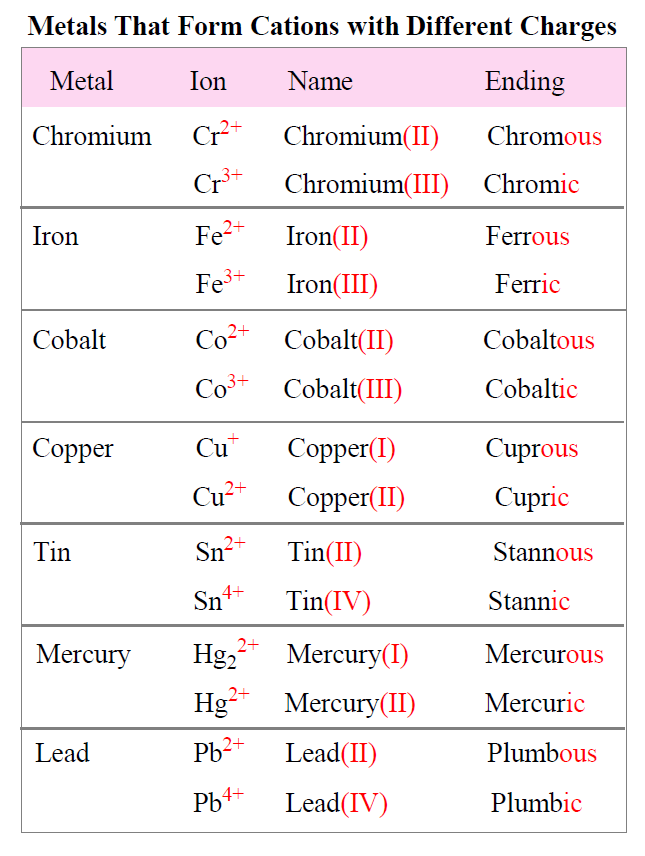
In contrast, Type II binary ionic compounds involve metals that can exhibit more than one common ionic charge. These metals are typically found in the transition metal block (Groups 3-12) and some elements in the p-block. For example, iron can exist as Fe$^{2+}$ or Fe$^{3+}$, and copper can exist as Cu$^{+}$ or Cu$^{2+}$. Because the charge of the cation is not immediately obvious from its position in the periodic table, a Roman numeral is used in the name to indicate the specific charge of the metal ion. This system of nomenclature, known as the Stock system, is essential for unambiguously identifying these compounds. The ability of these metals to form ions with different charges allows for the creation of materials with a wider range of electronic and magnetic properties, making them critical components in advanced technologies such as catalysts, pigments, and magnetic storage media.

Naming Binary Ionic Compounds: A Language of Chemical Structure
The ability to accurately name and represent binary ionic compounds is fundamental to their study and application in science and technology. A consistent nomenclature system ensures clear communication among researchers, engineers, and technicians, preventing errors in synthesis, formulation, and application. The naming convention for binary ionic compounds is systematic and follows specific rules, allowing for the deduction of the compound’s chemical formula from its name and vice versa.
Cation and Anion Naming Conventions
The naming of a binary ionic compound begins with the name of the cation, followed by the name of the anion. For simple cations, the element’s name is used directly (e.g., sodium, magnesium, aluminum). For transition metals that can exhibit multiple charges, the element’s name is followed by a Roman numeral in parentheses indicating its charge (e.g., iron(II), iron(III), copper(I)). The anion is named by taking the root of the nonmetal’s name and adding the suffix “-ide” (e.g., chloride, oxide, sulfide). For example, NaCl is named sodium chloride, and CuO is named copper(II) oxide. This systematic approach to naming ensures that each binary ionic compound has a unique identifier, facilitating its integration into databases, scientific literature, and manufacturing specifications, all critical for technological advancement.
Determining Chemical Formulas
Conversely, from the name, the chemical formula can be determined by ensuring that the total positive charge of the cations balances the total negative charge of the anions, resulting in a neutral compound. For instance, if we have calcium chloride, we know calcium forms a Ca$^{2+}$ ion, and chlorine forms a Cl$^{-}$ ion. To balance the charges, we need two chloride ions for every one calcium ion, giving the formula CaCl${2}$. For a compound like iron(III) oxide, iron(III) indicates Fe$^{3+}$, and oxide indicates O$^{2-}$. To balance these charges, we need two Fe$^{3+}$ ions (total charge +6) and three O$^{2-}$ ions (total charge -6), resulting in the formula Fe${2}$O$_{3}$. This ability to translate between names and formulas is a cornerstone of chemical understanding, enabling precise control over the composition of materials used in high-tech applications, from microelectronics to advanced composites.
Binary Ionic Compounds in Technology: Pillars of Innovation
The significance of binary ionic compounds extends far beyond theoretical chemistry; they are integral to a vast array of technological applications that shape our daily lives and drive future innovation. Their unique properties, stemming from their ionic nature and crystal structure, make them indispensable materials in numerous cutting-edge fields.
Energy Storage and Conversion: Powering the Future
One of the most impactful applications of binary ionic compounds lies in the realm of energy. Lithium-ion batteries, the powerhouses of our portable electronics and electric vehicles, rely heavily on binary ionic compounds like lithium cobalt oxide (LiCoO${2}$) and lithium iron phosphate (LiFePO${4}$). The movement of lithium ions (Li$^{+}$) through the electrolyte and electrodes is the fundamental mechanism of charge storage and release. These compounds are chosen for their ability to reversibly intercalate (insert and extract) lithium ions and for their electrochemical stability. Similarly, in solid oxide fuel cells (SOFCs), binary ionic compounds like yttria-stabilized zirconia (YSZ) act as solid electrolytes, facilitating the transport of oxygen ions (O$^{2-}$) at high temperatures, thereby converting chemical energy directly into electrical energy. The development of more efficient and sustainable energy technologies is heavily dependent on the continued discovery and engineering of novel binary ionic compounds.
Materials Science and Advanced Manufacturing: Building Tomorrow’s World
Binary ionic compounds are also foundational in materials science, contributing to the development of advanced ceramics, catalysts, and specialized coatings. Aluminum oxide (Al${2}$O${3}$), commonly known as alumina, is a prime example. Its extreme hardness, high melting point, and electrical insulation properties make it ideal for applications ranging from spark plug insulators and cutting tools to the substrates for electronic circuits. Silicon carbide (SiC), another binary ionic compound, possesses exceptional hardness, thermal conductivity, and resistance to wear and high temperatures, finding use in abrasives, semiconductor manufacturing, and high-performance automotive components. Furthermore, many binary ionic compounds serve as critical catalysts in chemical processes, enabling the efficient production of fuels, plastics, and pharmaceuticals. The precise control over stoichiometry and crystal structure of these compounds allows engineers to fine-tune their catalytic activity and selectivity, a crucial aspect of sustainable and efficient industrial processes.
Electronics and Optics: Enabling Digital and Visual Technologies
In the realm of electronics and optics, binary ionic compounds play a vital role. For instance, sodium chloride (NaCl), though simple, is used in some optical applications due to its transparency to a wide range of wavelengths. More sophisticated applications involve compounds like magnesium fluoride (MgF$_{2}$) as anti-reflective coatings on lenses and displays, enhancing light transmission and reducing glare. Certain transition metal oxides, which are binary ionic compounds, exhibit fascinating magnetic and electrical properties that are exploited in data storage devices and sensors. The continuous pursuit of smaller, faster, and more energy-efficient electronic devices relies on the ongoing research and development of novel binary ionic compounds with tailored electronic and optical characteristics.
In conclusion, binary ionic compounds represent a fundamental class of chemical substances with profound implications for the technological advancements that define our modern world. From the batteries that power our digital lives to the advanced materials that enable new forms of transportation and energy, these compounds are indispensable. Understanding their formation, nomenclature, and properties is not just an academic pursuit but a critical step in harnessing their potential for continued innovation and progress in the ever-evolving landscape of technology.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.