Atypical Teratoid Rhabdoid Tumor (ATRT) is a rare and highly aggressive form of cancer that primarily affects the central nervous system of young children. While the clinical reality of this diagnosis is sobering, the landscape of its treatment is being radically transformed by the “Tech” sector. From the application of artificial intelligence (AI) in molecular profiling to the development of high-precision radiotherapy hardware, technology is the primary driver in turning a once-unmanageable diagnosis into a field of active, data-driven hope.
This article explores the technological innovations currently redefining the fight against ATRT, focusing on how diagnostic software, robotic surgery, and bio-informatics are converging to create a new standard of care.
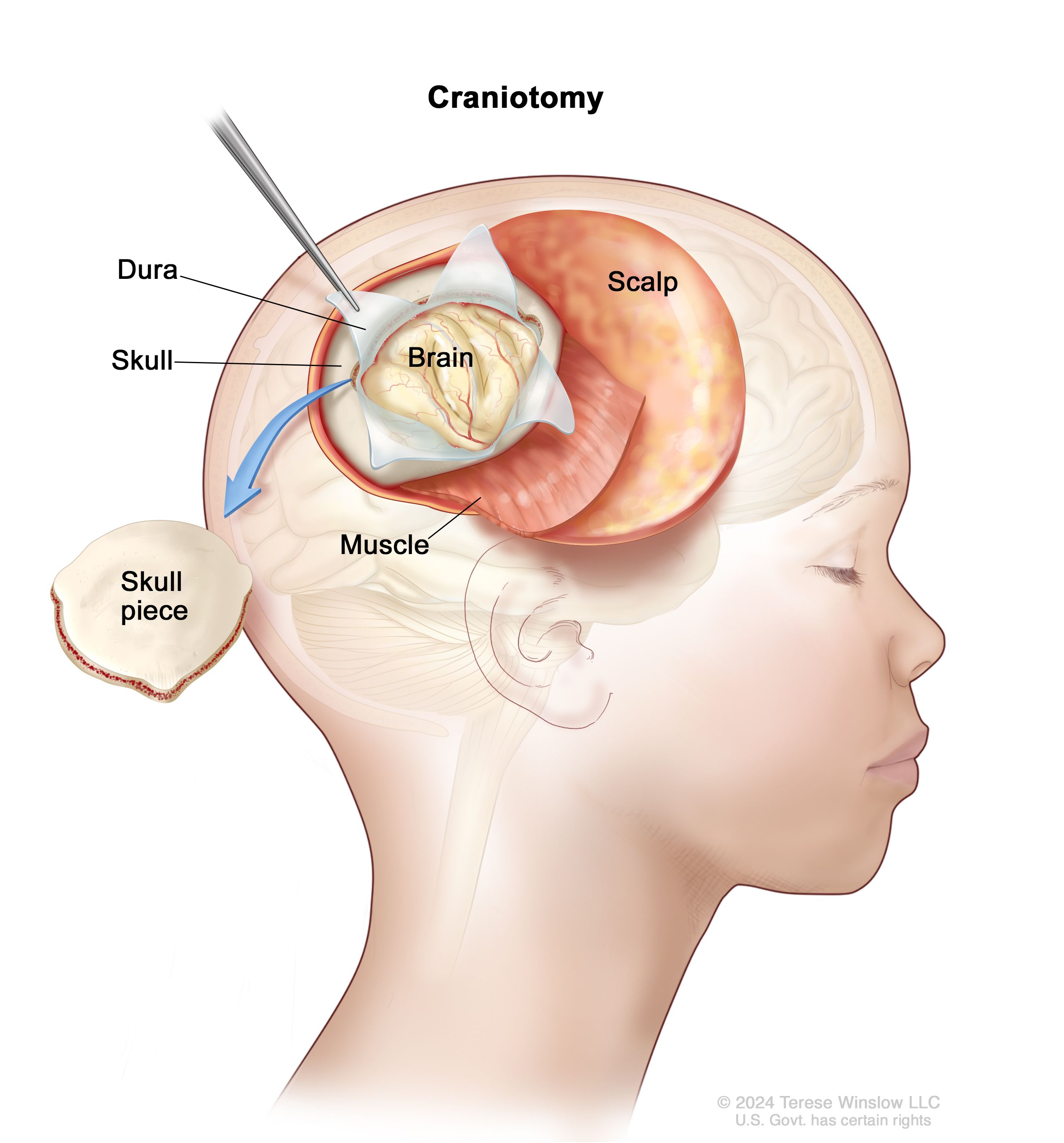
Precision Diagnostics: High-Throughput Sequencing and Molecular Tech
The most significant hurdle in treating ATRT has historically been its rapid progression and the difficulty of distinguishing it from other pediatric brain tumors. Modern technology has solved this through the integration of Next-Generation Sequencing (NGS) and advanced molecular diagnostic tools.
The Role of Next-Generation Sequencing (NGS)
At the heart of ATRT research is the SMARCB1 (or more rarely, SMARCA4) gene mutation. Tech-driven NGS platforms allow clinicians to sequence a child’s entire genome or exome within days rather than months. These high-throughput machines use complex fluidics and optical imaging to read millions of DNA fragments simultaneously. By identifying the specific genetic “signature” of ATRT at the digital level, software can confirm a diagnosis with near-total accuracy, ensuring that treatment begins based on data rather than clinical guesswork.
Epigenetic Profiling and Machine Learning
Beyond simple sequencing, “Methylation Array Technology” has become a gold standard. This involves looking at the chemical “tags” on DNA that turn genes on or off. Because ATRT has a very specific epigenetic profile, software algorithms trained on global databases can analyze these tags to categorize the tumor into specific subgroups (ATRT-SHH, ATRT-TYR, or ATRT-MYC). Machine learning models are now used to compare a patient’s unique molecular data against thousands of stored samples, predicting with high statistical confidence which subgroup the patient belongs to, which in turn dictates the specific technological intervention required.
Therapeutic Innovations: Robotics, Proton Therapy, and Targeted Drug Delivery
Once diagnosed, the technical challenge shifts to intervention. Because ATRT usually occurs in infants, the margin for error is non-existent. Traditional radiation can damage developing brain tissue, leading to the rise of highly specialized medical hardware and software.
Precision Proton Beam Therapy
Proton therapy represents a massive leap in medical physics and software engineering. Unlike traditional X-rays, which pass through the body, proton therapy uses massive particle accelerators (cyclotrons) controlled by sophisticated software to deliver a “Bragg Peak.” This is a concentrated burst of energy that stops exactly at the tumor site. The technology allows engineers to map the tumor in 3D and program the beam to avoid critical areas like the brainstem or optic nerves. The software calculates dosages to the millimeter, a feat of digital precision that was impossible two decades ago.
Nanotechnology and the Blood-Brain Barrier
One of the greatest “Tech” challenges in oncology is the blood-brain barrier (BBB), which prevents most medicines from reaching the brain. Researchers are currently developing “Nanocarriers”—microscopic technological vehicles designed to transport chemotherapy agents directly into the tumor. These nanoparticles are engineered at the molecular level to respond to specific triggers, such as the pH level of the tumor environment or an external magnetic field, releasing their payload only when they have successfully breached the target zone. This reduces systemic toxicity and maximizes the efficacy of the drug.
Artificial Intelligence and Big Data in Pediatric Oncology

The rarity of ATRT means that no single hospital has enough data to master it alone. This is where “Big Data” and cloud computing enter the frame, acting as a force multiplier for researchers around the globe.
AI-Driven Imaging Analysis
Radiologists are now supplemented by computer vision AI. When a child undergoes an MRI, AI software can analyze the scans to detect subtle changes in tumor volume or blood flow that are invisible to the human eye. By utilizing “Radiomics,” technology converts visual images into mineable data. This allows clinicians to track the “delta”—the rate of change—of a tumor in real-time, adjusting the technological treatment plan before the tumor has a chance to develop resistance.
Global Data Warehousing and Predictive Modeling
Cloud-based platforms like the “Children’s Brain Tumor Network” (CBTN) allow researchers to share genomic data and clinical outcomes across continents. Using predictive modeling, scientists can run “in silico” (on a computer) trials. Instead of testing a new drug on a patient first, they can simulate how a specific molecular structure will interact with an ATRT cell profile using high-performance computing. This accelerates the drug discovery pipeline by years, filtering out ineffective compounds before they ever reach a clinical trial.
Digital Health Platforms and Remote Patient Monitoring
The technological fight against ATRT does not end in the operating room or the lab. For survivors and those in active treatment, the focus shifts to “MedTech” for long-term monitoring and quality of life.
Telemedicine and Specialized Pediatric Care
Because ATRT is so rare, the world’s leading experts are often located in different time zones than the patients. High-definition telemedicine platforms and secure data-sharing portals allow for “Virtual Tumor Boards.” Here, surgeons, oncologists, and radiologists from around the world can view the same high-resolution 3D models of a patient’s brain in real-time, collaborating on a surgical plan via low-latency video tech. This democratization of expertise ensures that a child in a rural area has access to the same technological insights as one in a major urban center.
Wearable Tech for Post-Treatment Surveillance
For children who have completed treatment, the risk of recurrence or late-term side effects is a constant concern. Modern wearable technology is being adapted to monitor neurological health. Sensors that track gait, balance, and fine motor skills can upload data to a cloud platform. If the software detects a slight deviation in a child’s movement patterns—which might indicate a neurological shift—it can automatically alert the medical team for an early intervention MRI. This shift from “reactive” to “proactive” monitoring is entirely a product of the digital health revolution.
Future Outlook: The Intersection of Quantum Computing and Genetic Engineering
As we look toward the next decade, the “Tech” niche in ATRT research is moving toward even more futuristic horizons, specifically involving the synthesis of quantum computing and advanced gene editing.
Quantum Simulations for Drug Discovery
Current supercomputers struggle to simulate the complex folding of proteins associated with the SMARCB1 mutation. Quantum computing, however, has the potential to model these molecular interactions at the subatomic level. This could allow for the design of “designer molecules” that perfectly fit into the mutated receptors of ATRT cells, effectively “turning off” the cancer’s ability to replicate.

CRISPR and In-Vivo Gene Editing
The ultimate technological goal is not just to treat the tumor, but to fix the underlying genetic error. CRISPR-Cas9 technology is being researched as a method to “edit” the DNA of patients. While still in the experimental stages for brain tumors, the goal is to develop delivery systems (like viral vectors or lipid nanoparticles) that can carry the gene-editing machinery into the central nervous system to restore the function of the SMARCB1 gene. This represents the pinnacle of “Tech” in medicine: treating a biological problem with an engineering solution.
In conclusion, while “what is ATRT cancer” is a question rooted in biology, the answer to “how do we beat it” is increasingly found in technology. The synergy of high-speed processing, AI-driven insights, and precision engineering is transforming the landscape of pediatric neuro-oncology. As these technologies continue to mature and integrate, the outlook for children diagnosed with ATRT moves closer toward a future defined by precision, recovery, and long-term health.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.