Agarose, a fundamental component in numerous biotechnological applications, might seem like a specialized term confined to research labs. However, understanding its origins and composition unlocks a deeper appreciation for the technology that underpins many of the advancements we see in fields like genetic engineering, diagnostics, and even food science. This exploration delves into the materials from which agarose is derived, the intricate processes involved in its purification, and its critical role within the technological landscape.
The Marine Source: Unveiling Agarose’s Natural Origins
At its core, agarose is a biopolymer, a large molecule composed of repeating subunits, naturally occurring within certain marine organisms. Its primary source is seaweed, specifically a variety of red algae belonging to the Rhodophyceae class. These marine plants, often found in temperate and tropical waters, produce a complex polysaccharide called agar. Agar itself is a gel-forming hydrocolloid, and agarose is the principal purified component responsible for its unique gelling properties.

Identifying the Key Algal Families
While many species of red algae contain agar, commercially viable sources are typically concentrated within specific families known for their high agar content and desirable chemical properties. The most prominent among these include:
- Gracilaria: This genus is a significant global source of agar. Species within Gracilaria are cultivated and harvested worldwide, particularly in East Asia and South America, due to their adaptability to various marine environments and their yield of high-quality agar. The specific chemical structure of agar derived from Gracilaria often lends itself well to the production of purified agarose with excellent gel strength and clarity.
- Gelidium: Another crucial genus for agar production, Gelidium species are known for yielding agar with exceptionally strong gelling capabilities and thermal stability. Historically, Gelidium was a primary source, and while Gracilaria has gained prominence, Gelidium remains important for specific high-performance applications where superior gel properties are paramount. The cultivation and harvesting of Gelidium can be more challenging, often relying on wild harvesting in specific coastal regions.
- Pterocladia: This genus also contributes to the global agar supply. Species like Pterocladia capillacea are harvested and processed for their agar content, which possesses properties suitable for various applications, including some biotechnological uses.
The selection of a particular algal species is influenced by factors such as regional availability, cost-effectiveness of harvesting and cultivation, and the desired properties of the final agar product. The ecological impact of harvesting is also a growing consideration, driving research into sustainable aquaculture practices for these valuable marine resources.
The Polysaccharide Backbone: Understanding Agar Structure
Agar is not a single, homogenous substance but rather a mixture of two main polysaccharide fractions: agarose and agaropectin. Both are composed of repeating disaccharide units, but their chemical structures and properties differ significantly, with agarose being the critical component for advanced biotechnological applications.
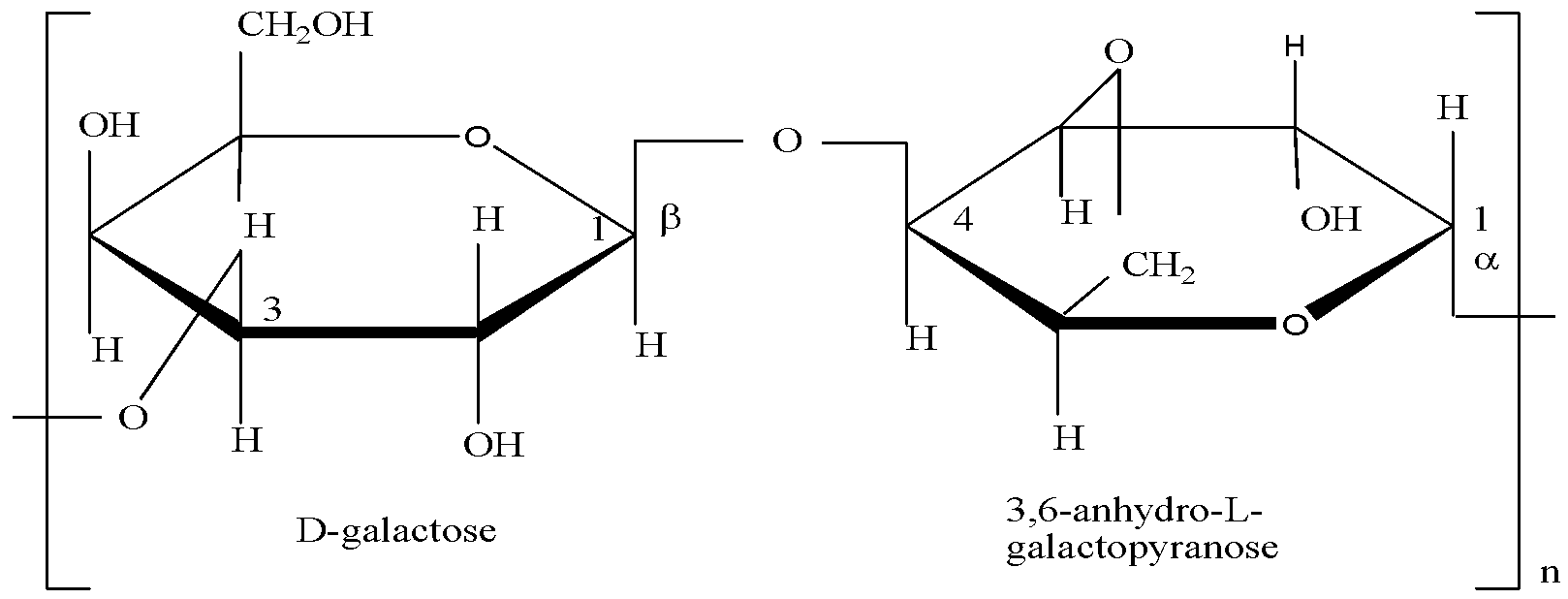
- Agarose: This fraction is primarily composed of repeating units of D-galactose and 3,6-anhydro-L-galactopyranose. The regular, linear structure of agarose allows its molecules to align in a helical fashion when dissolved in hot water. Upon cooling, these helices aggregate, forming a three-dimensional network of double helices that trap water molecules, resulting in the characteristic gel formation. Agarose is relatively neutral in charge, making it an ideal matrix for separating nucleic acids based on size without significant interaction with their negatively charged phosphate backbone.
- Agaropectin: This fraction is more complex and heterogeneous. It also contains galactose units but is heavily substituted with charged groups, such as sulfate and pyruvate. These charged groups make agaropectin more soluble in water and less prone to forming strong gels compared to agarose. Agaropectin contributes to the overall viscosity of agar but can interfere with certain biotechnological applications, particularly those requiring high purity and precise separation.
The technological significance of agarose lies precisely in its high degree of purity. For applications like gel electrophoresis, it is essential to isolate agarose from the more complex agaropectin. This isolation process is a critical step in producing the specialized agarose products used in laboratories worldwide.
The Technological Transformation: From Algae to Purified Agarose
The journey from raw seaweed to the highly purified agarose used in scientific research and diagnostics is a testament to sophisticated extraction and purification technologies. This multi-stage process is designed to remove impurities, particularly the charged agaropectin, and to achieve specific grades of agarose tailored for different applications.
Extraction: Releasing Agar from the Seaweed Matrix
The initial step involves extracting the agar from the harvested red algae. This process typically begins with cleaning the seaweed to remove sand, epiphytes, and other debris. The cleaned seaweed is then subjected to several treatments to break down cellular structures and release the agar:
- Alkaline Treatment: Often, the seaweed is treated with an alkali solution, such as sodium hydroxide. This process helps to modify the agar structure, breaking some of the glycosidic bonds and enhancing the yield of extractable agar. The alkaline treatment also plays a role in increasing the clarity and gel strength of the final product.
- Hot Water Extraction: The treated seaweed is then boiled in water, sometimes with the addition of acids or chelating agents to aid in the release of agar. The hot water dissolves the agar, forming a viscous solution. This extraction is a crucial step where the water-soluble agar polysaccharide is leached out from the algal biomass.
The temperature and duration of the extraction are carefully controlled to maximize agar yield while minimizing degradation of the polysaccharide chains. Multiple extraction cycles may be employed to ensure maximum recovery.

Purification: Isolating the Gel-Forming Powerhouse
Once the crude agar extract is obtained, the critical phase of purification begins to isolate agarose from agaropectin and other soluble components. This is where the technological sophistication becomes most apparent, as various methods are employed to achieve different purity levels.
- Precipitation and Filtration: The hot agar solution is often cooled, allowing the agar to gel. This gel is then subjected to freezing and thawing cycles, which helps to further expel water and impurities. Filtration is then used to separate the solid agar gel from the liquid phase. Repeated washing steps with water are employed to remove soluble impurities.
- Selective Precipitation: To separate agarose from agaropectin, specific chemical treatments can be used. For instance, certain salts or organic solvents can selectively precipitate agarose, leaving the more soluble agaropectin in solution. This is a key step in concentrating the agarose fraction.
- Ion-Exchange Chromatography: For exceptionally high-purity agarose, ion-exchange chromatography is often employed. This technique utilizes charged resins to bind and separate molecules based on their charge. While agarose is largely neutral, residual charged impurities (including agaropectin) can be effectively removed by passing the agarose solution through appropriate ion-exchange columns.
- Ultrafiltration and Dialysis: These membrane-based techniques are used to remove small molecules, salts, and residual impurities. Ultrafiltration uses semipermeable membranes to separate molecules based on size, while dialysis allows the diffusion of small solutes across a membrane, leaving larger molecules like agarose behind.
The choice of purification method depends on the intended application of the agarose. For routine laboratory electrophoresis, a certain level of purity is sufficient. However, for more sensitive applications, such as DNA sequencing or high-resolution protein separation, ultra-pure agarose is essential, requiring more rigorous and advanced purification techniques.
Drying and Milling: Preparing for Application
After purification, the agarose is typically in a hydrated or semi-hydrated form. The final steps involve drying and milling the purified agarose into a powder.
- Drying: Various drying methods, such as freeze-drying (lyophilization) or spray drying, are used to remove water from the agarose. Freeze-drying is often preferred for maintaining the integrity of the polysaccharide structure and ensuring good rehydration properties.
- Milling: The dried agarose is then milled into a fine powder. The particle size distribution of the powder is important for its dissolution characteristics and ease of use in laboratory settings. Uniform particle size ensures consistent and reproducible gel formation.
The resulting agarose powder is then packaged under controlled conditions to prevent moisture absorption and contamination, ready for distribution to laboratories and industries worldwide. The entire process, from harvesting seaweed to producing a high-grade agarose powder, represents a sophisticated interplay of biological sourcing and advanced chemical engineering.
Agarose in Action: Enabling Technological Advancements
The intricate process of producing agarose is justified by its indispensable role in a wide array of technological applications. Its unique ability to form stable, porous gels makes it an ideal matrix for separation, immobilization, and containment, driving innovation across numerous scientific disciplines.
Gel Electrophoresis: Visualizing the Blueprint of Life
Perhaps the most widely recognized application of agarose is in gel electrophoresis, a cornerstone technique in molecular biology. Agarose gels serve as a molecular sieve, allowing scientists to separate DNA and RNA fragments based on their size and charge.
- DNA and RNA Separation: When an electric field is applied across an agarose gel, negatively charged nucleic acid molecules migrate through the pores of the gel. Smaller fragments can move more readily through the gel matrix, traveling further and faster than larger fragments. This differential migration allows for the visualization and analysis of DNA or RNA populations, essential for gene cloning, PCR product analysis, DNA fingerprinting, and diagnostic testing.
- Gel Strength and Pore Size: The concentration of agarose used directly influences the gel strength and pore size. Higher agarose concentrations create denser gels with smaller pores, ideal for separating smaller nucleic acid fragments. Conversely, lower concentrations produce less dense gels with larger pores, suitable for separating larger DNA molecules. The purity of the agarose is critical here; impurities can lead to inconsistent pore sizes and distorted separation patterns.
The development of specialized agarose grades, such as molecular biology grade or low melting point agarose, further enhances its utility in electrophoresis. Molecular biology grade agarose is highly purified to minimize interference with enzymatic reactions, while low melting point agarose allows for easier manipulation and recovery of DNA fragments after electrophoresis.
Other Biotechnological Applications: Beyond Electrophoresis
The utility of agarose extends far beyond gel electrophoresis, enabling a diverse range of advanced biotechnological applications:
- Cell Culture and Tissue Engineering: Agarose can be used as a scaffold for growing cells and tissues in vitro. Its biocompatibility and ability to form a soft, hydrated matrix make it suitable for creating 3D cell cultures, which better mimic the in vivo environment. This is crucial for studying cell behavior, drug screening, and regenerative medicine.
- Immobilization of Enzymes and Antibodies: The porous nature of agarose gels allows for the immobilization of enzymes, antibodies, and other biomolecules. This process entraps the biomolecules within the gel matrix, maintaining their activity while providing a stable support. Immobilized enzymes are widely used in industrial bioprocesses for biocatalysis, while immobilized antibodies are key components in immunoassays and biosensors for detecting specific analytes.
- Microbiology and Food Science: In microbiology, agar is the primary solidifying agent in culture media, providing a nutrient-rich surface for bacterial growth. While purified agarose is not typically used for standard microbiological media (where whole agar is employed), its principles underpin the technology. In food science, purified agar can be used as a gelling agent in specific applications requiring high clarity and texture control, although it is less common than in biotechnology.
- Chromatography Media: Agarose-based beads are extensively used as stationary phases in various chromatographic techniques, particularly affinity chromatography. These beads are often chemically modified to create specific binding sites for target molecules, enabling the purification of proteins, antibodies, and other biomolecules with high specificity.
The continued development of new purification techniques and chemical modifications of agarose promises to further expand its applications, solidifying its status as a vital material in the ongoing evolution of biotechnology and related fields. The seemingly simple origin of agarose from the depths of the ocean belies its profound impact on modern scientific inquiry and technological progress.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.