In the rapidly evolving landscape of biotechnology and nanotechnology, the line between biological systems and engineering continues to blur. At the center of this convergence lies a fundamental biological component that functions remarkably like a piece of high-end hardware: the integral membrane protein (IMP). While traditional biology might view these as mere cellular structures, modern technology perceives them as sophisticated gatekeepers, sensors, and transducers that manage the “input/output” operations of the living cell.
Understanding what an integral membrane protein is—and how we are currently leveraging that knowledge through AI, computational modeling, and bio-electronic interfaces—is essential for understanding the future of health-tech and synthetic biology.
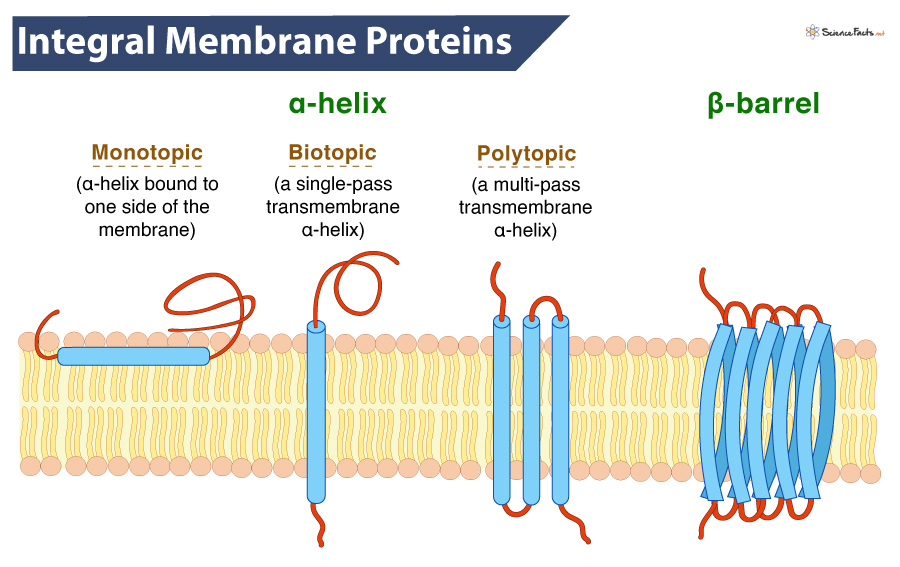
The Biological Hardware: Understanding the Structure of Integral Membrane Proteins (IMPs)
To understand integral membrane proteins from a tech perspective, one must first view the cell membrane as a sophisticated firewall or a semi-permeable boundary. The membrane itself is a lipid bilayer, an oily barrier that separates the internal “data” of the cell from the chaotic external environment. Integral membrane proteins are the specialized components embedded directly into this hardware.
The Transmembrane Domain: Engineering Nature’s Circuitry
Unlike peripheral proteins, which merely sit on the surface, integral membrane proteins are permanently anchored within the lipid bilayer. This is achieved through transmembrane domains—segments of the protein specifically “coded” with hydrophobic amino acids. In engineering terms, this is a masterpiece of material science. These domains allow the protein to span the entire width of the membrane, creating a physical bridge between the inside and the outside of the cell.
This spanning capability is what allows IMPs to function as the cell’s primary communication interface. They are the physical “ports” through which signals and molecules are processed, much like a USB-C port facilitates both power delivery and data transfer for a laptop.
From Alpha-Helices to Beta-Barrels: The Logic of Protein Design
The structural architecture of these proteins generally falls into two “design patterns”: alpha-helices and beta-barrels. Alpha-helical proteins are the most common and are often involved in signal transduction. Beta-barrels, on the other hand, frequently form pores or channels.
From a structural engineering standpoint, the stability of these designs is paramount. Because the interior of the cell membrane is highly hydrophobic (water-repelling), the protein must fold in a way that shields its sensitive parts while exposing its “anchor” points. This spatial logic is now being used by bio-engineers to design synthetic proteins that can survive in non-biological environments, such as industrial sensors or filtration systems.
The Role of AI and Machine Learning in Protein Folding (AlphaFold and Beyond)
For decades, identifying the structure of an integral membrane protein was one of the “Grand Challenges” of biology. Because these proteins are embedded in lipids, they are notoriously difficult to crystallize for traditional X-ray imaging. However, the tech industry recently provided a solution that has fundamentally changed the field: Deep Learning.
Solving the Crystallography Problem with Computational Power
Google DeepMind’s AlphaFold and its successors have revolutionized our understanding of IMPs. By treating protein sequences as a language and using transformer-based neural networks (similar to the technology behind LLMs), AI can now predict the 3D structure of an integral membrane protein with atomic-level accuracy.
This shift from “wet lab” experimentation to “dry lab” computation has accelerated drug discovery by orders of magnitude. We no longer need to spend years trying to physically map a protein; we can simulate its structure in a digital environment, allowing researchers to identify potential “docking sites” for new medications in a matter of hours.
Deep Learning and the Prediction of Membrane Topology
Beyond simple structure, AI tools are now being used to predict how these proteins interact with the complex lipid environment. This is known as “membrane topology.” In the tech world, this is equivalent to running a stress test on a new hardware component within a simulated operating system. By predicting how an IMP will flex, rotate, and respond to voltage changes within the membrane, tech companies are creating “digital twins” of cellular interfaces, enabling the design of more effective biological-digital bridges.
Bio-Technology Applications: IMPs as the Frontier of Nanotech and Sensors
The unique properties of integral membrane proteins have made them a focal point for the development of new gadgets and diagnostic tools. We are moving away from seeing IMPs as subjects of study and toward using them as actual components in technological devices.
Nanopore Sequencing: Using Proteins for Real-Time Data Processing
One of the most successful applications of IMP technology is Nanopore sequencing. Companies like Oxford Nanopore Technologies use an integral membrane protein—specifically a pore-forming protein—as a sensor. As a strand of DNA is pulled through the protein pore, it causes characteristic disruptions in an electrical current.
This is essentially a biological “reader head” for genetic data. It allows for portable, real-time DNA sequencing that can be plugged into a laptop via USB. This integration of a biological protein into a silicon-based device is perhaps the purest example of how IMPs are being commodified as “Bio-Tech” hardware.
Biosensors and Bio-Electronic Interfaces
Researchers are currently developing biosensors that use integral membrane proteins to detect specific environmental pollutants or pathogens. By wiring an IMP to a graphene transistor, a change in the protein’s shape (caused by binding to a target molecule) can be instantly converted into a digital signal. This “lab-on-a-chip” technology relies entirely on the structural integrity and sensitivity of the integral membrane protein, effectively turning a biological gateway into a digital switch.
Pharmaceutical Tech: The Software Approach to Drug Discovery
In the pharmaceutical industry, integral membrane proteins are the “High-Value Targets.” Over 50% of all modern drugs target these proteins because they are the most accessible points for intervention.
G-Protein Coupled Receptors (GPCRs) as Functional APIs
G-Protein Coupled Receptors (GPCRs) are a massive class of integral membrane proteins that act as the cell’s Application Programming Interface (API). They wait for an external ligand (a signal) and then trigger a specific “callback function” inside the cell.
Modern pharmacology is increasingly taking a “software” approach to these receptors. Rather than using “brute-force” chemical testing, researchers use molecular docking software to design small molecules that act like custom-coded keys for these GPCR ports. This precision reduces side effects and increases the efficiency of the drug, much like optimizing code to reduce CPU overhead.
The Future of Precision Medicine through Synthetic Biology
With the advent of CRISPR and advanced synthetic biology, we are now entering an era where we can “program” our own integral membrane proteins. Tech-driven startups are looking into designing custom IMPs that can be inserted into a patient’s cells to act as “smart” receptors. These engineered proteins could, for example, detect the presence of cancer markers and automatically trigger the production of an immune response. This represents the ultimate convergence of software logic and biological matter.
The Security and Ethical Landscape of Bio-Digital Convergence
As we master the ability to model, manipulate, and manufacture integral membrane proteins, we must also address the digital and biological security implications. When biological systems become programmable, they also become vulnerable to “exploits.”
Biosecurity in the Era of Programmable Matter
The ability to design custom pores or channels in a membrane could, in theory, be used to create novel toxins or bypass natural cellular defenses. This has led to the emergence of “Biosecurity Tech,” where AI systems are used to monitor synthetic DNA orders to ensure that no one is attempting to manufacture dangerous integral membrane proteins.
Just as we secure our digital networks with encryption and firewalls, the future of biotechnology will require “biological firewalls” to protect the integrity of the integral membrane proteins that keep our cells functioning. The intersection of cybersecurity and synthetic biology is set to become one of the most critical tech sectors of the next decade.
In conclusion, the integral membrane protein is far more than a biological definition; it is a sophisticated piece of nano-machinery that sits at the heart of the modern biotech revolution. From AI-driven structural predictions to the development of nanopore “hardware,” our ability to understand and interface with these proteins will define the next generation of technological advancement. Whether as drug targets, sensors, or the building blocks of synthetic life, IMPs are the essential interfaces of the 21st century.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.