The fundamental properties of elements, often explored through the lens of their atomic radii, are not merely academic curiosities. In the rapidly evolving landscape of technology, a deep understanding of these atomic characteristics is becoming increasingly crucial for innovation. From the development of advanced semiconductors and novel catalysts to the creation of ultra-lightweight yet strong materials, the size of an atom directly influences its behavior and, consequently, the performance and capabilities of the technologies it enables. This article delves into the element possessing the largest atomic radius and explores the profound technological implications stemming from this characteristic.

The Champion of Size: Understanding Francium and its Atomic Radius
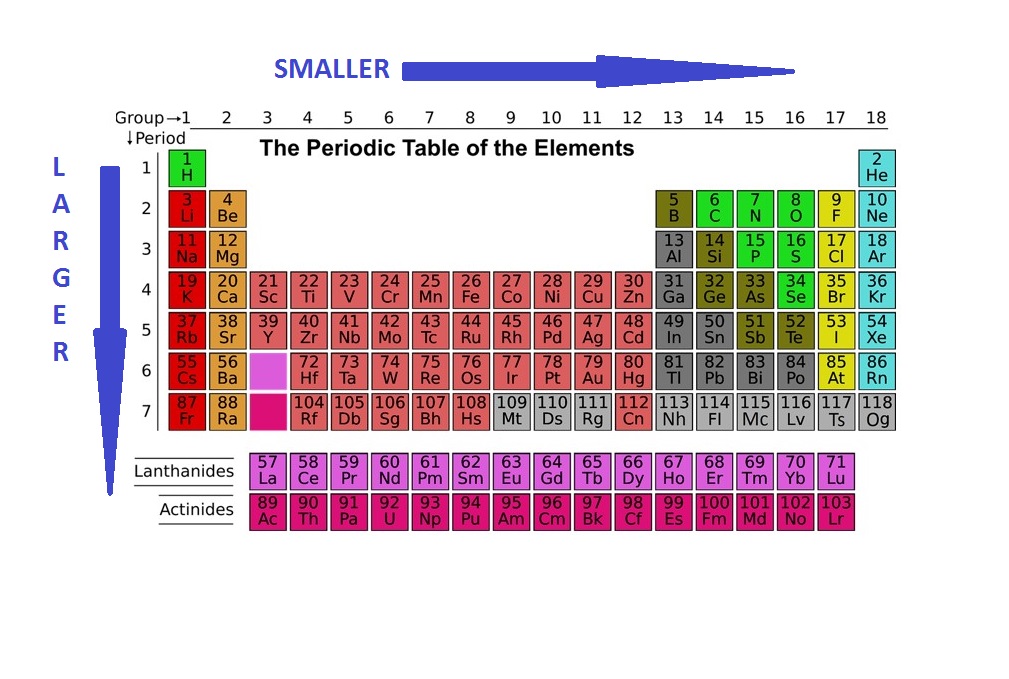
To identify the element with the largest atomic radius, we must journey to the far reaches of the periodic table, specifically to the alkali metals. These elements, located in Group 1, are known for their highly reactive nature and their tendency to readily lose their outermost electron. As we descend Group 1, a predictable trend emerges: atomic radius increases. This phenomenon is due to several key factors.
Electron Shell Expansion and Shielding Effects
The primary driver behind increasing atomic radius down a group is the addition of electron shells. Each new period on the periodic table signifies the filling of an additional principal energy level, placing valence electrons further from the nucleus. For instance, lithium (Period 2) has its valence electron in the second shell, while sodium (Period 3) has it in the third, and potassium (Period 4) in the fourth. This outward expansion of electron shells directly correlates with a larger atomic radius.
Furthermore, the inner electrons effectively “shield” the outer valence electrons from the full attractive force of the positively charged nucleus. As more electron shells are added, this shielding effect becomes more pronounced. While the nuclear charge also increases down a group, the combined effect of increased distance and enhanced shielding means that the valence electrons are held less tightly, resulting in a larger, more diffuse electron cloud and thus a greater atomic radius.
Francium: The Apex of Atomic Size
Following this trend, Francium (Fr) emerges as the element with the largest atomic radius. Occupying the bottom of Group 1 and belonging to Period 7, Francium’s outermost electron resides in the seventh electron shell. This extreme distance from the nucleus, coupled with the significant shielding provided by the six inner electron shells, grants Francium its exceptionally large atomic radius.
However, it is crucial to note that Francium is an extremely rare and highly radioactive element. Its most stable isotope has a half-life of only about 22 minutes, meaning it decays rapidly. This inherent instability and scarcity significantly limit its direct practical applications in bulk material technologies. Despite this, the principle of large atomic radius, exemplified by Francium, has profound implications for materials science and technology.
Technological Relevance of Large Atomic Radii
While Francium itself may not be found in your smartphone or the chassis of an electric vehicle, the understanding of why it possesses such a large atomic radius is fundamental to designing and engineering new materials with specific, desirable properties. The concept of atomic size influences how atoms interact with each other, dictating the spacing within crystal lattices, the strength of interatomic bonds, and the overall physical and chemical behavior of a substance.
Materials Science and Nanotechnology: Tailoring Properties
The ability to precisely control the size and arrangement of atoms is at the heart of modern materials science and nanotechnology. Researchers leverage the understanding of atomic radii to:
-
Develop Advanced Alloys: By alloying different metals, engineers can create materials with enhanced strength, corrosion resistance, or conductivity. The atomic radii of the constituent elements play a critical role in determining the resulting crystal structure and properties of the alloy. For instance, introducing larger or smaller atoms into a metallic lattice can distort it, leading to hardening or changes in electrical behavior.
-
Design Catalysts: Catalysts are essential for a vast array of industrial processes, from fuel refining to chemical synthesis. The surface area and electronic properties of a catalyst are paramount, and these are directly influenced by the atomic size of the constituent elements. Elements with larger atomic radii might offer more accessible active sites for chemical reactions or create specific electronic environments conducive to particular transformations.
-
Explore Novel Semiconductor Materials: The performance of electronic devices is dictated by the properties of semiconductor materials. While silicon remains dominant, research into alternative semiconductor alloys and structures often involves elements with varying atomic radii to fine-tune band gaps, carrier mobility, and thermal conductivity. Understanding how atomic size affects interatomic distances and bonding is crucial for predicting and controlling these electronic properties.
- Create Lightweight, High-Strength Composites: In aerospace and automotive industries, there is a constant demand for materials that are both strong and lightweight. The incorporation of elements with specific atomic radii into composite structures can influence the interfacial adhesion between different materials and the overall mechanical integrity of the final product.
Nanoscale Engineering and Quantum Effects
At the nanoscale, where the behavior of matter deviates significantly from macroscopic observations, atomic radii become even more critical. Nanotechnology deals with structures and devices on the order of nanometers, often comprising only a few thousand atoms. In this realm, individual atoms and their relative sizes dictate the unique quantum mechanical properties that emerge.
-
Quantum Dots and Nanoparticles: The synthesis of quantum dots and other nanoparticles with specific optical and electronic properties relies heavily on controlling the size of the constituent atoms and how they assemble. The precise spacing between atoms, influenced by their radii, determines the energy levels within these nanostructures, leading to tunable light emission or absorption characteristics. This is vital for applications in display technology, solar cells, and bio-imaging.
-
Atomic Layer Deposition (ALD) and Epitaxy: These advanced fabrication techniques allow for the precise deposition of atomic layers onto substrates. The controlled growth of thin films and nanostructures using ALD and epitaxy is fundamentally guided by the atomic radii of the precursor elements and the resulting interatomic distances in the deposited material. This enables the creation of complex heterostructures with tailor-made electronic and optical functionalities.
-
Surface Science and Adsorption: In heterogeneous catalysis and sensor technology, the interaction of molecules with material surfaces is key. The size of the surface atoms, dictated by their atomic radii, influences the binding energy and orientation of adsorbed molecules, impacting reaction rates and sensing sensitivity.
Challenges and Opportunities Presented by Atomic Size
The pursuit of technological advancement is intrinsically linked to mastering the control and manipulation of matter at the atomic level. While large atomic radii, as exemplified by Francium, highlight the upper bounds of atomic size, the challenges and opportunities lie in understanding and utilizing the spectrum of atomic sizes.
Precision in Fabrication and Simulation
The ability to predict and control the precise arrangement of atoms with varying radii is paramount. This requires sophisticated computational tools and experimental techniques.
-
Computational Chemistry and Materials Modeling: Modern computing power allows for the simulation of atomic interactions and the prediction of material properties based on fundamental atomic characteristics, including atomic radii. Density functional theory (DFT) and other quantum mechanical simulation methods are indispensable for designing new materials and understanding complex phenomena at the atomic scale. These simulations can predict how atoms of different sizes will pack, bond, and behave under various conditions, saving significant experimental time and resources.
-
Advanced Characterization Techniques: Techniques like Atomic Force Microscopy (AFM) and Transmission Electron Microscopy (TEM) provide unprecedented views of materials at the atomic level. These tools enable researchers to verify theoretical predictions and to directly observe how atoms of different sizes arrange themselves within a material. This feedback loop between theory and experiment is crucial for refining our understanding and capabilities.
Exploring the Extremes: From Tiny to Tremendous
While Francium represents one extreme of atomic size, the exploration of elements with very small atomic radii (like Helium or Neon) also presents unique technological opportunities. These elements are often used in creating inert atmospheres for sensitive electronic manufacturing or as coolants in high-performance computing. The interplay between the smallest and largest atomic radii, and the vast range in between, offers a rich palette for technological innovation.
-
Designing for Specific Interatomic Distances: Many technological applications require materials with specific interatomic distances. For example, in the development of certain types of lasers or optical fibers, precise spacing between atoms is critical for light-matter interactions. By selecting elements with appropriate atomic radii, scientists can engineer these precise distances.
-
Understanding Bonding Strength and Material Properties: The strength of chemical bonds between atoms is influenced by their size and the overlap of their electron orbitals. Elements with larger atomic radii often form weaker bonds due to greater electron cloud diffusion. This understanding is vital for designing materials that can withstand specific mechanical stresses or environmental conditions.

Conclusion: The Enduring Significance of Atomic Radius in Technological Progress
In conclusion, while Francium holds the title for the element with the largest atomic radius, its direct utility is limited by its extreme instability. However, the principle it represents – the profound impact of atomic size on material properties – is a cornerstone of modern technological advancement. From the microscopic realm of quantum dots to the macroscopic performance of advanced alloys, the careful consideration and manipulation of atomic radii are indispensable for designing the next generation of groundbreaking technologies. As our ability to probe, predict, and engineer matter at the atomic level continues to advance, the seemingly simple concept of an atom’s size will undoubtedly remain a critical driver of innovation across the entire spectrum of technological disciplines.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.