In the high-stakes world of global pharmaceuticals, the “classification” of a product often dictates its destiny. While a scientist might view classification through the lens of molecular structures and neurotransmitter receptors, a brand strategist views it as the foundational pillar of market positioning. Seroquel (quetiapine) represents one of the most significant case studies in modern brand management. Developed by AstraZeneca, Seroquel transcended its chemical roots to become a multi-billion-dollar brand, a feat achieved not just through chemistry, but through a masterful orchestration of brand identity, strategic classification, and lifecycle management.
To understand what classification Seroquel belongs to from a branding perspective is to understand the evolution of the “Blockbuster” drug. This analysis explores how Seroquel was classified within the market, how its identity was crafted to dominate a competitive landscape, and the strategic maneuvers that allowed it to maintain brand equity long after its patent expired.
The Brand Classification of Blockbuster Pharmaceuticals
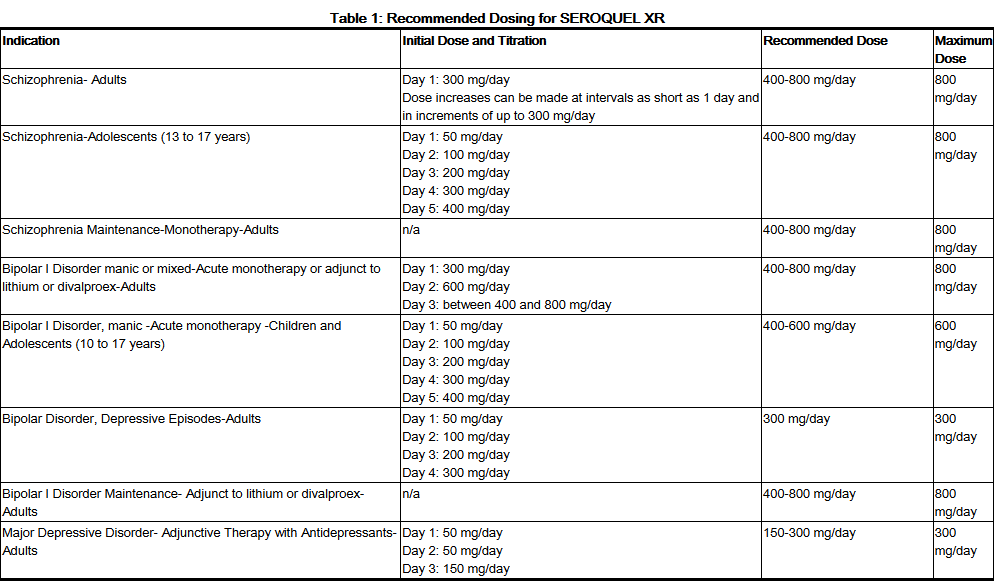
In the pharmaceutical industry, a “Blockbuster” is more than a sales figure; it is a classification of status. For a drug to reach this tier—typically defined by exceeding $1 billion in annual revenue—it must possess a brand identity that appeals to a broad cross-section of stakeholders, including healthcare providers (HCPs), insurers, and patients.
Defining the “Atypical” Brand Identity
Seroquel’s primary brand classification is that of an “atypical antipsychotic.” From a marketing standpoint, the word “atypical” was a gift. It allowed AstraZeneca to differentiate the product from the “typical” first-generation antipsychotics which were often associated with severe, visible side effects and a “heavy-handed” clinical feel. The “atypical” classification allowed Seroquel to be positioned as a modern, sophisticated, and more “human-centric” solution. The brand identity was built around the promise of efficacy without the same degree of motor-function impairment, positioning it as a premium choice in the mental health space.
Moving Beyond Clinical Labels to Consumer Perception
While its clinical classification was rigid, its brand perception was designed to be fluid. AstraZeneca did not want Seroquel to be pigeonholed as a treatment for only the most severe conditions like schizophrenia. Instead, the brand strategy involved “broadening the tent.” By pursuing classifications for bipolar disorder and later seeking adjunct status for major depressive disorder, the brand evolved from a niche psychiatric tool into a versatile mental health “utility” brand. This strategic expansion of its classification allowed it to tap into much larger patient populations, significantly increasing its market penetration.
Strategic Positioning: How Seroquel Carved Its Niche
A brand’s classification is only as effective as its positioning. Seroquel entered a market already occupied by heavyweights like Risperdal and Zyprexa. To survive and thrive, AstraZeneca’s brand team had to identify a “White Space” in the market—a specific need that competitors were not meeting effectively.
The Transition from Niche to Mass Market
Seroquel’s positioning strategy focused heavily on its sedative properties, which, while technically a side effect, were branded as a benefit for patients suffering from the agitation and insomnia associated with mood disorders. This “benefit-led” branding allowed the drug to be classified in the minds of clinicians as the go-to option for “agitated” patients. By owning the “agitation and sleep” niche within the broader antipsychotic category, Seroquel created a distinct brand personality that was more approachable than its more “clinical-feeling” rivals.
Differentiation in a Crowded Competitive Landscape
In brand strategy, differentiation is the key to avoiding commoditization. Seroquel’s differentiation was fueled by its “Seroquel XR” (extended release) formulation. This wasn’t just a clinical update; it was a rebranding exercise. The XR classification allowed the brand to refresh its visual identity, update its messaging, and—most importantly—extend its market exclusivity. By positioning the XR version as a more convenient, once-daily “lifestyle-friendly” option, AstraZeneca effectively re-classified the drug from an acute intervention to a chronic management tool, fostering long-term brand loyalty.
The Lifespan of a Brand: From Patent Protection to Generic Competition

Every pharmaceutical brand faces the “Patent Cliff”—the moment its legal protection ends and generic competitors enter the market. The way a brand handles this transition determines whether its classification shifts from “Premium Asset” to “Legacy Commodity.”
Maintaining Brand Equity Post-Exclusivity
When Seroquel lost its patent protection, it faced a sudden influx of quetiapine generics. However, the Seroquel brand name carried significant equity. In the minds of many patients and physicians, the “Seroquel” brand represented a known quantity of quality and reliability that generic versions lacked. AstraZeneca’s strategy involved maintaining a “Premium Generic” presence in certain markets and utilizing the Seroquel XR brand to keep patients within the proprietary ecosystem. This approach highlights how a strong brand classification can act as a shield against price-driven competition.
The Impact of Generic “Classifications” on Brand Loyalty
The pharmaceutical market often sees a “split” in brand loyalty once a drug goes generic. There is the “Economic Class,” which prioritizes cost, and the “Brand Loyalists,” who remain attached to the original name. AstraZeneca successfully navigated this by reinforcing the trust associated with the Seroquel name. Even today, the “Seroquel” brand remains a dominant search term and a household name, proving that the brand classification has outlasted the chemical exclusivity.
Corporate Identity and the Role of AstraZeneca
A product’s brand does not exist in a vacuum; it is deeply tied to the corporate identity of its parent company. Seroquel became the flagship of AstraZeneca’s neuroscience portfolio, and its success (and controversies) significantly shaped the company’s global reputation.
Synergy Between Corporate and Product Branding
AstraZeneca’s corporate brand—focused on innovation and “Pushing the Boundaries of Science”—was perfectly aligned with Seroquel’s market entry. The success of Seroquel provided the capital and the reputational “clout” for AstraZeneca to acquire other assets and expand its R&D. In turn, the company’s global reach ensured that Seroquel was classified as a “Global Standard of Care,” available in over 80 countries. This synergy between the corporate entity and the product brand created a feedback loop of perceived authority and reliability.
Crisis Management and Brand Reputation in the Pharma Space
No discussion of Seroquel’s brand classification is complete without addressing the challenges of litigation and off-label marketing controversies. From a branding perspective, these incidents threatened to re-classify Seroquel as a “Controversial” or “Risky” brand. AstraZeneca’s response—a combination of massive legal settlements and a pivot toward more transparent, data-driven communication—is a masterclass in brand damage control. By refocusing the narrative on patient outcomes and the rigors of FDA-approved indications, the company managed to preserve the brand’s core value, preventing a permanent “re-classification” into the territory of failed or dangerous products.
The Future of Pharmaceutical Branding in a Digital Age
As we look toward the future, the way we classify and brand drugs like Seroquel is changing. The rise of digital health, personalized medicine, and AI-driven diagnostics is creating new categories for established brands.
Personalization and the Next Evolution of Drug Classifications
We are moving away from “one-size-fits-all” brand classifications. In the future, a drug like Seroquel may be classified not just by its chemical category, but by the “Patient Persona” it best serves, identified through genetic markers or digital biomarkers. The branding of the future will be hyper-targeted. Seroquel’s legacy provides the blueprint for this: a brand that started with a specific clinical classification and, through strategic marketing, evolved into a versatile, multi-faceted identity that stood the test of time.
Conclusion: The Lasting Legacy of the Seroquel Brand
“What classification is Seroquel?” The answer depends on who you ask. To a pharmacist, it is an atypical antipsychotic. To an investor, it was a legendary revenue driver. But to a brand strategist, Seroquel is a masterclass in Category Leadership. It is a brand that took a complex clinical classification and transformed it into a clear, compelling, and highly profitable market position.
By understanding the interplay between clinical science and brand strategy, Seroquel moved beyond being a mere pill to becoming a fundamental component of the mental health landscape. Its story serves as a reminder that in any industry—whether Tech, Money, or Pharma—the way you classify your brand is the way the world will ultimately value it. AstraZeneca didn’t just sell a chemical compound; they built an enduring brand identity that survived the rigors of regulation, competition, and time.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.