In the rapidly evolving landscape of medical technology, the question “what classification is amlodipine” transcends the boundaries of simple biology and enters the realm of data science, informatics, and software engineering. While a physician views amlodipine as a clinical tool for managing hypertension, a healthtech architect views it as a specific entry in a complex database—a string of metadata governed by global standards.
Understanding the technical classification of amlodipine is essential for developing electronic health records (EHRs), pharmacy management systems, and AI-driven diagnostic tools. This article explores how modern technology categorizes this crucial compound, the digital frameworks that support its identification, and how machine learning is redefining pharmacological taxonomies.

The Informatics of Pharmacology: How Data Systems Categorize Amlodipine
At its core, the classification of a drug in a digital environment is not about words, but about hierarchical codes. In the world of healthtech, “classification” refers to the placement of a substance within a structured taxonomy that software can interpret.
The ATC Classification System and Data Interoperability
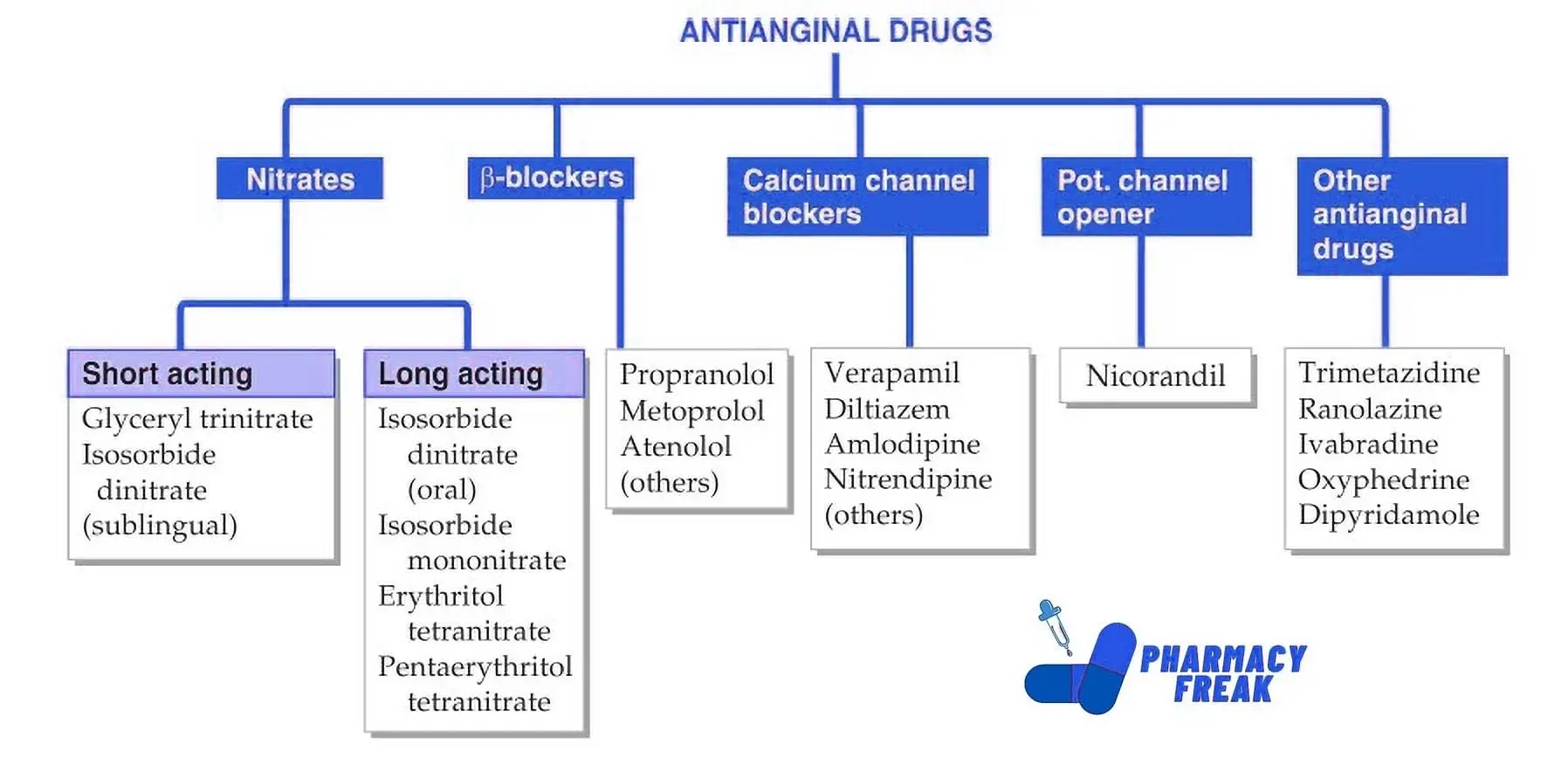
The most prominent technical framework for drug classification is the Anatomical Therapeutic Chemical (ATC) system, managed by the World Health Organization (WHO). In this digital hierarchy, amlodipine is classified under the code C08CA01.
To a piece of software, this code is far more useful than the name “amlodipine.” The code “C” tells the system it belongs to the Cardiovascular System; “C08” identifies it as a Calcium Channel Blocker; “C08C” specifies it as a selective calcium channel blocker with mainly vascular effects; and “C08CA” narrows it down to dihydropyridine derivatives. This nested data structure allows EHRs to perform automated cross-referencing, ensuring that if a patient is flagged for cardiovascular issues, the software can instantly identify all medications within the “C” category.
Relational Databases and Medication Metadata
In modern software architecture, amlodipine exists as a primary key or an object within a relational database. When a developer builds a pharmacy app, they rely on databases like RxNorm or the National Drug Code (NDC) directory.
Amlodipine’s classification within these databases includes metadata such as molecular weight, half-life, and contraindications. For developers, the “classification” of amlodipine is a vital attribute that dictates how the UI displays the drug, how the backend calculates dosage warnings, and how the API interacts with insurance databases for billing.
AI and Machine Learning in Drug Categorization
The traditional classification of amlodipine is static, but artificial intelligence is introducing a dynamic layer to how we understand this medication. AI tools are now used to re-classify drugs based on predictive modeling and chemical structure analysis.
Predictive Modeling for Calcium Channel Blockers
Machine learning algorithms, particularly those utilizing deep learning, can analyze the chemical structure of amlodipine (a dihydropyridine derivative) to predict its behavior in various simulated environments. In tech-driven drug discovery, “classification” moves from “what it is” to “what it can do.”
By using neural networks to process quantitative structure-activity relationship (QSAR) data, researchers can classify amlodipine’s efficacy relative to newer compounds. This technical classification helps bio-informatics software identify potential “off-target” effects that manual classification might overlook.
Pattern Recognition in Chemical Structures
Computer vision and pattern recognition software allow for the automated classification of drugs based on molecular imaging. For amlodipine, AI tools identify the 1,4-dihydropyridine ring structure. This digital fingerprinting is essential for quality control in automated pharmaceutical manufacturing and for identifying counterfeit medications. When a high-speed camera on a production line scans a tablet, the software classifies it in milliseconds, ensuring that the physical product matches the digital record.
Data Security and Interoperability in Clinical Software
When we discuss the classification of amlodipine in a technical context, we must also address how that information is securely transmitted and interpreted across different software platforms. This is where standards like FHIR (Fast Healthcare Interoperability Resources) become critical.
FHIR Standards and Medication Coding
FHIR is a standard for exchanging healthcare information electronically. When a hospital’s system sends a prescription for amlodipine to a retail pharmacy’s system, the “classification” must be identical across both platforms.
Using JSON or XML formats, FHIR resources define amlodipine not just by its classification code, but by its “Medication” resource type. This technical standard ensures that regardless of the software vendor—whether it’s Epic, Cerner, or a custom-built startup app—the classification remains consistent, preventing lethal medication errors that could arise from data misalignment.
Algorithmic Checks and Drug-Drug Interactions
One of the most significant tech applications of amlodipine’s classification is in Drug-Drug Interaction (DDI) engines. These are software modules integrated into prescribing tools.
Because amlodipine is classified as a calcium channel blocker that is metabolized by the CYP3A4 enzyme, the DDI engine uses a lookup table to check for conflicts. If a physician attempts to prescribe a drug classified as a “CYP3A4 inhibitor” (like certain antifungals), the software triggers an immediate alert. This is a purely technical process: a logic gate in the code compares the classification of Drug A with the classification of Drug B and returns a boolean “true” or “false” for potential toxicity.
The Future of Healthtech: Personalized Medicine and Genomic Classification
As we move toward the era of personalized medicine, the classification of amlodipine is becoming even more granular. We are transitioning from general chemical classification to individual-centric data classification.
Pharmacogenomics and Data Integration
The future of healthtech lies in pharmacogenomics—the study of how genes affect a person’s response to drugs. In this context, the technical classification of amlodipine might include “Genomic Compatibility” markers.
Software platforms are being developed to integrate a patient’s DNA sequence with their medication list. For a patient with a specific genetic polymorphism in their calcium channels, the software might re-classify amlodipine from “Preferred Treatment” to “Sub-optimal Treatment.” This level of data integration requires massive computational power and sophisticated data pipelines to ensure that genomic data and pharmacological classifications are synced in real-time.
Blockchain and Traceability in the Supply Chain
The classification of amlodipine is also a cornerstone of pharmaceutical supply chain technology. Using blockchain, every batch of amlodipine is given a unique digital identity.
In this decentralized ledger, the drug’s classification, origin, expiration date, and temperature logs during shipping are all recorded. This tech ensures that the classification of the drug is “Verified” and “Authentic.” If a batch is compromised, the blockchain allows for an instantaneous, automated recall by targeting only the specific digital signatures associated with that batch, showcasing the power of combining pharmacological classification with distributed ledger technology.

Conclusion: The Digital Identity of Amlodipine
In the final analysis, identifying the classification of amlodipine within the tech sector is about understanding the intersection of chemistry and code. It is no longer enough to say that amlodipine is a calcium channel blocker; in the digital age, it is a C08CA01 ATC code, a FHIR Medication Resource, and a data point in a machine learning model.
As software continues to eat the world of medicine, the technical classification of drugs becomes the foundation upon which safe, efficient, and innovative healthcare is built. For developers, data scientists, and healthtech entrepreneurs, amlodipine represents a perfect case study in how rigorous data standards and cutting-edge technology can transform a simple chemical compound into a sophisticated tool for global health management. By mastering these digital classifications, we pave the way for a more integrated, intelligent, and secure medical future.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.