Genomics, a field that has rapidly ascended from theoretical biological concept to a cornerstone of modern technological innovation, represents a profound shift in our understanding of life itself. More than just studying individual genes, genomics is the interdisciplinary science of exploring the entire genome of an organism – its complete set of DNA, including all of its genes, their interconnections, and their collective influence. It stands distinct from classical genetics, which primarily focuses on single genes and their inheritance patterns, by taking a holistic, systems-level approach. At its heart, genomics is deeply intertwined with cutting-edge technology, leveraging advanced sequencing platforms, sophisticated bioinformatics tools, and artificial intelligence to unravel the complex blueprint that defines every living thing.
This technological revolution began with the deciphering of the human genome and has since expanded exponentially, impacting medicine, agriculture, environmental science, and beyond. It’s a field driven by data, computation, and relentless innovation, pushing the boundaries of what we can discover and achieve through an understanding of life’s fundamental code. As we delve deeper into the intricate world of genomics, it becomes clear that its essence is fundamentally technological, offering unprecedented insights and empowering transformative applications across various sectors.
The Technological Core of Genomics: Sequencing and Bioinformatics
The ability to “read” and interpret the vast stretches of DNA that constitute a genome is the foundational technological achievement underpinning genomics. This process relies on two primary pillars: high-throughput sequencing technologies and the sophisticated computational tools of bioinformatics.
Next-Generation Sequencing (NGS) Revolution
The advent of Next-Generation Sequencing (NGS), also known as massively parallel sequencing, truly democratized genomics, transforming it from a prohibitively expensive and time-consuming endeavor into a routine, high-throughput process. Prior to NGS, the gold standard was Sanger sequencing, a laborious method capable of reading only short DNA fragments at a time. NGS technologies, however, enabled the simultaneous sequencing of millions of DNA fragments, drastically increasing speed, reducing costs, and improving scalability.
Modern NGS platforms, such as those developed by Illumina, Pacific Biosciences (PacBio), and Oxford Nanopore Technologies, utilize diverse biochemical and engineering principles. Illumina’s sequencers, for instance, employ a “sequencing by synthesis” approach, detecting fluorescently tagged nucleotides as they are incorporated into a growing DNA strand. PacBio’s Single Molecule, Real-Time (SMRT) sequencing offers longer read lengths, crucial for resolving repetitive regions and complex genomic architectures, by monitoring DNA polymerase activity in real-time. Oxford Nanopore takes a unique approach, passing DNA molecules through a nanopore protein, with electrical current fluctuations indicating the sequence of individual bases. Each technology presents its own trade-offs in terms of read length, accuracy, throughput, and cost, allowing researchers to select the most appropriate tool for their specific genomic investigation. The continuous innovation in these sequencing platforms – pushing for higher accuracy, longer reads, faster turnaround, and lower costs – remains a critical driver of genomic progress.
Bioinformatics: The Data Science of Life
The sheer volume of data generated by NGS technologies would be meaningless without the computational power and analytical sophistication of bioinformatics. This discipline is the bridge between raw sequencing data and biological insight, integrating computer science, statistics, mathematics, and biology to store, retrieve, analyze, and interpret complex biological information.
Bioinformatics tools and software pipelines are essential at every stage of genomic data processing. This begins with quality control checks on raw reads, followed by crucial steps like sequence alignment (mapping millions of short reads back to a reference genome), variant calling (identifying differences like single nucleotide polymorphisms or insertions/deletions), and genome assembly (piecing together reads to reconstruct a novel genome from scratch). Specialized algorithms and statistical models are employed for gene annotation, identifying coding regions and regulatory elements, and for comparative genomics, which examines evolutionary relationships between species.
The demands of genomic data processing necessitate robust computational infrastructure, often leveraging cloud computing platforms for scalability and parallel processing. Machine learning and artificial intelligence are increasingly integrated into bioinformatics workflows to identify subtle patterns, predict gene function, or uncover disease associations that might elude traditional statistical methods. Specialized databases, such as NCBI’s GenBank or ENSEMBL, serve as repositories for genomic sequences and annotations, providing crucial resources for researchers worldwide. Without the continuous advancement of bioinformatics, the deluge of genomic data would remain an unmanageable torrent, rather than a wellspring of profound biological understanding.
Genomics in Action: Transformative Tech Applications
The technological prowess of genomics translates into tangible, life-changing applications across diverse sectors, demonstrating its capacity to innovate and solve complex problems.
Precision Medicine and Personalized Healthcare
Perhaps the most heralded application of genomics is in the realm of medicine, giving rise to “precision medicine” or “personalized healthcare.” By analyzing an individual’s unique genetic makeup, genomics allows for a highly tailored approach to disease prevention, diagnosis, and treatment. Genetic predispositions to certain diseases (e.g., specific cancers, cardiovascular conditions, neurological disorders) can be identified early, enabling proactive monitoring and preventative strategies.
A particularly impactful area is pharmacogenomics, which uses genomic information to predict an individual’s response to specific drugs. Genetic variations can influence how a person metabolizes a drug, determining its efficacy and the likelihood of adverse reactions. This allows clinicians to select the optimal drug and dosage, minimizing trial-and-error and improving patient outcomes, especially in fields like oncology, psychiatry, and cardiology. Genomic sequencing is also becoming instrumental in diagnosing rare diseases, often providing answers to families who have endured long diagnostic odysseys, and in guiding targeted therapies for various cancers by identifying specific mutations driving tumor growth.
Biotechnology and Agricultural Innovation
Beyond human health, genomics is revolutionizing biotechnology and agriculture. The ability to precisely read and manipulate genetic code has profound implications for improving crop yields, enhancing disease resistance, and boosting the nutritional value of food. Genomic selection, for example, uses high-density genetic markers to predict desirable traits in plants and animals more rapidly and accurately than traditional breeding methods, accelerating the development of superior varieties.
Furthermore, genomic insights are fundamental to advanced gene editing technologies like CRISPR-Cas9. By understanding the precise genomic locations and functions of genes, scientists can accurately target and modify DNA sequences to introduce beneficial traits (e.g., drought resistance in crops, disease immunity in livestock) or correct deleterious mutations. This technological capability promises more sustainable and efficient agricultural practices, addressing global food security challenges.

Environmental Genomics and Bioremediation
Genomics also extends its reach to the environment, particularly through the field of metagenomics. This involves sequencing genetic material directly from environmental samples (e.g., soil, water, air) to study the collective genomes of microbial communities without the need for culturing individual organisms. This provides unprecedented insights into microbial diversity, ecological roles, and interactions within complex ecosystems.
Environmental genomics has diverse applications: monitoring ecosystem health, identifying novel enzymes or metabolic pathways for industrial applications, and tracking the spread of antibiotic resistance genes in the environment. In bioremediation, genomic data helps identify and optimize microbial communities capable of degrading pollutants, offering natural solutions for cleaning up contaminated sites. Understanding the genomic makeup of environmental organisms is crucial for developing sustainable biotechnological processes and safeguarding our planet’s biodiversity.
Emerging Technologies and Future Frontiers in Genomics
The pace of innovation in genomics shows no signs of slowing, with several emerging technologies poised to further expand its capabilities and applications.
Single-Cell Genomics
Traditionally, genomic sequencing provided an average genetic profile from a bulk sample of millions of cells. Single-cell genomics, however, offers a revolutionary leap by enabling the analysis of DNA or RNA from individual cells. This technology is critical for understanding cellular heterogeneity within tissues, unraveling developmental processes, and investigating disease mechanisms at an unprecedented resolution. For instance, in cancer research, single-cell sequencing can identify rare tumor cells with unique mutations that might drive metastasis or drug resistance, insights that are often masked in bulk analyses. This capability allows for a much more nuanced understanding of complex biological systems and disease progression.
AI and Machine Learning in Genomic Discovery
The sheer volume and complexity of genomic data make it an ideal playground for artificial intelligence (AI) and machine learning (ML) algorithms. These advanced computational techniques are becoming indispensable for extracting meaningful patterns and predictions from genomic datasets. AI can accelerate drug discovery by predicting protein structures, identifying potential therapeutic targets, and screening vast chemical libraries for drug candidates. In disease diagnostics, ML models can be trained to identify subtle genomic biomarkers indicative of disease onset or progression, often outperforming human interpretation. Furthermore, AI is automating and optimizing bioinformatics pipelines, making data analysis faster, more accurate, and more accessible, freeing human researchers to focus on higher-level interpretation and experimental design.
Gene Editing Technologies Beyond CRISPR
While CRISPR-Cas9 revolutionized gene editing, the field continues to evolve with the development of even more precise and versatile tools. Technologies like “base editing” and “prime editing” offer enhanced precision by allowing for single-base changes or small insertions/deletions without inducing double-strand DNA breaks, which can be associated with off-target effects and chromosomal rearrangements. Base editors can convert one DNA base pair into another (e.g., C to T, or A to G) directly, while prime editors can perform a wider range of precise edits, including all 12 possible point mutations, small insertions, and deletions. These advancements promise greater therapeutic potential for correcting a broader spectrum of genetic diseases with improved safety and specificity, pushing the boundaries of what is possible in genomic interventions.
Challenges and Ethical Considerations in the Genomic Era
Despite its immense promise, the widespread adoption and responsible development of genomics are accompanied by significant technical, logistical, and ethical challenges.
Data Management and Security
Genomic data is not only massive in scale but also incredibly sensitive, containing deeply personal information about an individual’s health, ancestry, and predispositions. The management, storage, and sharing of this data pose substantial technological hurdles. Secure cloud infrastructure, robust encryption protocols, and sophisticated access controls are paramount to prevent data breaches and misuse. Compliance with stringent data privacy regulations like GDPR in Europe and HIPAA in the United States is complex and demanding. Developing standardized formats and interoperable platforms for genomic data exchange, while maintaining privacy, is an ongoing technical challenge that requires collaborative efforts from researchers, healthcare providers, and technology developers.
Interpretational Complexities and Clinical Integration
Translating raw genomic data into actionable clinical insights is far from straightforward. The human genome contains millions of variants, and distinguishing between benign changes and those that are pathogenic or medically significant requires sophisticated analytical tools and expert interpretation. There’s often a gap between identifying a genetic variant and understanding its functional consequence or clinical relevance. This necessitates highly skilled bioinformaticians, genetic counselors, and clinicians trained in genomic medicine. Integrating genomic testing and data into routine clinical workflows also presents logistical challenges, including educating healthcare providers, developing clear guidelines for genomic test ordering and interpretation, and ensuring that clinical information systems can effectively handle and display genomic data.
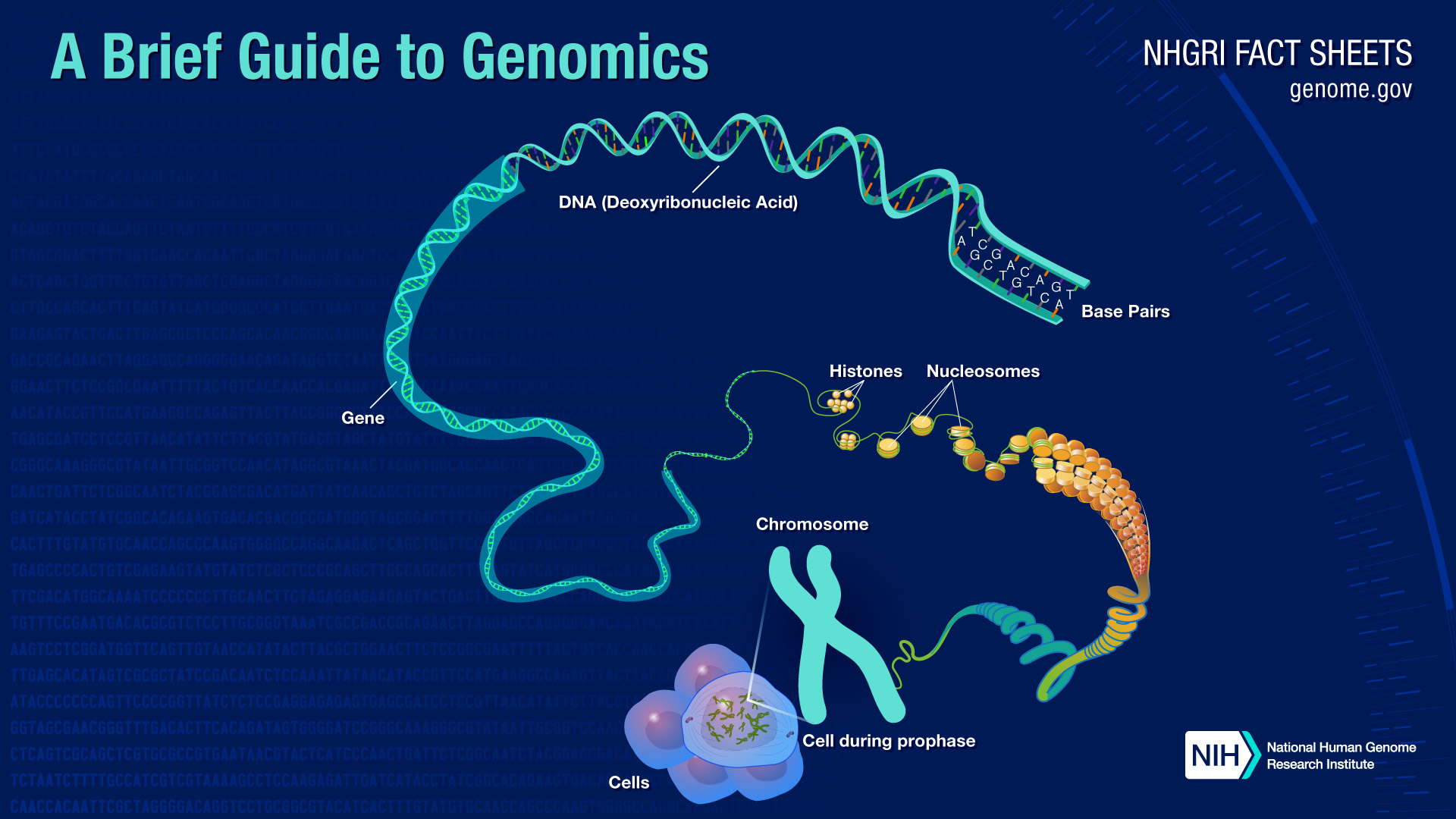
Ethical and Societal Implications
The power of genomics also brings forth profound ethical and societal questions. Ensuring equitable access to these advanced technologies is a critical concern, as disparities could exacerbate existing health inequalities. The potential for genetic discrimination in areas like employment or insurance coverage is a serious issue that requires robust regulatory frameworks and legal protections. Furthermore, the ability to predict genetic predispositions raises complex questions about individual autonomy, informed consent, and the psychological impact of knowing one’s future health risks. As gene-editing technologies advance, the ethical debates surrounding human germline editing (changes passed to future generations) and the potential for “designer babies” highlight the need for careful societal deliberation, transparent public discourse, and thoughtful policy development to ensure these powerful technologies are used responsibly and for the benefit of all humanity.
In conclusion, genomics is a transformative field that stands firmly at the intersection of biology and cutting-edge technology. From the intricate machinery of next-generation sequencers to the sophisticated algorithms of bioinformatics and AI, technology is the engine driving our ability to read, interpret, and ultimately interact with the fundamental code of life. Its applications are already reshaping healthcare, agriculture, and environmental science, promising a future of personalized medicine, sustainable food production, and a deeper understanding of our planet. However, as with any powerful technology, genomics demands careful stewardship, requiring continuous innovation to address its technical challenges, robust frameworks to secure its sensitive data, and ongoing ethical deliberation to guide its responsible and equitable development for the benefit of society as a whole.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.