In the landscape of modern biotechnology, few innovations represent the marriage of biological intuition and high-tech engineering as perfectly as Antibody-Drug Conjugates (ADCs). Often described by researchers as “biological missiles” or “the magic bullets of oncology,” ADCs are a class of biopharmaceutical drugs designed as a targeted therapy for the treatment of cancer. Unlike traditional chemotherapy, which acts like a carpet bomb—affecting both healthy and cancerous cells—ADCs are engineered to deliver potent cytotoxic agents directly to specific cells. This article explores the intricate technology, architectural design, and computational advancements that make ADCs one of the most promising frontiers in the tech-driven medical era.

The Architecture of ADCs: A Three-Component Bio-Tech System
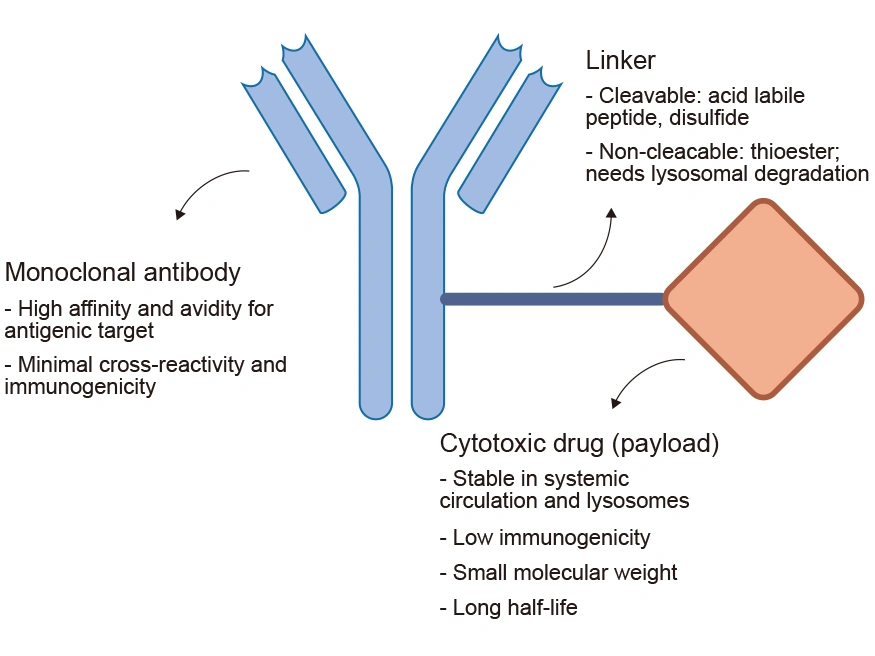
To understand an ADC, one must view it not as a single chemical entity, but as a complex piece of molecular machinery. The efficacy of an ADC depends on the harmonious integration of three distinct technological components: the monoclonal antibody (mAb), the cytotoxic payload, and the chemical linker.
The Monoclonal Antibody: The Biological GPS
The first component is the monoclonal antibody, which serves as the “targeting system” of the device. In the tech world, we might compare the mAb to a sophisticated GPS or a facial recognition algorithm. These proteins are engineered to bind with high affinity to specific antigens—proteins found predominantly on the surface of tumor cells. By selecting an antibody that ignores healthy tissue and locks onto malignant markers (such as HER2 or Trop-2), engineers ensure that the “payload” is only delivered where it is needed.
The Cytotoxic Payload: The Molecular Warhead
The payload is the active drug component designed to destroy the target cell. Interestingly, the payloads used in ADCs are often too toxic to be administered through traditional systemic injection; they would be lethal to the patient before reaching the tumor. However, when conjugated to an antibody, these highly potent small molecules (such as auristatins or maytansinoids) remain inert while in circulation. They only become active once the “delivery vehicle” has successfully docked and entered the target cell.
The Linker: The Critical Interface of Stability and Release
Perhaps the most sophisticated piece of engineering within the ADC is the linker. This is the chemical bridge that attaches the payload to the antibody. The linker must solve a difficult binary problem: it must be stable enough to hold the payload securely while the drug travels through the bloodstream (to prevent off-target toxicity), yet it must be “programmable” to break apart and release the drug instantly once it reaches the acidic or enzyme-rich environment inside a cancer cell.
The Biological Logic Gate: How ADCs Function as Targeted Hardware
The operation of an ADC follows a logical sequence that mirrors a complex software routine or an automated delivery system. This “mechanism of action” is what separates these biotechs from first-generation cancer treatments.
Antigen Recognition and Binding
The process begins when the ADC is infused into the patient’s bloodstream. The antibody component “scans” cell surfaces until it finds its matching antigen. This is a lock-and-key mechanism. Once the ADC binds to the surface of the tumor cell, it triggers a biological response known as receptor-mediated endocytosis. Essentially, the cell “swallows” the ADC, bringing it inside the cellular membrane.
Internalization and Endosomal Processing
Once inside the cell, the ADC is transported to specialized compartments called lysosomes. This is where the “environmental trigger” of the linker is activated. Depending on whether the linker is designed to be “cleavable” or “non-cleavable,” the high acidity or specific enzymes within the lysosome begin to dismantle the ADC structure. The linker snaps, and the cytotoxic payload is released from its antibody carrier.
The Bystander Effect: Expanding the Radius of Impact
In recent years, biotech engineers have optimized ADCs to exhibit what is known as the “bystander effect.” If the payload is lipid-soluble, once it has killed the primary target cell, it can diffuse out of that cell and enter neighboring cancer cells that might not have expressed the target antigen. This is a crucial “fail-safe” or “patch” in the technology, allowing the drug to clear out heterogeneous tumor clusters that would otherwise be resistant to the initial targeting.
Advanced Manufacturing and Computational Design in ADC Development
The creation of an ADC is an exercise in extreme precision. Historically, attaching drugs to antibodies resulted in “heterogeneous” mixtures—some antibodies might have one drug molecule attached, others might have eight. This lack of uniformity made it difficult to predict how the drug would behave.
Site-Specific Conjugation Technologies
Modern biotech has moved toward “site-specific conjugation.” Using enzymatic or genetic engineering, researchers can now dictate exactly where on the antibody the payload will be attached. This allows for a precise Drug-to-Antibody Ratio (DAR), typically optimized at 4 or 8. This level of control is akin to moving from hand-soldering circuits to automated robotic assembly lines; it ensures every “unit” of the drug performs identically to the next.
AI and Machine Learning in Linker Optimization
The tech industry’s greatest contribution to ADCs in the last five years has been the application of Artificial Intelligence (AI). Designing a linker that is stable in the blood but labile in the cell is a massive data problem. Machine learning algorithms now simulate millions of chemical permutations to predict which linker structures will yield the highest stability. This computational approach has shortened the “design-test-build” cycle of ADC development by years, allowing for more rapid iteration of these complex molecules.
Challenges in Bio-Engineering: Overcoming Resistance and Toxicity
Despite their technological brilliance, ADCs are not without engineering challenges. The human body is a dynamic environment, and tumors are adept at “hacking” the treatments designed to kill them.
Managing the Therapeutic Window
The primary challenge for ADC engineers is the “therapeutic window”—the gap between a dose that is effective and a dose that is toxic. If the linker is too unstable, the “warhead” drops off early, causing systemic side effects. If the antibody isn’t specific enough, it might dock at a healthy organ. Current biotech research is focused on refining the “targeting sensors” to reduce these errors, much like refining the sensors on an autonomous vehicle to prevent false positives.
Next-Gen Formats: Bispecifics and Pro-ADCs
To overcome resistance, tech-heavy firms are developing “Bispecific ADCs.” These are engineered with antibodies that can bind to two different antigens at once, ensuring a more secure “handshake” with the tumor cell. Additionally, “Pro-ADCs” are being developed; these remain masked by a protective coating that is only removed by specific enzymes found exclusively within the tumor microenvironment, adding an extra layer of “encryption” to the drug delivery process.

The Future of Biotech: ADCs as the Foundation for Personalized Oncology
As we look toward the future of medical technology, the role of Antibody-Drug Conjugates will only expand. We are moving away from the era of “one size fits all” medicine and into an era of “modular medicine.”
The modularity of the ADC platform is its greatest strength. If a new type of cancer antigen is discovered, engineers do not need to invent an entirely new drug class; they simply swap out the “targeting” antibody component for a new one, keeping the proven linker and payload architecture. This “plug-and-play” capability is revolutionizing how we approach rare diseases and resistant cancers.
Furthermore, the integration of real-time diagnostics—where a patient’s tumor is sequenced and a custom ADC is selected from a library of components—represents the ultimate goal of personalized bio-tech. By treating the human body as a biological system that can be precisely addressed via molecular engineering, ADCs are proving that the most advanced technology of the 21st century isn’t just in our pockets or our “cloud”—it’s being programmed to heal us from the inside out. Through the convergence of chemistry, biology, and computational power, ADCs are redefining the limits of what is possible in the fight against disease.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.