The intersection of clinical microbiology and information technology has fundamentally altered how healthcare providers approach common infections. Group B Streptococcus (GBS), or Streptococcus agalactiae, is a bacterium frequently identified in urinary tract screenings, particularly in pregnant populations and the elderly. While the medical community has long relied on standardized protocols to determine what antibiotics treat Group B Strep in urine, the modern landscape is being redefined by artificial intelligence, cloud-based diagnostic tools, and precision medicine software.
In the current HealthTech era, the focus is shifting away from broad-spectrum guesswork and toward data-driven, targeted interventions. By leveraging advanced algorithmic analysis and rapid diagnostic hardware, the tech industry is providing clinicians with the tools necessary to identify GBS faster and select the most effective antibiotic regimen with unprecedented accuracy.
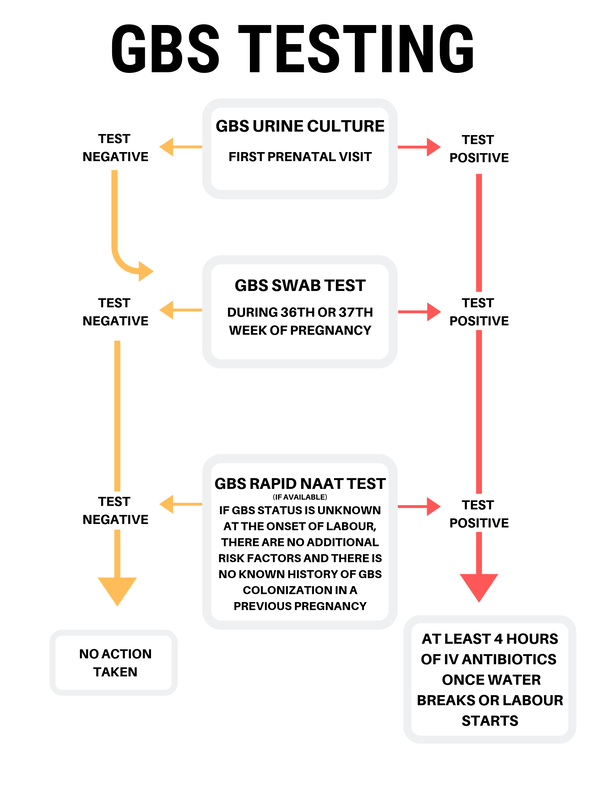
The Digital Transformation of Bacterial Identification and Urinalysis
The traditional method of identifying Group B Strep in urine involved manual culture growth, which often took 24 to 48 hours. Today, technology has streamlined this process, reducing the time-to-treatment through high-tech diagnostic platforms.
AI-Driven Automated Microscopy
One of the most significant leaps in HealthTech is the integration of Artificial Intelligence (AI) and Machine Learning (ML) into automated urinalysis systems. Modern digital scanners use neural networks to analyze high-resolution images of urine sediment. These systems are trained on millions of data points to recognize the specific morphological characteristics of GBS. By automating the visual identification process, labs can flag potential infections in minutes rather than days, allowing for a more rapid transition to targeted antibiotic therapy.
MALDI-TOF MS and Proteomic Identification
Matrix-Assisted Laser Desorption/Ionization-Time of Flight (MALDI-TOF) Mass Spectrometry represents a pinnacle of diagnostic technology. This hardware identifies bacteria by analyzing their protein “fingerprints.” When a sample of urine indicates a high bacterial load, MALDI-TOF tech can identify GBS with near-perfect accuracy by comparing the sample’s protein profile against a massive digital library. For the tech-savvy clinician, this means the question of “what is the pathogen?” is answered with a level of digital precision that traditional culture-based methods simply cannot match.
Antibiotic Stewardship Through Clinical Decision Support Systems (CDSS)
Once GBS is identified, the technological challenge shifts to selecting the appropriate treatment. This is where Clinical Decision Support Systems (CDSS) become vital. These software platforms are integrated into Electronic Health Records (EHR) to guide doctors on what antibiotics treat Group B Strep in urine based on real-time data.
Algorithmic Prescription Precision
Selecting an antibiotic—typically Penicillin, Ampicillin, or Cephalosporins for GBS—is no longer just a matter of memory. CDSS platforms analyze patient-specific data, including allergy profiles, kidney function, and potential drug interactions, to recommend the optimal dosage. If a patient is allergic to first-line beta-lactams, the software uses complex logic trees to suggest alternatives like Clindamycin or Vancomycin, while simultaneously checking for the most recent local resistance trends recorded in the cloud.
Digital Antibiograms and Resistance Tracking
One of the most powerful tools in the tech arsenal is the digital antibiogram. Unlike paper-based reports of the past, modern antibiograms are dynamic dashboards that track antibiotic resistance patterns within a specific hospital or geographic region. By using big data analytics, these tools help healthcare systems understand if GBS in their specific area is becoming resistant to common treatments. This ensures that the antibiotics prescribed are not just theoretically effective, but proven to work against the local strains currently in circulation.

Telemedicine and the Rise of Remote Diagnostic Tech
The “hospital at home” movement has necessitated the development of gadgets and apps that can manage GBS infections without a physical clinic visit. This shift is particularly important for managing asymptomatic bacteriuria in high-risk groups, where frequent monitoring is required.
IoT-Connected At-Home Testing Kits
The consumerization of healthcare tech has led to the development of smart urine testing kits. These gadgets often include a dipstick that interfaces with a smartphone app. Using the phone’s camera and a proprietary colorimetric algorithm, the app can detect signs of infection, such as nitrites or leukocyte esterase, and upload the data to a secure cloud server for physician review. While these kits may not always definitively identify GBS specifically, they act as an early-warning system that triggers a formal laboratory evaluation, streamlining the path to antibiotic treatment.
Virtual Care Integration
Telehealth platforms have evolved beyond simple video calls. They now incorporate integrated diagnostic workflows where data from home-testing kits or local labs is automatically synced into a virtual consultation dashboard. This allows a remote provider to view the GBS lab results, confirm the bacterial count, and electronically transmit a prescription for the appropriate antibiotic to the patient’s preferred pharmacy. This tech-driven workflow reduces the “friction” of traditional healthcare, ensuring that GBS in urine is treated before it can progress to more serious complications like pyelonephritis or sepsis.
The Future of Precision Medicine: Bioinformatics and CRISPR
As we look toward the future of technology in treating GBS, we are moving beyond traditional chemicals and toward biological programming. The fields of bioinformatics and synthetic biology are opening new doors for treating stubborn or recurrent infections.
Genomic Sequencing and Targeted Therapy
Next-Generation Sequencing (NGS) technology allows researchers to map the entire genome of a GBS strain found in a patient’s urine. This high-tech approach identifies specific virulence factors and resistance genes. In the future, rather than using broad-spectrum antibiotics that kill beneficial gut bacteria, bioinformatics software could help design “precision antibiotics” or small-molecule inhibitors that only target the specific GBS strain identified in the patient.
CRISPR and Programmable Antimicrobials
Perhaps the most exciting tech trend in microbiology is the application of CRISPR-Cas9 as an antimicrobial tool. Researchers are developing “programmable” treatments that use CRISPR technology to target and destroy the DNA of Group B Strep specifically. This would be delivered via engineered bacteriophages (viruses that eat bacteria). From a technology perspective, this represents a shift from “analog” chemical medicine to “digital” genetic medicine, where the “antibiotic” is essentially a piece of code that tells the treatment exactly which bacterial cells to eliminate.

Conclusion: A Tech-Enabled Standard of Care
The question of what antibiotics treat Group B Strep in urine is increasingly answered not just by medical textbooks, but by a sophisticated stack of healthcare technologies. From the AI that identifies the bacteria in a lab to the CDSS that ensures the correct prescription, and the telemedicine platforms that deliver care to the patient’s doorstep, technology is the backbone of modern infection management.
As we move forward, the integration of big data, IoT, and genetic engineering will continue to refine our approach. For patients and providers alike, this means more accurate diagnoses, fewer side effects from unnecessary medications, and a more efficient healthcare system. The fight against Group B Strep is no longer a manual struggle; it is a high-tech endeavor defined by data, speed, and precision.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.