For over a century, the lead-acid battery has remained the heartbeat of the internal combustion engine. While the automotive industry is currently undergoing a massive digital transformation, the fundamental technology behind the traditional 12-volt battery has remained remarkably consistent. At the core of this technology is a potent chemical solution: sulfuric acid. Understanding the role of this acid is not just a matter of basic chemistry; it is an exploration of how chemical energy is converted into the electrical power required to start engines and manage sophisticated onboard electronics.

The Engineering Foundation: Sulfuric Acid as the Primary Electrolyte
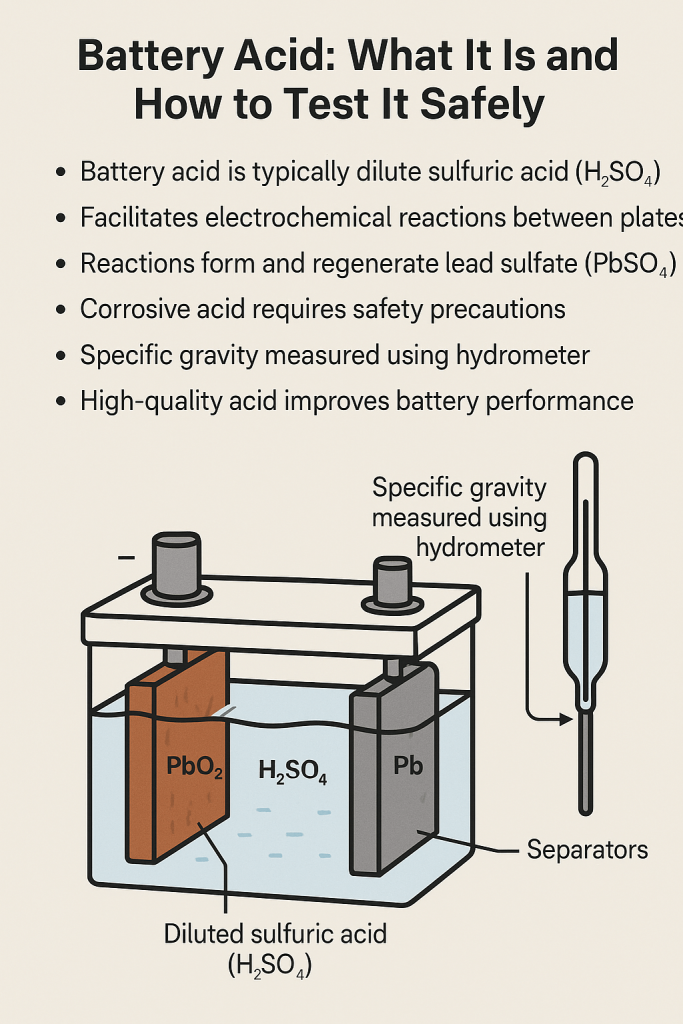
The “acid” in a car battery is almost exclusively sulfuric acid ($H2SO4$) diluted with purified water. This mixture is known as the electrolyte. In a standard lead-acid battery, the electrolyte typically consists of about 35% sulfuric acid and 65% water. This specific ratio is engineered to maximize the battery’s capacity to conduct electricity and facilitate the chemical reactions necessary for energy storage.
The Role of Electrolytes in Electron Transfer
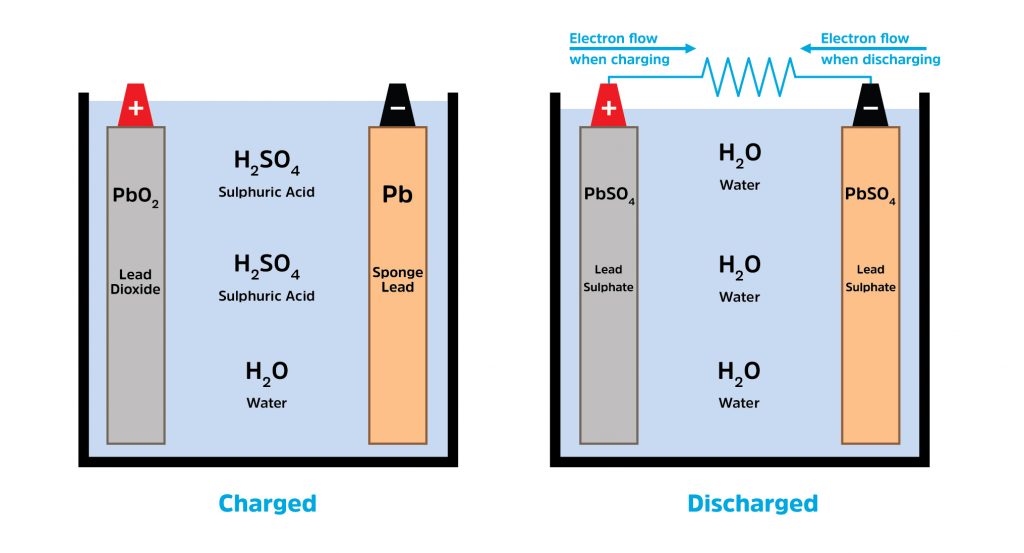
In the context of electrical engineering, an electrolyte is a substance that produces an electrically conducting solution when dissolved in a polar solvent, such as water. In a car battery, the sulfuric acid serves as the medium through which ions move between the lead plates. When you turn the ignition, a chemical reaction begins. The sulfuric acid reacts with the lead plates ($Pb$ and $PbO2$) to create lead sulfate ($PbSO4$) and water. This reaction releases electrons, which flow through the vehicle’s electrical system to power the starter motor, lights, and computer modules.
Specific Gravity and State of Charge
From a diagnostic technology standpoint, the concentration of the acid is a critical indicator of the battery’s health. Technicians use “specific gravity” to measure the density of the electrolyte compared to water. As a battery discharges, the sulfate from the acid adheres to the plates, leaving the remaining liquid more water-heavy and less dense. High-tech digital hydrometers can now analyze this density to provide precise data on the state of charge, allowing for predictive maintenance before a total power failure occurs.
The Thermal Dynamics of Battery Acid
The chemical composition of battery acid is also designed to withstand extreme temperature fluctuations. However, the technology has its limits. In extreme cold, the chemical reaction slows down, which is why batteries often struggle in winter. Conversely, excessive heat can cause the water within the acid mixture to evaporate, increasing the concentration of the acid and accelerating the degradation of the internal lead plates—a process known as “corrosion.”
Technological Evolutions: From Flooded Cells to AGM
While the basic chemistry of sulfuric acid remains the standard, the way this acid is housed and utilized has seen significant technological advancements. The industry has moved from “flooded” batteries, which required periodic manual maintenance, to sophisticated, sealed systems designed for the high-demand electronics of modern vehicles.
Traditional Flooded Lead-Acid (FLA) Technology
The oldest form of battery technology, FLA batteries, contains liquid electrolyte that moves freely within the battery casing. These are the most cost-effective solution but require the battery to be kept upright to prevent acid leaks. In earlier decades, drivers had to manually refill the battery with distilled water to replace what was lost during electrolysis. Modern “maintenance-free” FLA batteries have improved casing designs that recapture vapor, though the core “wet” technology remains the same.
Absorbent Glass Mat (AGM) Innovation
The pinnacle of lead-acid technology is the Absorbent Glass Mat (AGM) battery. In these units, the sulfuric acid is not a free-flowing liquid. Instead, it is absorbed into a fine fiberglass mat sandwiched between the lead plates. This design allows for a “starved” electrolyte system, meaning there is just enough acid to keep the mats wet but no liquid to spill. AGM tech offers superior vibration resistance and can be mounted in various orientations, making it the preferred choice for performance gadgets and high-end tech-heavy vehicles.
Gel Cell Batteries and Specialized Applications
Another variation is the Gel battery, where silica is added to the sulfuric acid to turn it into a thick, jelly-like substance. While less common in automotive starting applications due to their sensitivity to high-voltage charging, Gel batteries are frequently used in deep-cycle applications, such as for marine electronics or off-grid solar storage. This technology highlights the versatility of sulfuric acid when modified for specific discharge and recharge cycles.
Integrating Battery Tech with Modern Vehicle Systems
The battery is no longer a standalone component; it is an integrated part of a vehicle’s digital architecture. As cars have become “computers on wheels,” the demand for stable, reliable current from the lead-acid system has grown exponentially.
Smart Charging and Battery Management Systems (BMS)
Modern vehicles utilize Battery Management Systems (BMS) to monitor the health of the acid-based battery in real-time. These systems use sensors to track voltage, current, and temperature. By understanding the “Coulombic efficiency” of the sulfuric acid reaction, the BMS can adjust the alternator’s output. This prevents overcharging, which would otherwise “boil” the acid and damage the internal components, thereby extending the lifespan of the hardware through software optimization.
The Stop-Start Technology Challenge
The rise of “Stop-Start” technology—where the engine shuts off at red lights to save fuel—has placed a massive strain on traditional battery chemistry. Every time the engine restarts, the battery must deliver a high-current burst. This requires the sulfuric acid to react and recover rapidly. This technological demand led to the development of Enhanced Flooded Batteries (EFB), which utilize special carbon additives on the plates to allow the acid to react more efficiently during frequent cycling.
Digital Diagnostics and Telematics
In the era of the Internet of Things (IoT), car battery health is now monitored remotely. Telematics systems can send alerts to a user’s smartphone if the voltage of the lead-acid battery drops below a certain threshold. This digital layer sits on top of the chemical layer, ensuring that the 19th-century technology of lead and acid can keep up with 21st-century connectivity demands.
The Future of Energy Storage: Beyond Acid
As we move toward a future dominated by Electric Vehicles (EVs), the role of sulfuric acid is shifting. While the primary propulsion in an EV comes from Lithium-ion or Nickel-cobalt-manganese chemistries, the traditional lead-acid battery still plays a vital role in the tech ecosystem.
The 12-Volt Legacy in EVs
Even the most advanced Teslas and Lucids currently utilize a 12-volt lead-acid (or more recently, a small 12V Li-ion) battery to power secondary systems. This battery is responsible for the digital screens, interior lights, and—most importantly—the safety systems that engage the high-voltage drivetrain. The reliability of the sulfuric acid reaction makes it a “fail-safe” for critical systems that cannot afford the volatility sometimes associated with high-voltage lithium packs.
Recycling Tech and the Circular Economy
One of the most impressive “tech” stories regarding battery acid is the recycling infrastructure. Lead-acid batteries are the most recycled consumer product in the world, with a 99% recycle rate in many regions. Specialized facilities use automated technology to neutralize the sulfuric acid or convert it into sodium sulfate, which is then used in the manufacturing of laundry detergents and glass. The lead plates are melted down and reused, creating a closed-loop system that is far more mature than current lithium-ion recycling technologies.
Transitioning to Solid-State and Lithium Alternatives
Despite its reliability, the industry is looking at alternatives to acid-based systems. Solid-state batteries, which replace the liquid electrolyte with a solid ceramic or polymer, promise higher energy density and no risk of acid leaks. However, the cost-to-performance ratio of sulfuric acid remains unbeatable for standard automotive applications for the time being.
Conclusion
The question of “what acids are in car batteries” leads us into a complex world of chemical engineering and technological integration. Sulfuric acid remains the industry standard because of its efficiency, predictability, and ability to be managed by modern electronic systems. From the rugged simplicity of flooded cells to the high-performance engineering of AGM systems, the interaction between sulfuric acid and lead is a cornerstone of automotive technology. As we bridge the gap between internal combustion and fully electric futures, the chemical foundation provided by these acids continues to be the silent partner in our global mobility.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.