In the realm of biotechnology, enzymes are the “software” of the living world. They are highly specialized biological catalysts that accelerate chemical reactions, making life as we know it possible. However, from an industrial and technological perspective, enzymes have a critical “operating temperature.” When that temperature is exceeded, these biological machines fail. The question of “what happens to enzymes at high temperatures” is not just a biological curiosity; it is a fundamental challenge for the multi-billion-dollar bio-manufacturing and tech-driven pharmaceutical sectors.
To understand the tech behind thermal resilience, we must first look at the mechanics of failure and then explore how modern computational tools and synthetic biology are rewriting the code of these proteins to withstand extreme heat.
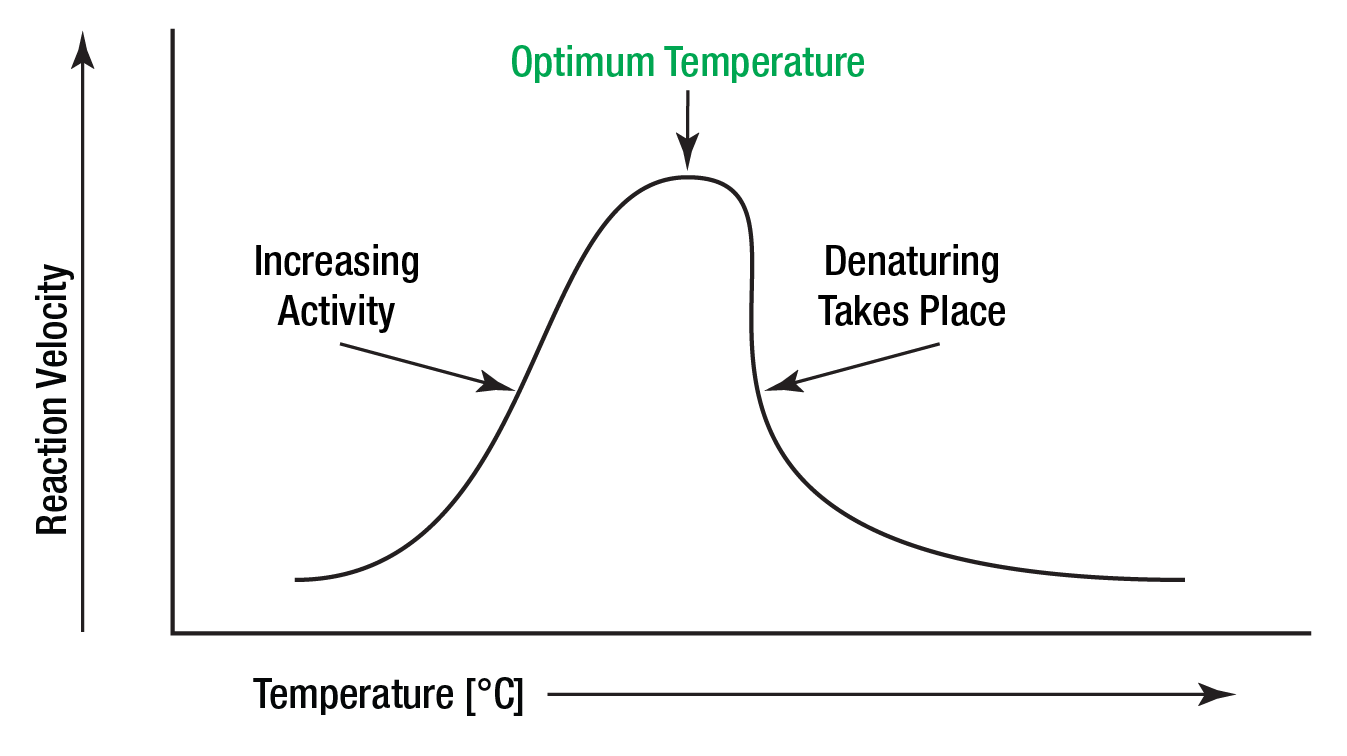
The Mechanics of Thermal Denaturation: When Biological Tech Fails
At the molecular level, an enzyme is a complex, three-dimensional structure folded into a very specific shape. Its function is entirely dependent on this geometry. When we ask what happens to enzymes at high temperatures, the answer is “denaturation.” In tech terms, this is the equivalent of a processor overheating until its internal components physically warp, rendering it incapable of executing instructions.
The Breakdown of Non-Covalent Bonds
Enzymes are held together by relatively weak forces: hydrogen bonds, ionic interactions, and van der Waals forces. As temperature rises, kinetic energy increases. The atoms within the enzyme vibrate more violently. Eventually, this kinetic energy overcomes the strength of the non-covalent bonds holding the protein together. The enzyme begins to vibrate out of its functional shape—a process known as unfolding. Once the “active site” (the part of the enzyme that performs the work) is distorted, the enzyme can no longer bind to its substrate.
Irreversible Aggregation and System Crashing
In many cases, once an enzyme unfolds at high temperatures, it doesn’t just stop working; it becomes “garbage data.” The hydrophobic (water-fearing) interior of the protein becomes exposed to the surrounding aqueous environment. These exposed parts of different unfolded proteins tend to stick to one another, forming clumps or aggregates. This is why a boiled egg white turns from clear to opaque and solid; the proteins have denatured and tangled. In an industrial bioreactor, this aggregation represents a total system failure, leading to costly downtime and loss of raw materials.
AI and Computational Protein Design: Re-coding for Heat Resistance
If nature’s default enzymes fail at high temperatures, the modern technological solution is to redesign them. We are no longer limited to the enzymes found in common soil bacteria. Through advanced software and AI, engineers are now “overclocking” biology by designing thermophilic (heat-loving) enzymes from scratch.
AlphaFold and the Predictive Revolution
Until recently, predicting how an enzyme would fold—or unfold—was one of the hardest problems in computer science. The arrival of DeepMind’s AlphaFold and similar AI tools has changed the landscape. By using deep learning to predict the 3D structure of proteins with atomic accuracy, researchers can now simulate “what happens to enzymes at high temperatures” in a digital environment before ever stepping into a lab. These AI models allow tech-engineers to identify “weak points” in an enzyme’s structure that are likely to break first under thermal stress.
Directed Evolution and Machine Learning Loops
Another core technology in this space is Directed Evolution, a method that won the Nobel Prize in Chemistry. It involves creating millions of random mutations in an enzyme’s DNA and screening for the ones that survive high heat. Today, this process is supercharged by machine learning. Instead of manual screening, high-throughput robotic platforms generate massive datasets of enzyme variants. Machine learning algorithms then analyze these datasets to predict which specific amino acid substitutions will provide the highest thermal stability, creating a feedback loop that rapidly produces “super-enzymes” capable of operating at near-boiling temperatures.

Industrial Applications: High-Temp Enzymes in the Tech Ecosystem
The drive to solve the heat problem in enzymes is fueled by the massive efficiency gains available in industrial tech. When enzymes can survive high temperatures, chemical reactions happen faster, the risk of microbial contamination is reduced, and the viscosity of fluids is lowered, making industrial pumping and mixing more energy-efficient.
Biofuel Production and Waste-to-Energy Tech
The conversion of cellulose (plant waste) into biofuels requires breaking down tough, fibrous materials. This process is significantly more efficient at high temperatures. Tech companies in the green energy sector utilize engineered “cellulases”—enzymes that can withstand the heat required to liquefy biomass. By maintaining structural integrity at 65°C to 80°C, these enzymes allow for faster throughput in biofuel refineries, bridging the gap between laboratory science and commercial viability.
Plastic Degradation and the Circular Economy
One of the most exciting frontiers in “Enzyme Tech” is the development of heat-stable PETases—enzymes that eat plastic. Most plastic waste must be heated to a “glass transition temperature” to become soft enough for enzymes to attack the molecular bonds. Standard enzymes would denature instantly at these temperatures. Technological breakthroughs in protein engineering have led to the creation of synthetic enzymes that remain active at 70°C, allowing for the rapid enzymatic recycling of plastic bottles into their original chemical building blocks, a feat previously thought impossible.
The Future of Extremophilic Synthetic Biology
As we look toward the future of biotechnology, the focus is shifting from simply “fixing” existing enzymes to harvesting the “tech” used by extremophiles—microorganisms that naturally live in volcanic vents and hot springs.
Learning from Thermophilic Microorganisms
Nature has already solved the problem of high-temperature enzyme stability in organisms like Thermus aquaticus. These “extremophiles” have evolved proteins with increased numbers of disulfide bridges (chemical “staples”) and more compact hydrophobic cores. The current tech trend involves using CRISPR and gene-editing tools to transplant these thermal-resistance “modules” into industrial microbes. This is akin to taking the cooling system from a high-performance racing engine and installing it into a standard passenger car.
Digital Twins and Real-Time Bioprocess Monitoring
The final piece of the high-temperature enzyme puzzle isn’t just the enzyme itself, but the digital infrastructure used to manage it. “Digital Twins”—virtual replicas of physical bioreactors—now use real-time sensor data to monitor the health of enzymes during a reaction. If the temperature fluctuates, AI-driven control systems can adjust the pH or pressure in milliseconds to stabilize the enzyme’s molecular environment. This synergy between molecular biology and IoT (Internet of Things) technology ensures that the answer to “what happens to enzymes at high temperatures” is no longer “failure,” but rather “controlled optimization.”

Conclusion: The New Biological Frontier
Understanding what happens to enzymes at high temperatures has evolved from a basic scientific inquiry into a sophisticated branch of engineering. By viewing enzymes as programmable hardware and their DNA as code, the tech industry has begun to overcome the thermal limitations of biology.
Through the lens of AI-driven design, directed evolution, and extremophilic research, we are building a new generation of biological tools. These heat-resistant enzymes are not just biological curiosities; they are the engines of the next industrial revolution, powering everything from sustainable jet fuel to the circular plastic economy. As we continue to refine the technology of protein stabilization, the “boiling point” of biology will continue to move higher, opening up possibilities for innovation that were once hindered by the fragile nature of life itself.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.