In the modern landscape of chemical engineering and laboratory research, the transition from manual calculation to digital precision has redefined how we approach fundamental constants. At first glance, asking “what is the molar mass of Ca(OH)₂” might seem like a basic academic exercise. However, in the context of high-tech industrial applications—ranging from carbon capture technology to semiconductor manufacturing—this figure is a critical variable in complex algorithmic models. Calcium hydroxide, commonly known as slaked lime, serves as a cornerstone reagent in various technological sectors, and its precise molar weight is the baseline for the digital simulations that drive modern innovation.
The Algorithmic Basis of Molar Mass Calculation
To understand how technology processes chemical data, one must look at how software interprets the periodic table. For a computer program, calculating the molar mass of Calcium Hydroxide (Ca(OH)₂) involves more than simple addition; it requires the integration of high-precision isotopic averages stored within chemical databases.
The Breakdown of Molecular Components
In a digital environment, a chemical formula is treated as a string of data. To calculate the molar mass, the software parses the string:
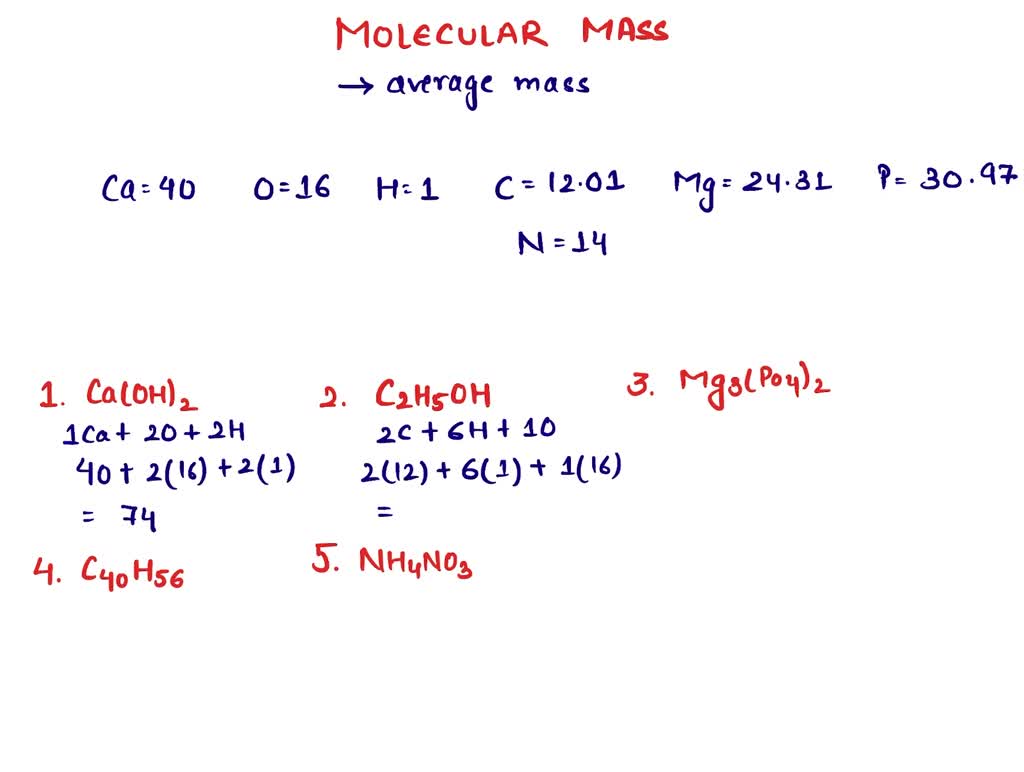
- Calcium (Ca): The primary metallic element, with a standard atomic weight of approximately 40.078 u.
- Oxygen (O): A non-metal with a weight of 15.999 u.
- Hydrogen (H): The lightest element, weighing 1.008 u.
The formula Ca(OH)₂ indicates one atom of Calcium and two functional groups of Hydroxide. The computational logic follows a nested calculation: $40.078 + 2 times (15.999 + 1.008)$. This results in a total molar mass of approximately 74.093 g/mol. While a student might round this to 74.1, high-end industrial software maintains the five-decimal precision necessary for large-scale automation.
Software Integration and Digital Libraries
Modern Laboratory Information Management Systems (LIMS) and stoichiometry calculators do not just perform these sums in isolation. They pull from live databases such as those maintained by IUPAC (International Union of Pure and Applied Chemistry). This ensures that if the scientific community refines an atomic weight due to better isotopic measurements, every calculation in a global pharmaceutical or tech firm is updated in real-time. This level of synchronization is vital for “Digital Twins”—virtual replicas of physical chemical processes—where even a 0.001% deviation in molar mass can lead to significant errors in predicted yields.
Software Ecosystems for Chemical Analysis and Simulation
The calculation of molar mass is the “Hello World” of chemical software. Today’s tech stack for chemists and industrial engineers includes a variety of tools that utilize the molar mass of Ca(OH)₂ to solve real-world problems.
Molecular Modeling and CAD in Chemistry
Tools like ChemDraw and BIOVIA Pipeline Pilot allow researchers to visualize the structure of Ca(OH)₂ and simulate its reactions. When designing a new water purification system, engineers use these tools to calculate exactly how many kilograms of calcium hydroxide are needed to neutralize a specific volume of acidic effluent. The software uses the molar mass to convert volumetric flow rates into precise chemical dosing instructions, which are then fed into automated SCADA (Supervisory Control and Data Acquisition) systems.
The Rise of AI and Machine Learning in Stoichiometry
Artificial Intelligence is currently revolutionizing how we interact with chemical data. Natural Language Processing (NLP) models are being trained on vast repositories of chemical literature to provide instant answers to complex queries. If a developer asks an AI assistant for the molar mass of Ca(OH)₂, the AI doesn’t just look up a static value. It analyzes the context—asking whether the user is working with anhydrous conditions or a specific concentration of lime milk—and provides the necessary stoichiometric adjustments.
Furthermore, machine learning algorithms are used in “Predictive Maintenance” for industrial equipment. By knowing the exact molar mass and chemical properties of Ca(OH)₂, software can predict how quickly calcium carbonate scale will build up in high-tech sensors, allowing for scheduled maintenance before a system failure occurs.
The Role of Ca(OH)₂ in Emerging Green Tech and Industrial Innovation

The specific properties of Calcium Hydroxide make it a vital component in several “Climate Tech” initiatives. Technology is not just about software; it is about the hardware and materials science that address global challenges.
Carbon Capture and Storage (CCS) Technologies
One of the most exciting tech trends is Direct Air Capture (DAC). Some of these systems utilize the chemical reaction between CO₂ and Ca(OH)₂. In these high-tech scrubbers, the molar mass is the fundamental unit of measurement for carbon sequestration efficiency. Tech companies in this space use IoT sensors to monitor the conversion of Calcium Hydroxide into Calcium Carbonate. By calculating the mass balance—based on the 74.09 g/mol figure—the system can report exactly how many tons of carbon have been removed from the atmosphere, providing verifiable data for carbon credit markets.
Semiconductor and Electronics Manufacturing
In the world of gadgets and hardware, Ca(OH)₂ is often used in the treatment of wastewater generated during the production of silicon wafers. The process of etching circuits requires highly acidic chemicals. Tech firms employ automated neutralizing stations that rely on the molar mass of Ca(OH)₂ to ensure that the pH of the wastewater is perfectly balanced before it is discharged. This precision prevents the corrosion of delicate infrastructure and meets the stringent environmental standards required for “Green Electronics” certification.
Wastewater Treatment and IoT Integration
Smart cities are increasingly integrating IoT (Internet of Things) sensors into their water management grids. These sensors measure the alkalinity and mineral content of water in real-time. When the system detects a drop in pH, it triggers a digital command to release a calculated amount of Ca(OH)₂. The cloud-based software calculates the required dosage using the molar mass as its core constant, ensuring that the water remains safe for the public while optimizing the cost of chemicals used.
Digital Security and Data Integrity in Chemical Research
As chemical data becomes more integrated into the cloud, the focus on digital security and data integrity has intensified. When we discuss the “molar mass of Ca(OH)₂” within a corporate database, we are discussing a piece of “Master Data.”
Protecting Proprietary Chemical Formulas
In the pharmaceutical and specialty chemical industries, the exact ratios used in a process—calculated using molar masses—are valuable trade secrets. Cyber-security tools are now specifically designed to protect “Recipe Data.” If a hacker gains access to the stoichiometric ratios of a proprietary process involving Ca(OH)₂, they can reverse-engineer the product. Therefore, digital security trends in this niche focus on encrypting calculation logs and using multi-factor authentication for LIMS access.
Blockchain and the Chemical Supply Chain
Blockchain technology is finding a home in the chemical industry to ensure the purity and origin of reagents. When a tech company buys Ca(OH)₂ for sensitive manufacturing, they need to know its exact purity. A blockchain ledger can store the batch’s chemical analysis, including its calculated molar mass and impurity profile. This creates an immutable digital “Passport” for the chemical, ensuring that the material arriving at the factory matches the digital specifications required by the automated systems.
The Future of Computational Chemistry: Quantum Computing
The ultimate frontier in calculating and applying molar mass lies in quantum computing. While current digital tools are excellent at basic stoichiometry, they struggle with the complex electron-cloud interactions that occur during chemical reactions at the molecular level.
Beyond Classical Calculations
Quantum computers, such as those being developed by IBM and Google, have the potential to simulate the behavior of Ca(OH)₂ in real-time at an atomic scale. This would allow researchers to see how every gram (based on its molar mass) interacts with pollutants or new materials in a way that traditional binary computers cannot.
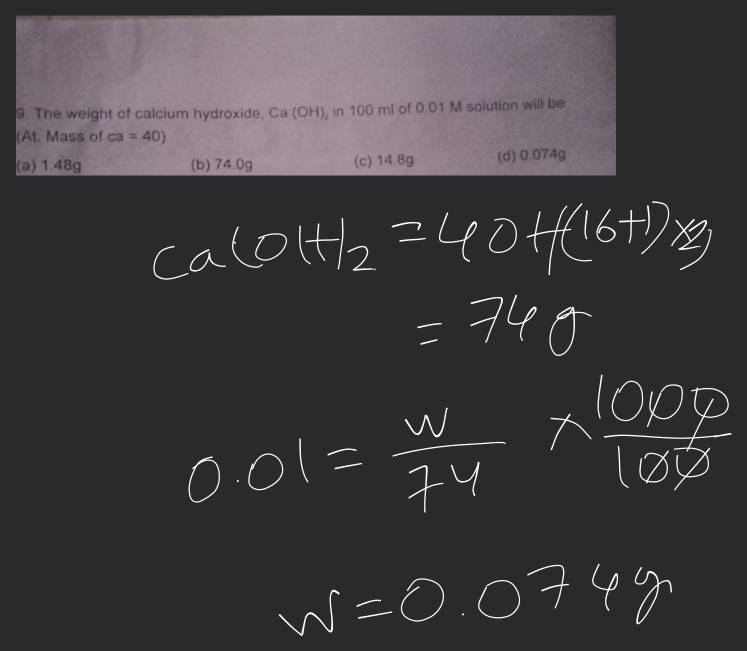
Accelerating Discovery
In the tech world, speed is everything. Quantum-enhanced simulations could reduce the time it takes to develop new calcium-based battery technologies or high-efficiency construction materials from years to days. By inputting the fundamental constants—the molar mass being the most basic—these computers can run millions of permutations to find the most efficient chemical path for the next generation of technological hardware.
In conclusion, while the question “what is the molar mass of Ca(OH)₂” begins as a simple numerical inquiry, its implications in the tech sector are vast. From the algorithms that power chemical software to the IoT systems protecting our environment, this fundamental constant is a critical piece of data in our increasingly digital world. Precision in stoichiometry is no longer just a requirement for the lab; it is a requirement for the global digital infrastructure.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.