The article title “what produces wbc” immediately triggers a cascade of inquiries, but when viewed through the lens of Tech, it unlocks a fascinating exploration of biological engineering, cellular manufacturing, and the sophisticated internal “factories” that keep us healthy. This isn’t about the biological process itself, but rather the technological understanding and manipulation of that process, from diagnostic tools to potential therapeutic innovations. Our focus will be on the technological marvels that allow us to identify, monitor, and even influence the production of White Blood Cells (WBCs), the indispensable soldiers of our immune system.
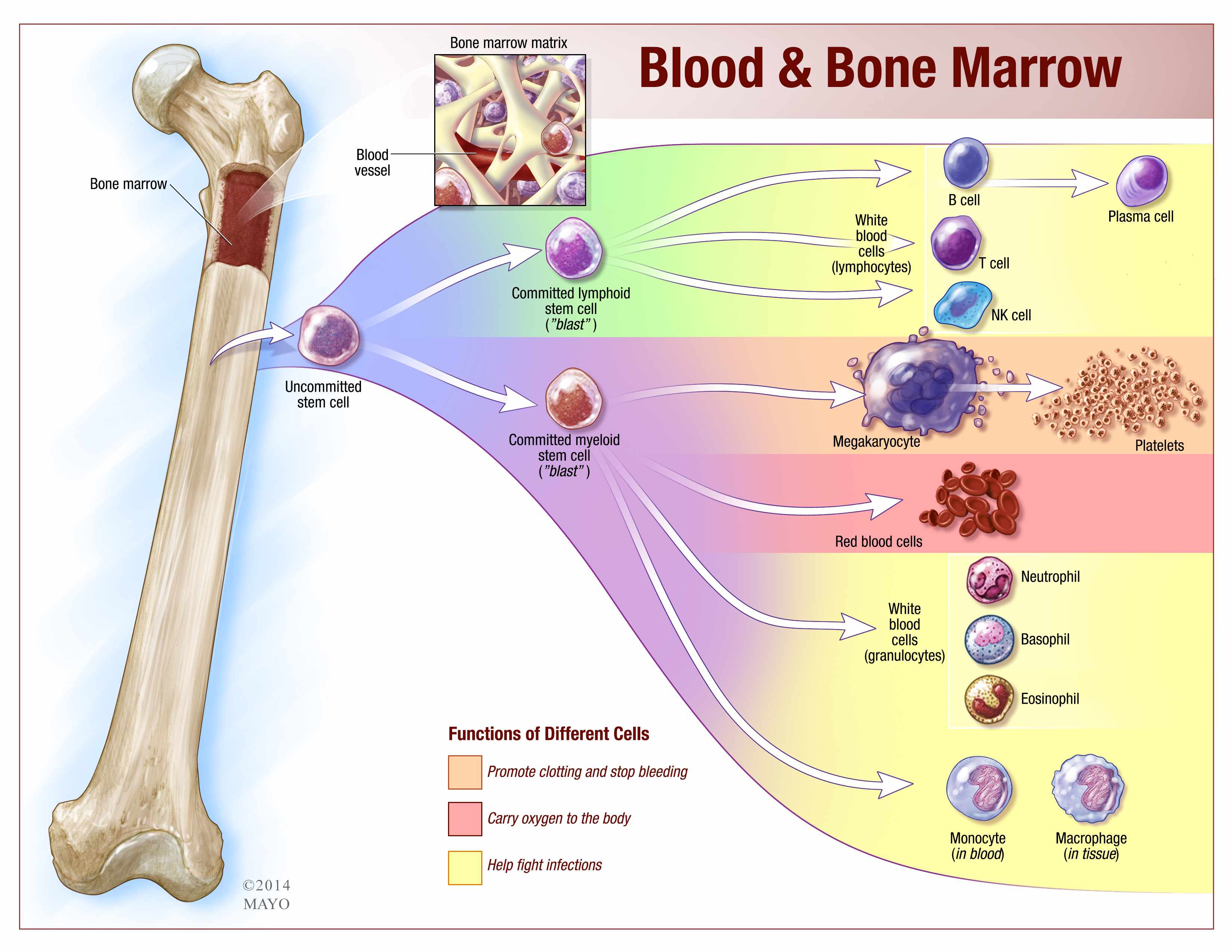
The Cellular Factories: Unveiling the Bio-Production Process through a Tech Lens
From a technological perspective, the production of WBCs is a remarkable feat of biological engineering. Our bodies, through intricate cellular mechanisms, act as highly efficient bio-factories. Understanding these processes is crucial for developing advanced diagnostic and therapeutic technologies.
Hematopoiesis: The Foundation of Cellular Manufacturing
At the heart of WBC production lies hematopoiesis, the complex biological process by which all blood cells, including leukocytes (WBCs), are generated. This process originates from hematopoietic stem cells (HSCs) residing primarily in the bone marrow. Technologically, our understanding of hematopoiesis has been revolutionized by advancements in molecular biology and genetics.
Stem Cell Technology and its Role in Monitoring Production
The identification and isolation of HSCs represent a significant technological breakthrough. Techniques like flow cytometry, a method that uses light scattering and fluorescence to sort and analyze individual cells, are indispensable in identifying HSCs. These technologies allow researchers to quantify stem cell populations and study their differentiation pathways. Furthermore, single-cell sequencing technologies enable us to delve into the genetic and epigenetic landscapes of individual HSCs, revealing the molecular switches that govern their decision to differentiate into various WBC lineages. This granular understanding is paramount for developing targeted interventions.
Gene Editing and the Future of Immune Cell Engineering
Technologies like CRISPR-Cas9 have opened up unprecedented possibilities for understanding and potentially manipulating WBC production. While still largely in the research and development phase for direct production enhancement, gene editing allows scientists to precisely modify genes involved in hematopoiesis. This could lead to the development of novel cellular therapies where specific immune cells are engineered to be more effective against diseases. For instance, researchers are exploring how to genetically modify HSCs to enhance their ability to produce specific types of T-cells or B-cells, crucial components of adaptive immunity. This is not about directly “producing” WBCs in a lab in the traditional sense of manufacturing, but rather about understanding and influencing the biological machinery that does the producing.
The Bone Marrow: A Sophisticated Biological Bioreactor
The bone marrow is not merely a passive site; it’s a dynamic microenvironment that orchestrates WBC production. This intricate ecosystem, teeming with stromal cells, growth factors, and signaling molecules, acts as a sophisticated biological bioreactor. Technologies are being developed to mimic or understand this environment ex vivo.
Bioreactor Designs for Hematopoietic Cell Culture
The development of advanced bioreactors is a testament to our ability to translate biological understanding into engineering solutions. These controlled environments are designed to optimize the conditions for stem cell proliferation and differentiation. Researchers are experimenting with different bioreactor designs, including microfluidic systems that can precisely control nutrient flow and cell-cell interactions, and 3D scaffolding technologies that mimic the extracellular matrix of the bone marrow. These bioreactors are crucial for research purposes, allowing scientists to study the fundamental processes of WBC production in a controlled setting, and hold potential for future therapeutic cell manufacturing.
In Vitro Models and Drug Discovery
In vitro models, often derived from induced pluripotent stem cells (iPSCs) or immortalized cell lines, are invaluable technological tools for studying WBC production. These models allow researchers to test the effects of various compounds and genetic manipulations on hematopoiesis without the ethical and logistical challenges of in vivo experimentation. High-throughput screening platforms, utilizing robotic automation and advanced imaging, can rapidly assess the impact of thousands of potential drug candidates on the differentiation and function of various WBC types. This accelerates the discovery of novel therapies that could modulate WBC production for therapeutic benefit.
Diagnostic Technologies: Quantifying and Characterizing WBC Production
Beyond understanding the production process itself, technology plays a critical role in diagnosing and monitoring the health of our WBC-producing systems. This involves a suite of sophisticated analytical tools.
Advanced Blood Analyzers: The Automated Workforce
Modern hematology analyzers are marvels of automation and precision. These sophisticated instruments perform complete blood counts (CBCs) with differential, providing not only the total number of WBCs but also breaking them down into their various subtypes (neutrophils, lymphocytes, monocytes, eosinophils, basophils).
Flow Cytometry in Clinical Diagnostics
Flow cytometry, beyond its research applications, is a cornerstone of clinical diagnostics for immune system disorders. It allows for the precise enumeration and characterization of different WBC populations, identifying abnormalities in their numbers, proportions, or surface marker expression. This is crucial for diagnosing conditions like leukemia, lymphoma, and immunodeficiencies, where WBC production or function is compromised. Advanced flow cytometers can analyze dozens of parameters simultaneously, providing a highly detailed snapshot of the immune system.

Imaging Technologies for Bone Marrow Assessment
When abnormalities are suspected, imaging technologies come into play. Techniques like MRI and CT scans can provide macroscopic views of the bone marrow, identifying any structural changes or infiltrations that might affect hematopoiesis. However, for microscopic assessment, advanced digital microscopy and AI-powered image analysis are transforming the interpretation of bone marrow aspirates and biopsies. These technologies can automate the detection of abnormal cells, quantify cell morphology, and assist pathologists in making more accurate and timely diagnoses.
Molecular Diagnostics: Unraveling the Genetic Blueprint
Molecular diagnostic technologies provide a deeper understanding of the genetic underpinnings of WBC production and the diseases that affect it.
Polymerase Chain Reaction (PCR) and Next-Generation Sequencing (NGS)
PCR-based assays are used to detect genetic mutations associated with certain blood cancers or inherited bone marrow failure syndromes. NGS, on the other hand, allows for comprehensive genomic profiling, identifying a wide range of genetic alterations that may be driving abnormal WBC production or function. This information is invaluable for personalized medicine, guiding treatment decisions and even predicting therapeutic responses. The ability to rapidly sequence and analyze the genomes of blood cells has revolutionized our understanding of oncogenesis and the development of targeted therapies.
Therapeutic Technologies: Intervening in WBC Production
The ultimate technological goal is to therapeutically intervene in WBC production to combat disease and enhance health. This is a rapidly evolving field.
Cell Therapies: Manufacturing Immune Cells for Treatment
The advent of cell therapies represents a paradigm shift in medicine. CAR T-cell therapy, for example, involves genetically engineering a patient’s own T-cells to recognize and attack cancer cells. This is a sophisticated form of “producing” specific, targeted immune cells ex vivo for therapeutic infusion.
Engineering Immune Cells for Cancer Therapy
The process of creating CAR T-cells is a highly technological endeavor. It involves isolating T-cells from a patient, genetically modifying them in a specialized laboratory to express chimeric antigen receptors (CARs), expanding these engineered cells to therapeutic numbers, and then re-infusing them back into the patient. This complex manufacturing process relies on precise genetic engineering techniques, sterile cell culture, and rigorous quality control measures. The success of CAR T-cell therapy highlights the potential of harnessing and augmenting the body’s own immune system through technological intervention.
Hematopoietic Stem Cell Transplantation (HSCT): The Ultimate Cellular Reset
HSCT, commonly known as bone marrow transplant, is a well-established therapeutic technology for a range of hematological malignancies and genetic disorders. It involves replacing a patient’s diseased bone marrow with healthy stem cells, which then repopulate the hematopoietic system and resume normal WBC production.
Ex Vivo Gene Therapy and Cell Engineering for HSCT
While traditional HSCT relies on healthy donor stem cells, emerging technologies are focusing on ex vivo gene therapy and cell engineering to enhance the efficacy and safety of transplants. This can involve genetically modifying a patient’s own stem cells to correct genetic defects or to make them more resistant to chemotherapy. This represents a sophisticated technological approach to address the root cause of many blood disorders, enabling the production of healthy, functional WBCs from corrected progenitor cells.
Immunomodulatory Drugs: Fine-Tuning the Production Lines
Pharmaceutical companies are continuously developing immunomodulatory drugs that can precisely influence WBC production. These drugs, often designed using sophisticated computational modeling and drug discovery platforms, can either stimulate or suppress the activity of specific WBC lineages.
Targeted Therapies for Autoimmune Diseases
In autoimmune diseases, the immune system mistakenly attacks the body’s own tissues. Technologies are being employed to develop drugs that can specifically dampen the production or activity of the overactive WBCs responsible for this attack, without compromising the overall immune response. This requires a deep understanding of the molecular signaling pathways that govern WBC differentiation and function, allowing for the design of highly specific therapeutic agents.
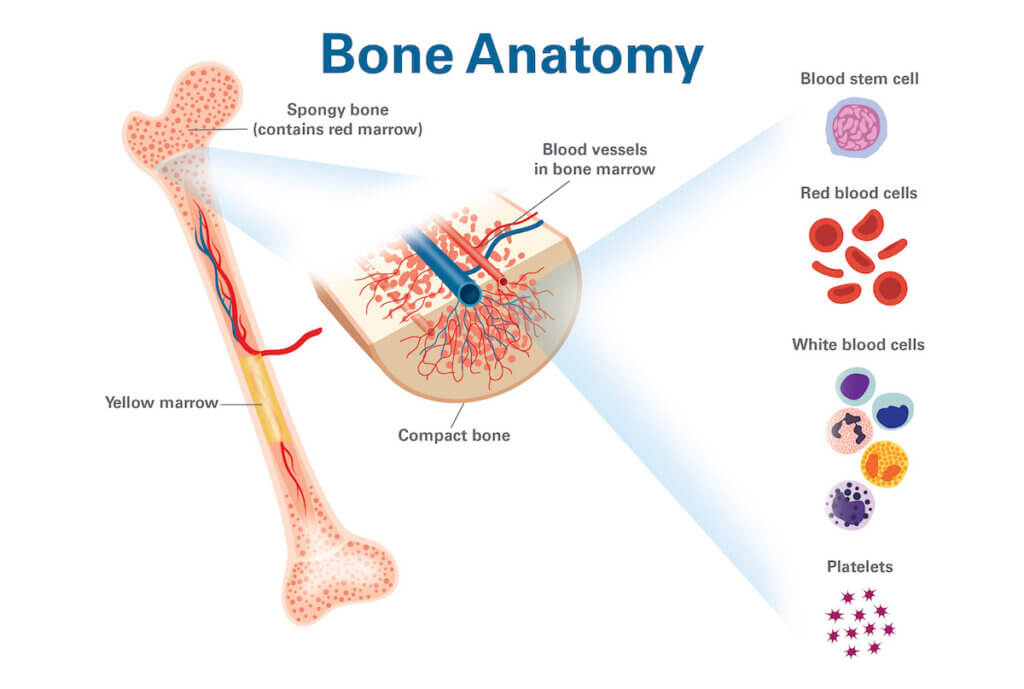
Growth Factors and Cytokines in Clinical Applications
Recombinant DNA technology has enabled the production of synthetic versions of natural growth factors and cytokines, such as G-CSF (granulocyte colony-stimulating factor). These biological molecules are used to stimulate the bone marrow to produce more neutrophils, a crucial type of WBC, particularly in patients undergoing chemotherapy or those with certain types of neutropenia. This is a direct technological application of understanding and manipulating the signals that drive WBC production.
In conclusion, while the question “what produces wbc” has a fundamental biological answer, exploring it through a Tech lens reveals a profound interplay between biology and engineering. From the intricate molecular machinery of hematopoiesis, understood through advanced sequencing and gene editing, to the sophisticated bioreactors and diagnostic analyzers that monitor and influence these processes, technology is at the forefront of understanding and harnessing the power of our immune system’s defenders. The ongoing advancements in cell therapies, gene editing, and targeted drug development promise even more innovative ways to address diseases by directly influencing the production and function of WBCs, ushering in a new era of precision medicine.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.