Enzymes, the sophisticated catalysts of life, are proteins that drive nearly every biochemical reaction within living organisms and across countless industrial and technological processes. Their unparalleled efficiency and specificity make them indispensable in biotechnology, medicine, manufacturing, and environmental science. However, the delicate balance of their three-dimensional structure is paramount to their function. When this structure is compromised, a process known as denaturation occurs, leading to a cascade of effects that can halt critical operations, reduce product efficacy, and incur substantial economic losses within technology-driven sectors.
The Fundamental Impact on Biotechnological Processes
At their core, enzymes are molecular machines. Each enzyme possesses a unique three-dimensional shape, crucial to its specific function. Within this structure lies an “active site,” a precisely contoured region where substrate molecules bind and undergo chemical transformation. The integrity of this active site is entirely dependent on the enzyme’s intricate folding pattern, maintained by various weak bonds (hydrogen bonds, ionic bonds, hydrophobic interactions).
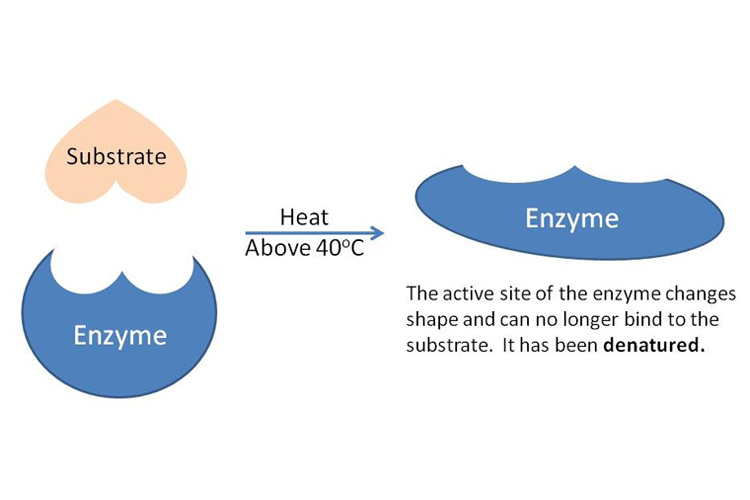
Denaturation is the process by which these delicate bonds are disrupted, causing the enzyme to lose its specific three-dimensional shape. Common culprits include extreme temperatures, drastic changes in pH, high concentrations of salts, organic solvents, or even intense mechanical agitation. As the enzyme unfolds, its active site is deformed, rendering it unable to bind effectively with its substrate or catalyze the intended reaction. The consequence is a complete or partial loss of catalytic activity.
In many technological applications, denaturation is an irreversible process, meaning the enzyme cannot regain its original structure and function even if optimal conditions are restored. This permanent inactivation presents a significant hurdle for maintaining the reliability and efficiency of enzyme-dependent technologies. From diagnostic kits to industrial bioreactors, understanding and mitigating denaturation is central to process design and product stability.
Critical Challenges in Pharmaceutical and Medical Technology
The pharmaceutical and medical technology sectors heavily rely on the precise function of enzymes, making enzyme denaturation a critical concern with profound implications for human health and economic viability.
Drug Manufacturing
Enzymes are increasingly employed in pharmaceutical synthesis due to their specificity, efficiency, and environmental friendliness. They catalyze stereoselective reactions, reducing the need for harsh chemicals and multi-step syntheses. However, if the enzymes used in these processes denature, the synthesis pathway is disrupted, leading to incomplete reactions, reduced yields of active pharmaceutical ingredients (APIs), or even the formation of undesirable byproducts. This not only results in significant material and energy waste but can also introduce safety concerns if product purity is compromised. For therapeutic enzymes themselves, which are administered directly to patients (e.g., digestive enzymes, clot-dissolving agents), denaturation before or during storage leads to inactive drugs, rendering them ineffective and potentially dangerous. Maintaining the stability of these protein-based drugs from production to patient administration is a complex challenge.
Challenges in Diagnostic Technologies
Enzyme-based diagnostic tools, such as glucose meters, pregnancy tests, and numerous clinical assays, are cornerstones of modern healthcare. These devices depend on the specific and measurable reactions catalyzed by immobilized or soluble enzymes. For instance, glucose meters use glucose oxidase to detect blood sugar levels. If the enzymes in these test strips or reagents denature, their sensitivity and accuracy plummet. A denatured enzyme could provide false negatives or positives, leading to incorrect diagnoses, delayed treatment, or unnecessary medical interventions. Ensuring the long-term stability and shelf-life of these enzyme-containing products is paramount for reliable diagnostic outcomes, often requiring precise storage conditions and robust packaging.
Biomedical Implants and Drug Delivery Systems
Emerging medical technologies incorporate enzymes into biomedical implants and advanced drug delivery systems. For example, enzymes might be used to degrade a biodegradable implant at a controlled rate or to release a drug in response to specific physiological cues. Denaturation in such applications would directly compromise the intended function, leading to premature degradation, uncontrolled drug release, or a complete failure of the device. The body’s own environment, with its varying temperatures, pH levels, and proteolytic enzymes, can also contribute to the denaturation of therapeutic enzymes or enzyme-loaded systems, posing significant design challenges for in vivo applications.
Industrial Applications: When Efficiency Fails
Beyond healthcare, enzyme denaturation profoundly impacts a vast array of industrial technologies, where optimal enzyme activity is directly linked to process efficiency, product quality, and economic returns.
Food and Beverage Processing
The food and beverage industry is a massive consumer of enzyme technology, utilizing them for everything from bread making (amylases for starch breakdown) and brewing (proteases, amylases) to cheese production (rennet) and fruit juice clarification (pectinases). Denaturation in these processes can lead to product spoilage, altered textures, undesirable flavors, and reduced shelf life. For instance, if enzymes in a dough denature prematurely, the bread may not rise correctly. In brewing, denatured enzymes can result in poor fermentation or unwanted haze. Even pasteurization, a heat-treatment process designed to eliminate harmful microorganisms, must be carefully balanced to avoid denaturing beneficial enzymes that contribute to product quality.
Biofuel Production and Waste Management
The quest for sustainable energy sources has placed enzymes at the forefront of biofuel production. Cellulases and amylases are critical for breaking down complex biomass (like agricultural waste) into fermentable sugars. However, these processes often occur under harsh conditions—high temperatures, extreme pH—that are prime triggers for enzyme denaturation. When enzymes denature, the efficiency of biomass conversion drops significantly, making biofuel production economically unviable. Similarly, in enzyme-aided waste treatment and bioremediation, denatured enzymes fail to break down pollutants effectively, prolonging environmental contamination and increasing operational costs.
Detergents and Cleaning Solutions
Commercial detergents widely incorporate enzymes (proteases, lipases, amylases) to break down stains. These enzymes must maintain stability and activity in varying water temperatures, pH levels, and in the presence of other cleaning chemicals. Denaturation of detergent enzymes directly translates to reduced cleaning power and product dissatisfaction. Manufacturers invest heavily in stabilizing these enzymes to ensure their products perform consistently throughout their shelf life.

Textile and Paper Industries
Enzymes play roles in textile processing (e.g., desizing, bio-polishing) and paper manufacturing (e.g., de-inking, improving pulp drainage). Denaturation can lead to inefficient processing, requiring more chemicals or energy, producing inferior quality products, and increasing waste. For instance, inefficient enzymatic desizing can result in poor dye uptake and uneven fabric finishes.
Technological Solutions and Innovations to Prevent Denaturation
Recognizing the critical impact of denaturation, researchers and engineers have developed sophisticated technological strategies to enhance enzyme stability and preserve their function across diverse applications.
Enzyme Engineering
A leading approach is enzyme engineering, which involves modifying an enzyme’s genetic sequence to alter its amino acid composition and, consequently, its three-dimensional structure. Techniques like directed evolution (mimicking natural selection in the lab) and rational design (predicting structural changes based on computational models) are used to create enzymes that are inherently more resistant to high temperatures, extreme pH, or the presence of organic solvents. This results in “designer enzymes” tailored for specific industrial conditions, greatly expanding their utility and economic viability.
Immobilization Techniques
Enzyme immobilization involves physically restricting enzyme movement by attaching them to an inert, insoluble support material. This can be achieved through various methods:
- Adsorption: Enzymes bind weakly to a surface.
- Covalent Bonding: Stronger, permanent attachment to a support.
- Entrapment: Enzymes are encapsulated within a polymer matrix or gel.
- Cross-linking: Enzymes are chemically linked to each other to form insoluble aggregates.
Immobilization often confers greater stability, protects enzymes from harsh environments, allows for easier separation from reaction products, and enables repeated use, significantly reducing operational costs in bioreactor systems.
Formulation Chemistry
Strategic formulation of enzyme-containing products involves adding stabilizing agents. These can include sugars (e.g., trehalose), polyols (e.g., glycerol), specific ions, or polymers that interact with the enzyme to maintain its structure or create a protective microenvironment. This approach is vital for extending the shelf life of diagnostic kits, pharmaceutical preparations, and industrial enzyme solutions.
Advanced Bioreactor Design
Modern bioreactor technologies are designed with sophisticated control systems that precisely monitor and regulate environmental conditions such such as temperature, pH, and substrate concentration. By maintaining optimal parameters, these bioreactors minimize the risk of denaturation, ensuring sustained enzyme activity and maximizing product yield in biotechnological manufacturing processes.
The Role of AI and Computational Biology in Enzyme Stability
The advent of artificial intelligence (AI) and computational biology is revolutionizing the approach to enzyme stability, offering powerful tools for prediction, design, and optimization.
Predictive Modeling
AI algorithms, particularly machine learning models, are trained on vast datasets of protein sequences, structures, and stability data. These models can predict an enzyme’s stability under various conditions and identify specific amino acid residues or structural motifs that are prone to denaturation. This predictive capability significantly accelerates the rational design of more stable enzymes, reducing the need for extensive trial-and-error experimentation.
High-Throughput Screening
Machine learning combined with high-throughput screening technologies allows for the rapid analysis of millions of enzyme variants. AI can help prioritize which enzyme mutations are most likely to yield improved stability, thereby streamlining the discovery of novel enzymes with enhanced robustness from large genomic and metagenomic libraries.
Optimizing Reaction Conditions
In industrial settings, AI-driven process control systems can monitor real-time data from bioreactors and dynamically adjust parameters like temperature, pH, and substrate feed rates. By continuously optimizing conditions, AI ensures enzymes operate at peak efficiency while minimizing the risk of denaturation due to environmental fluctuations.

Drug Discovery
For enzyme-based therapeutics, AI is being used to design proteins with improved stability in biological environments, reduced immunogenicity, and enhanced target specificity. This computational approach offers a path toward developing more effective and safer enzyme drugs.
The persistent challenge of enzyme denaturation underscores the intricate relationship between structure and function in biological catalysts. As technology advances, a deeper understanding of this phenomenon, coupled with innovative engineering and computational approaches, continues to unlock new possibilities for enzyme applications across all sectors, driving both scientific progress and economic growth.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.