Nucleotides are the fundamental monomers that make up nucleic acids, the essential molecules of life: deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Understanding the structure of a nucleotide is paramount to comprehending the intricate mechanisms of genetics, heredity, and the vast field of biotechnology. In the realm of technology, particularly in areas like bioinformatics, genetic sequencing, drug discovery, and synthetic biology, a deep appreciation for these molecular building blocks is not just academic but foundational to innovation and application. This article will delve into the three primary components that constitute a nucleotide, exploring their individual roles and how they collectively form the basis of genetic information and its manipulation.

The Pentose Sugar: The Sweet Backbone of Genetic Code
At the heart of every nucleotide lies a pentose sugar, a five-carbon sugar molecule. This sugar serves as the structural backbone to which the other two key components – a phosphate group and a nitrogenous base – are attached. The subtle difference in this pentose sugar between DNA and RNA is crucial and dictates many of their distinct biological functions and technological applications.
Deoxyribose: The Stable Foundation for DNA
In deoxyribonucleic acid (DNA), the pentose sugar is deoxyribose. The name “deoxyribose” itself hints at its defining characteristic: it is a ribose sugar that has lost an oxygen atom. Specifically, at the second carbon position of the sugar ring, deoxyribose has a hydrogen atom (-H) instead of the hydroxyl group (-OH) found in ribose. This seemingly small alteration has profound implications for DNA’s stability. The absence of the hydroxyl group at the 2′ carbon makes DNA less reactive and more resistant to hydrolysis, particularly in alkaline conditions. This enhanced stability is critical for DNA’s role as the long-term repository of genetic information, capable of withstanding cellular wear and tear over extended periods.
Technologically, the stability of deoxyribose is leveraged in numerous applications. In forensic science, the ability of DNA to remain intact even under adverse conditions allows for reliable genetic profiling from trace evidence. In diagnostic kits for genetic diseases, the longevity of DNA samples ensures accurate and reproducible results. Furthermore, the development of DNA sequencing technologies, the bedrock of modern genomics, relies on the predictable chemical behavior of deoxyribose and its attached bases to accurately read the genetic code. The robust nature of the deoxyribose backbone is also a key consideration in the design of gene therapies and the development of synthetic DNA constructs for various research and industrial purposes.
Ribose: The Dynamic Partner in RNA
In contrast, ribonucleic acid (RNA) utilizes ribose as its pentose sugar. Ribose is a typical pentose sugar that possesses a hydroxyl group (-OH) attached to its second carbon atom (the 2′ carbon). This additional hydroxyl group makes RNA more chemically reactive than DNA. While this reactivity might seem like a disadvantage, it is precisely what allows RNA to perform its diverse and dynamic roles within the cell. RNA is not primarily a long-term storage molecule; rather, it acts as a messenger, a structural component, and a catalytic enzyme, all of which benefit from its inherent chemical flexibility.
The technological implications of ribose’s reactivity are significant. For instance, in the field of gene expression analysis, the transient nature of messenger RNA (mRNA) is exploited to study cellular responses to stimuli. The ability to detect and quantify specific mRNA molecules provides insights into which genes are actively being transcribed. Moreover, RNA interference (RNAi) technologies, powerful tools for gene silencing, depend on the specific interactions and degradation pathways of short RNA molecules, which are influenced by the ribose sugar. The development of therapeutic RNAs, such as mRNA vaccines, also hinges on understanding and managing the chemical properties conferred by the ribose backbone. The inherent instability of RNA, compared to DNA, necessitates careful handling and storage in laboratory settings and during manufacturing processes, a challenge that has driven innovation in molecular biology techniques and reagent development.
The Phosphate Group: The Energy Link and Structural Connector
The second crucial component of a nucleotide is the phosphate group. This inorganic molecule, consisting of a central phosphorus atom bonded to four oxygen atoms, plays a dual role: it provides the energetic currency for molecular processes and acts as the vital link that connects individual nucleotides together to form the long chains of nucleic acids.
The Role in Energy Transfer: ATP and Beyond

In its simplest form, a nucleotide can exist as a nucleoside monophosphate, meaning it has one phosphate group attached. However, nucleotides can also exist with two or three phosphate groups, such as adenosine diphosphate (ADP) and adenosine triphosphate (ATP). ATP is arguably the most important molecule in cellular energy transfer. The bonds between the phosphate groups in ATP are high-energy bonds, meaning that a significant amount of energy is released when one or two of these phosphate groups are cleaved through hydrolysis. This released energy powers a vast array of cellular activities, from muscle contraction to the synthesis of complex molecules.
Within the technological sphere, the energy transfer capabilities of nucleotides, particularly ATP, are foundational to many biochemical assays and diagnostic tools. For example, in PCR (Polymerase Chain Reaction), the amplification of DNA sequences, the inclusion of nucleotide triphosphates (dNTPs) – which include ATP, GTP, CTP, and TTP – is essential, not only as building blocks but also because the energy released from their triphosphate form drives the polymerization process. Moreover, research into bioenergetics and the development of artificial energy-generating systems often draw inspiration from the efficiency of ATP hydrolysis. The development of biosensors that detect cellular energy levels or metabolic activity frequently relies on measuring the concentrations of ATP or its derivatives.
Linking Nucleotides: The Phosphodiester Bond
When nucleotides assemble into DNA or RNA polymers, the phosphate group becomes the crucial linker. It forms a phosphodiester bond, connecting the 5′ carbon of one pentose sugar to the 3′ carbon of the next pentose sugar in the chain. This creates the characteristic sugar-phosphate backbone of nucleic acids. Each phosphodiester bond adds to the overall length of the DNA or RNA molecule, and the precise sequence of these nucleotides, dictated by the attached nitrogenous bases, encodes the genetic information.
The formation and cleavage of phosphodiester bonds are central to numerous molecular biology techniques. DNA ligases, enzymes that catalyze the formation of these bonds, are indispensable tools in genetic engineering for joining DNA fragments. Conversely, nucleases, enzymes that cleave phosphodiester bonds, are used for DNA and RNA manipulation, purification, and degradation. The stability of the phosphodiester bond is a key factor in the integrity of genetic material, and understanding its chemistry is vital for the development of novel therapeutic agents that target nucleic acid metabolism or stability, such as some antiviral drugs. In computational biology and bioinformatics, algorithms are designed to analyze the structure and function of DNA and RNA based on the sequential arrangement of nucleotides linked by these phosphodiester bonds.
The Nitrogenous Base: The Language of Genetic Information
The third and perhaps most iconic component of a nucleotide is the nitrogenous base. These organic molecules, characterized by their ring structures containing nitrogen atoms, are the carriers of genetic information. The sequence of these bases along a DNA or RNA strand dictates the traits of an organism, the structure of proteins, and the regulation of cellular processes. There are five primary nitrogenous bases found in nucleic acids, categorized into two main groups: purines and pyrimidines.
Purines: Adenine and Guanine
The purines are double-ringed nitrogenous bases. In both DNA and RNA, the purines are adenine (A) and guanine (G). Adenine and guanine are complementary to specific pyrimidines in base pairing within double-stranded nucleic acids. In DNA, adenine pairs with thymine (T) via two hydrogen bonds, and guanine pairs with cytosine (C) via three hydrogen bonds. This specific base pairing is fundamental to DNA replication and transcription.
The presence and pairing of adenine and guanine are critical in various technological applications. In DNA sequencing, the accurate identification of A and G is essential for reconstructing the genetic code. In synthetic biology, engineered organisms are designed with specific DNA sequences containing particular ratios of A and G for desired functionalities. The study of mutations, such as point mutations where an A or G is substituted for another base, is a cornerstone of genetic diagnostics and cancer research, often facilitated by advanced sequencing technologies. Furthermore, the chemical properties of purines influence the binding affinity of drugs to DNA, a principle exploited in the design of anticancer agents that intercalate or bind to DNA.
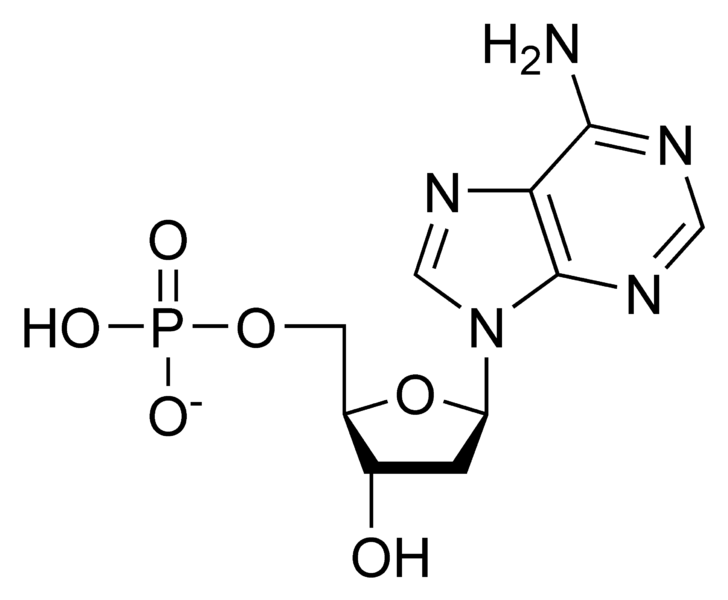
Pyrimidines: Cytosine, Thymine, and Uracil
The pyrimidines are single-ringed nitrogenous bases. The three main pyrimidines are cytosine (C), thymine (T), and uracil (U). Cytosine is found in both DNA and RNA, pairing with guanine. Thymine is found exclusively in DNA, pairing with adenine. Uracil, on the other hand, is found exclusively in RNA, where it replaces thymine and also pairs with adenine. This substitution of uracil for thymine in RNA is a key difference that contributes to RNA’s distinct chemical properties and its diverse functional roles.
The differential presence of thymine and uracil is a key indicator and target in molecular biology. For instance, the detection of uracil in a DNA sample can indicate DNA degradation or damage, as uracil arises from the deamination of cytosine. This is a critical measurement in assessing the stability of genetic material. In the development of antiviral and anticancer drugs, molecules that mimic pyrimidine bases are often used as antimetabolites, interfering with DNA or RNA synthesis in rapidly dividing cells or viruses. The precise sequence of pyrimidines, along with purines, dictates protein synthesis through codons in mRNA. Technologies like gene expression profiling rely on the accurate quantification of mRNA sequences rich in these pyrimidine bases. The understanding of how these bases interact, forming the double helix or participating in RNA folding, is a fundamental aspect of bioinformatics and computational structural biology, enabling the prediction of nucleic acid structures and functions.
In conclusion, the nucleotide, a seemingly simple molecular unit, is a masterpiece of biological engineering. Its three core components – the pentose sugar, the phosphate group, and the nitrogenous base – work in concert to store, transmit, and express the genetic information that underpins all life. For those operating within the technological landscape, from bioinformaticians to genetic engineers and medical researchers, a thorough understanding of nucleotide structure is not merely an academic exercise but a prerequisite for driving innovation, developing groundbreaking technologies, and unlocking the full potential of the biological world.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.