In the intricate symphony of modern automotive engineering, many components work tirelessly, often out of sight and out of mind, to ensure our vehicles operate efficiently and responsibly. Among these unsung heroes, few play a more critical role in public health and environmental protection than the catalytic converter. Far from being a mere muffler, this ingenious device stands as a testament to humanity’s ability to innovate solutions to complex problems, transforming harmful tailpipe emissions into something significantly less toxic. Without it, the air quality in our cities would be drastically worse, and the environmental footprint of billions of vehicles would be staggering. This comprehensive guide delves into the essence of the catalytic converter, exploring its scientific underpinnings, its evolution, its importance, and the challenges it faces in an ever-advancing technological landscape.
The Unsung Hero of Clean Air: Understanding Its Core Function
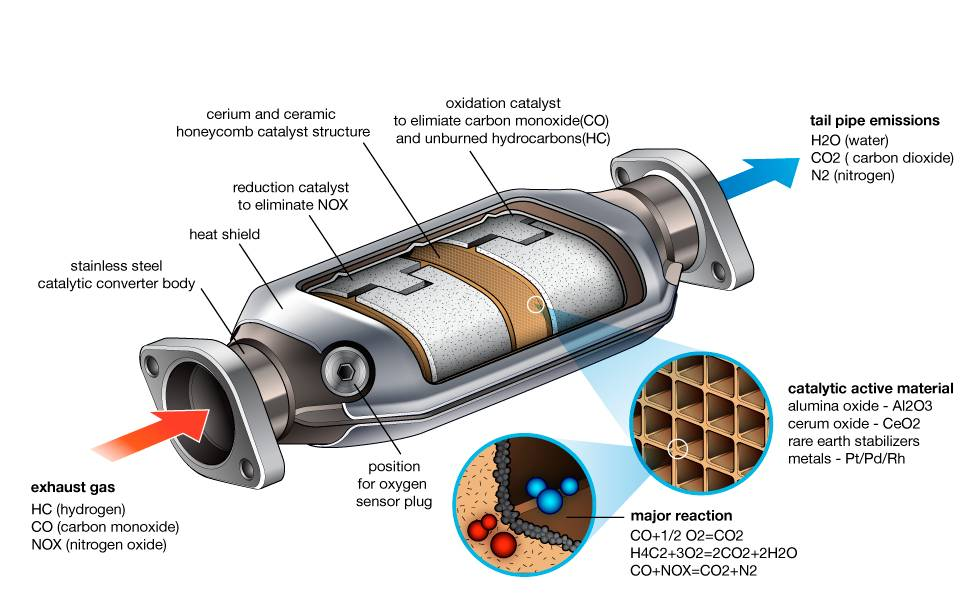
At its heart, a catalytic converter is an exhaust emission control device that converts toxic by-products of combustion in an internal combustion engine to less toxic substances by catalyzing redox reactions. It’s a compact, yet incredibly powerful, chemical processing plant nestled within your vehicle’s exhaust system, typically positioned between the engine and the muffler. Its primary mission is to safeguard the air we breathe from the pollutants generated every time a gallon of fuel is combusted.
A Chemical Reactor on Wheels
Imagine a miniature chemical laboratory constantly running behind your engine. That’s essentially what a catalytic converter is. It doesn’t filter pollutants like a sieve; instead, it initiates and accelerates chemical reactions that change the molecular structure of harmful gases. This process, known as catalysis, involves special materials that encourage chemical changes without themselves being consumed in the reaction. The beauty of this technology lies in its efficiency and passive operation, demanding no direct energy input from the driver other than the exhaust gases themselves.
The Pollutants It Tackles
Modern vehicles, especially those designed for regulatory compliance in most developed nations, employ “three-way” catalytic converters. The “three-way” refers to the three main classes of harmful compounds it is designed to mitigate:
- Nitrogen Oxides (NOx): A group of gases, primarily nitric oxide (NO) and nitrogen dioxide (NO2), formed at high temperatures in the engine. NOx contributes to smog, acid rain, and respiratory illnesses. The converter’s role is to reduce these into harmless nitrogen gas (N2) and oxygen gas (O2).
- Carbon Monoxide (CO): A colorless, odorless, and highly poisonous gas produced by incomplete combustion. When inhaled, it binds to hemoglobin in blood, preventing oxygen transport. The converter oxidizes CO into less harmful carbon dioxide (CO2).
- Unburnt Hydrocarbons (HC) / Volatile Organic Compounds (VOCs): These are essentially unburnt fuel particles that escape the combustion chamber. They contribute to smog and can be carcinogenic. The converter oxidizes HCs into carbon dioxide (CO2) and water vapor (H2O).
By tackling these three major pollutants simultaneously, the catalytic converter dramatically reduces the environmental and health impact of gasoline and diesel engines.
How It Works: The Science Behind the Conversion
The efficacy of a catalytic converter lies in its ingenious design and the specific materials it employs. It’s not just a box; it’s a carefully engineered system designed to maximize surface area and catalytic activity.
The Precious Metals Catalyst
The heart of any catalytic converter is its “catalyst” – a small quantity of precious metals. The most common catalytic materials include:
- Platinum (Pt): Excellent for oxidizing carbon monoxide and hydrocarbons.
- Palladium (Pd): Also very effective for oxidation reactions, and often used in conjunction with platinum.
- Rhodium (Rh): Crucial for the reduction of nitrogen oxides.
These metals are incredibly expensive, contributing significantly to the cost of the converter itself. Their high cost also makes catalytic converters a frequent target for theft. These metals are applied in extremely thin layers, often measured in microns, to maximize their surface area and catalytic efficiency.
The “Three-Way” Magic
The term “three-way” accurately describes the converter’s dual function involving both reduction and oxidation reactions occurring simultaneously:
- Reduction Catalyst (First Stage): This section typically uses platinum and rhodium to focus on reducing nitrogen oxides. When NOx molecules pass over the catalyst, they lose oxygen atoms, which are adsorbed by the catalyst. The nitrogen atoms then bond with other nitrogen atoms to form harmless N2 gas, and the oxygen atoms combine to form O2.
- 2NOx → N2 + xO2
- Oxidation Catalyst (Second Stage): This section, usually containing platinum and palladium, targets carbon monoxide and hydrocarbons. Here, these pollutants react with the excess oxygen molecules present in the exhaust gas (or freed from the NOx reduction).
- 2CO + O2 → 2CO2
- CxH2x+2 + [(3x+1)/2]O2 → xCO2 + (x+1)H2O
These two stages often occur within the same ceramic monolith, carefully designed to facilitate both types of reactions under optimal temperature conditions.
The Substrate and Washcoat
The precious metals aren’t just solid chunks; they are meticulously dispersed over a large surface area to maximize contact with exhaust gases. This is achieved through:
- The Substrate: Typically made of a ceramic honeycomb structure (cordierite) or, less commonly, stainless steel. This structure provides thousands of tiny channels, vastly increasing the surface area without significantly impeding exhaust flow. The honeycomb design ensures that exhaust gases are spread out evenly and have maximum exposure to the catalytic surfaces.
- The Washcoat: A thin layer of aluminum oxide (alumina) applied over the ceramic substrate. The washcoat is porous and rough, further increasing the effective surface area by many orders of magnitude. It also acts as a binder for the precious metals, holding them securely while allowing their catalytic properties to function. Some washcoats also contain “oxygen storage” components like cerium oxide, which can release or absorb oxygen depending on the exhaust gas mixture, helping to stabilize the air-fuel ratio and optimize converter efficiency.
Evolution and Importance in Automotive Technology

The catalytic converter is not a static piece of technology; its design and integration have evolved significantly since its widespread adoption, driven by increasingly stringent environmental regulations and advancements in materials science.
A Brief History of Emission Control
The concept of a catalytic converter was patented by Eugene Houdry in France in 1950, but it wasn’t until the 1970s that they became common in cars. The United States Clean Air Act of 1970 was a pivotal moment, mandating significant reductions in vehicle emissions and effectively forcing automakers to adopt catalytic converters. Early versions were “two-way” converters, only addressing CO and HCs. The advent of the “three-way” converter in the 1980s, combined with sophisticated engine management systems and oxygen sensors, marked a major leap forward, allowing for the simultaneous reduction of NOx.
Modern Enhancements: Sensors and Beyond
Modern catalytic converters don’t operate in isolation. They are integral to a sophisticated ecosystem of sensors and computer controls that optimize their performance:
- Oxygen Sensors (O2 Sensors): Located both before and after the catalytic converter, these sensors monitor the oxygen content in the exhaust gas. The pre-cat sensor helps the Engine Control Unit (ECU) adjust the air-fuel mixture to ensure it’s optimal for catalytic conversion (stoichiometric ratio). The post-cat sensor monitors the converter’s efficiency by comparing oxygen levels; a significant difference indicates a properly functioning converter.
- Engine Control Unit (ECU): The “brain” of the engine, the ECU uses data from various sensors (including oxygen sensors, mass airflow, throttle position, etc.) to precisely manage fuel injection and ignition timing. This precise control is crucial for maintaining the ideal exhaust gas composition that the catalytic converter needs to operate efficiently.
- Warm-up Catalysts and Close-Coupled Converters: Because catalytic converters only work effectively at high temperatures (typically above 250-300°C), modern vehicles often place them very close to the engine manifold (“close-coupled”) to heat up faster after a cold start, minimizing emissions during the critical warm-up phase. Some also incorporate electric heaters for even faster activation.
Its Role in Vehicle Performance and Longevity
While primarily for emissions, a healthy catalytic converter indirectly contributes to overall vehicle performance and longevity. A clogged or damaged converter creates back pressure in the exhaust system, which can severely restrict engine power, reduce fuel efficiency, and even cause engine damage dueasting to increased heat and stress. Conversely, a free-flowing and efficient converter ensures optimal exhaust scavenging, allowing the engine to breathe properly and perform as designed.
Common Issues and Technological Challenges
Despite its robust design, the catalytic converter is not immune to problems. Understanding these issues is key to maintenance and appreciation of the technology.
Signs of a Failing Converter
A catalytic converter can fail for several reasons, often due to engine issues that send unburnt fuel or oil into the exhaust, leading to “poisoning” or “melting” of the catalyst. Common symptoms include:
- Reduced Engine Performance: Sluggish acceleration, reduced power, poor fuel economy due to excessive back pressure.
- Rattling Noise: Internal ceramic substrate can break apart, causing a metallic rattle, especially when starting the engine or accelerating.
- Check Engine Light (CEL): Often triggered by oxygen sensor readings indicating inefficient catalytic conversion (e.g., P0420 or P0430 codes).
- Sulphur or Rotten Egg Smell: Unconverted hydrogen sulfide passing through the exhaust system can produce this distinctive odor.
- Failed Emissions Test: The most direct indicator of a converter not performing its job.
The Problem of Theft
One of the most unfortunate challenges associated with catalytic converters is their susceptibility to theft. Due to the valuable precious metals (platinum, palladium, rhodium) contained within them, they are a lucrative target for thieves. Removing a converter can be done relatively quickly with the right tools, leading to significant repair costs for vehicle owners and contributing to a growing illegal market for these materials. This issue highlights the intrinsic value of the technology beyond its environmental benefits.
Future of Emission Control Tech
As the automotive industry shifts towards electrification, the role of the traditional catalytic converter will naturally diminish in pure electric vehicles. However, for hybrid vehicles and the vast number of internal combustion engine (ICE) vehicles still on the road, advanced catalytic technologies continue to be developed. This includes new catalyst formulations that require less precious metal, operate more efficiently at lower temperatures, and are more resistant to poisoning. There’s also research into alternative aftertreatment systems and more integrated emission control technologies that work seamlessly with engine design.
Maintaining Your Catalytic Converter and Looking Ahead
While seemingly a “fit and forget” component, proper vehicle maintenance significantly impacts the lifespan and efficiency of your catalytic converter.
Tips for Longevity
- Regular Engine Maintenance: Address misfires, oil consumption, and rich fuel mixtures promptly. These issues can send unburnt fuel or oil into the converter, leading to overheating and damage.
- Avoid Short Trips: Catalytic converters need to reach operating temperature to be effective. Frequent short trips can prevent this, leading to carbon buildup and reduced efficiency over time.
- Use the Correct Fuel: Using the recommended octane and avoiding leaded gasoline (which is generally obsolete but historically a problem) prevents catalyst poisoning.
- Address Warning Lights Immediately: A “Check Engine” light, especially one related to emission control, should never be ignored. Early diagnosis can prevent expensive converter replacement.

The Environmental Imperative
The catalytic converter remains a cornerstone of environmental protection in the automotive sector. Its technology has been instrumental in cleaning up urban air and reducing the overall ecological impact of transportation for decades. As we move towards a future dominated by electric vehicles, understanding the catalytic converter’s past and present role helps us appreciate the scale of engineering challenges overcome in the pursuit of cleaner air and a healthier planet. Its legacy will undoubtedly influence future innovations in emission control, even as propulsion technologies continue to evolve.
In conclusion, the catalytic converter is far more than a simple exhaust component; it is a sophisticated piece of chemical engineering that exemplifies the power of technology to address pressing environmental concerns. From its precious metal catalysts to its intricate honeycomb structure, every aspect of its design is geared towards transforming harmful pollutants into benign gases, quietly safeguarding our planet one exhaust puff at a time. It stands as a profound technological achievement, an essential guardian of public health, and a continuous reminder of the engineering ingenuity within our vehicles.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.