In the intricate tapestry of chemical reactions, few processes are as fundamental and widely applicable as radical chain reactions. These reactions, driven by the fleeting yet potent existence of free radicals, form the bedrock of countless industrial processes and natural phenomena. At the heart of these chains lies the propagation step, a crucial stage where the radical is regenerated, perpetuating the reaction cycle. Understanding the characteristics of this vital step is paramount for chemists seeking to control, optimize, and harness the power of radical chemistry.
The Essence of Radical Chain Propagation
Radical chain propagation represents the core mechanism by which a radical reaction sustains itself. Unlike initiation, which creates the initial radicals, or termination, which extinguishes them, propagation is the phase where the chain actively grows and transforms reactants into products. The defining characteristic of this step is its self-perpetuating nature, where a radical species reacts with a stable molecule to generate a new radical and a stable product. This cyclical regeneration ensures that a single initiation event can lead to the transformation of a vast quantity of reactants.
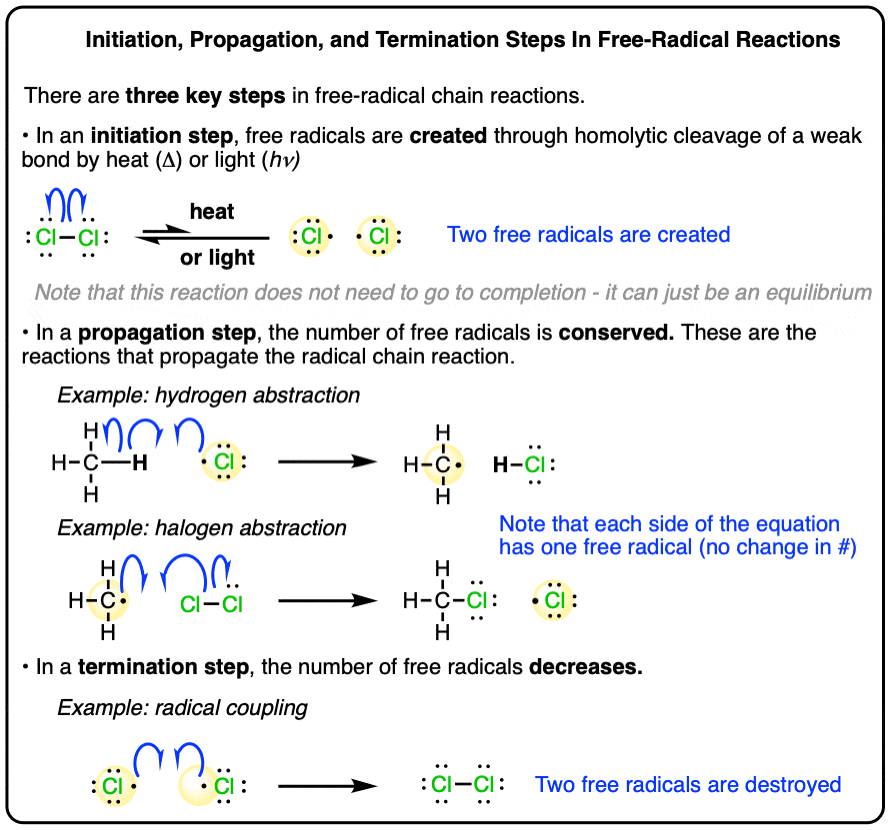
The Regenerative Cycle
The fundamental equation for a radical chain propagation step can be generalized as:
R• + X-Y → R-X + Y•
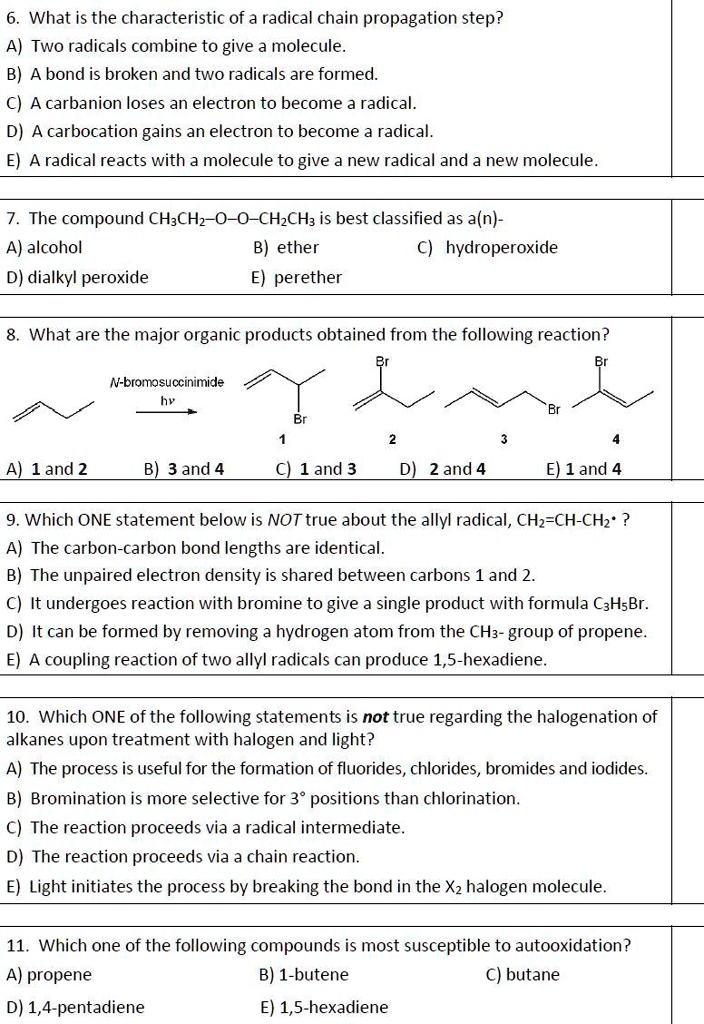
Here, R• represents a free radical, X-Y is a reactant molecule, R-X is a product molecule, and Y• is a newly formed free radical. The key here is that the radical on the left (R•) is consumed, but a new radical (Y•) is simultaneously generated. This newly formed radical can then go on to react with another molecule of X-Y, continuing the chain. This iterative process is what distinguishes propagation from other reaction steps.
Energetic Considerations
For a propagation step to be favorable and efficient, the bond being broken in the reactant molecule (X-Y) must be weaker than or comparable in strength to the new bond being formed (R-X). This is because breaking a bond requires energy, while forming a bond releases energy. If the energy released from forming the new bond significantly exceeds the energy required to break the old one, the reaction will proceed exothermically, providing the driving force for the propagation. Conversely, if bond breaking requires substantially more energy than is released by bond formation, the propagation step will be energetically unfavorable and unlikely to occur.
Selectivity and Specificity
While the fundamental principle of radical regeneration is universal, the specific outcome of a propagation step is heavily influenced by the nature of the radical and the reactant molecule. This leads to a remarkable degree of selectivity. A particular radical will often preferentially abstract an atom or add to a specific site within a larger molecule. This specificity is governed by factors such as:
- Radical stability: More stable radicals are generally less reactive and will exhibit higher selectivity. They will preferentially react with weaker bonds or electron-rich sites.
- Bond strengths: As mentioned earlier, the relative strengths of bonds play a crucial role. Radicals will tend to abstract atoms from weaker bonds.
- Steric hindrance: Bulky radicals or substituents on the reactant molecule can impede the approach of the radical, influencing where the reaction occurs.
- Electronic effects: The electron density distribution within the reactant molecule can direct the attack of the radical. Electron-deficient sites are more susceptible to attack by electron-rich radicals, and vice-versa.
These factors allow chemists to design reactions with a high degree of control, targeting specific bonds or positions within complex molecules.
Key Features of Radical Chain Propagation
The characteristic of a radical chain propagation step is not merely its ability to regenerate a radical; it is a multifaceted phenomenon characterized by several key features that dictate the course and efficiency of the overall reaction. These features provide the framework for understanding and manipulating radical processes.
Unimolecular vs. Bimolecular Propagation
Propagation steps can broadly be categorized into two types based on the number of molecules involved:
- Bimolecular Propagation: This is the most common scenario, involving the reaction between a radical and a stable molecule, as exemplified by the general equation R• + X-Y → R-X + Y•. Two species collide and react, leading to the formation of a new radical and a product. Examples include hydrogen atom abstraction from alkanes by halogens or alkyl radicals, and addition of radicals to alkenes.
- Unimolecular Propagation: While less common, unimolecular propagation can occur in specific cases where a radical can undergo an internal rearrangement or fragmentation to generate a new radical. This typically involves a highly unstable radical undergoing a transformation to a more stable, albeit still radical, species. For instance, certain peroxy radicals can undergo cyclization reactions.
The distinction between these two types is significant for understanding reaction kinetics and mechanisms. Bimolecular steps are generally second-order with respect to the concentrations of the reacting species, while unimolecular steps are first-order.
Energetic Driving Force and Thermodynamics
The thermodynamic favorability of a propagation step is a critical determinant of its occurrence. As discussed, the bond dissociation energy (BDE) plays a pivotal role. A propagation step will proceed readily if the BDE of the bond being broken in the reactant is lower than the BDE of the new bond being formed in the product. For instance, the abstraction of a hydrogen atom from an alkane by a chlorine radical is favorable because the C-H bond is weaker than the H-Cl bond.
However, it’s important to consider not just bond strengths but also the overall enthalpy change of the reaction. A propagation step with a negative enthalpy change (exothermic) is thermodynamically favored. While a reaction might be kinetically facile, it will not proceed indefinitely if it is energetically unfavorable.
Kinetic Considerations: Activation Energy
Beyond thermodynamics, the kinetics of a propagation step are dictated by its activation energy (Ea). The activation energy is the minimum energy required for the reaction to occur. A lower activation energy means the reaction proceeds faster, as a larger proportion of molecules will possess sufficient energy to overcome the energy barrier upon collision.
Factors influencing the activation energy of a propagation step include:
- The nature of the radical and the attacking atom/group: Steric and electronic factors can influence the ease with which the radical approaches and interacts with the reactant.
- The strength of the bond being broken: Weaker bonds generally have lower activation energies for cleavage.
- The transition state geometry: The specific arrangement of atoms in the transition state plays a significant role in determining the activation energy.
Chemists often aim to lower the activation energy of desired propagation steps through catalyst design or by modifying reaction conditions to enhance the overall reaction rate.
The Role of Propagation in Complex Radical Systems
The seemingly simple cycle of radical generation and consumption in propagation steps belies their profound impact on complex chemical systems. These steps are not isolated events but are intricately woven into the fabric of larger reaction mechanisms, influencing product distribution, reaction rates, and even the overall feasibility of a chemical transformation.
Chain Length and Efficiency
The efficiency of a radical chain reaction is often quantified by its chain length (CL). The chain length represents the average number of product molecules formed per initiation event. A high chain length signifies an efficient propagation mechanism, where termination events are relatively infrequent. The characteristics of the propagation step directly influence the chain length. If propagation steps are fast and have low activation energies, they can outcompete termination steps, leading to longer chains. Conversely, slow or energetically unfavorable propagation steps will result in short chains and lower overall yields.
Influence on Product Selectivity
As highlighted earlier, the inherent selectivity of propagation steps is a cornerstone of controlling product formation. In complex molecules with multiple potential reaction sites, the propagation step will dictate which bonds are broken and which new bonds are formed. For instance, in the chlorination of an alkane, primary, secondary, and tertiary hydrogens have different BDEs and associated activation energies for abstraction by a chlorine radical. This difference in reactivity, stemming from the propagation step, leads to a mixture of chlorinated products, with the distribution dictated by the relative rates of abstraction at each position. Understanding these subtle differences in propagation step characteristics allows for targeted synthesis.
Interaction with Other Reaction Steps
Propagation steps do not operate in isolation. They are in constant interplay with initiation and termination steps.
- Initiation: The rate and efficiency of the initiation step determine the initial concentration of radicals available to propagate the chain. A slow initiation can bottleneck the entire process, even if propagation is very facile.
- Termination: Termination steps, where two radicals combine to form a stable molecule, effectively remove radicals from the system and break the chain. The rate of termination relative to propagation is crucial. If termination is too fast, the chain will be short-lived. If propagation is too fast relative to termination, side reactions or runaway reactions could occur.
The balance between these three types of steps is a delicate equilibrium that dictates the overall outcome of a radical chain reaction. Designing effective radical processes often involves carefully tuning conditions to favor efficient propagation while minimizing premature termination.

Conclusion: The Unsung Hero of Radical Chemistry
The radical chain propagation step, though often overshadowed by the more dramatic initiation or the finality of termination, is the engine that drives radical reactions. Its defining characteristic is the self-perpetuating cycle where a radical reacts with a stable molecule to generate both a product and a new radical, thus continuing the chain. This regenerative process, governed by energetic considerations, bond strengths, and kinetic barriers, allows for the transformation of vast quantities of material from a single initiating event.
The inherent selectivity of propagation steps, dictated by factors such as radical stability and electronic effects, offers chemists a powerful tool for controlling reaction outcomes and designing sophisticated synthetic pathways. By understanding and manipulating the nuances of these propagation mechanisms, from the distinction between unimolecular and bimolecular events to the precise energetic driving forces and activation energies, researchers can optimize reaction efficiencies, enhance product selectivity, and ultimately unlock the full potential of radical chemistry in diverse scientific and industrial applications. The radical chain propagation step is, indeed, the unsung hero, silently orchestrating the complex dance of chemical transformation.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.