When we lift the hood of a modern vehicle, we are often greeted by a complex landscape of plastic shrouds, wiring looms, and high-performance engineering. Yet, tucked away in the corner is a component that has remained fundamentally unchanged in its core chemistry for over 160 years: the lead-acid battery. While the external casing and the digital systems that manage it have evolved into high-tech marvels, the “acid” inside remains the lifeblood of the automotive electrical system.
In the context of modern technology and engineering, understanding what this acid is—and how it functions as a critical hardware interface—is essential for anyone interested in the intersection of chemical engineering and digital automotive systems.

The Core Component: Sulfuric Acid and the Electrolytic Solution
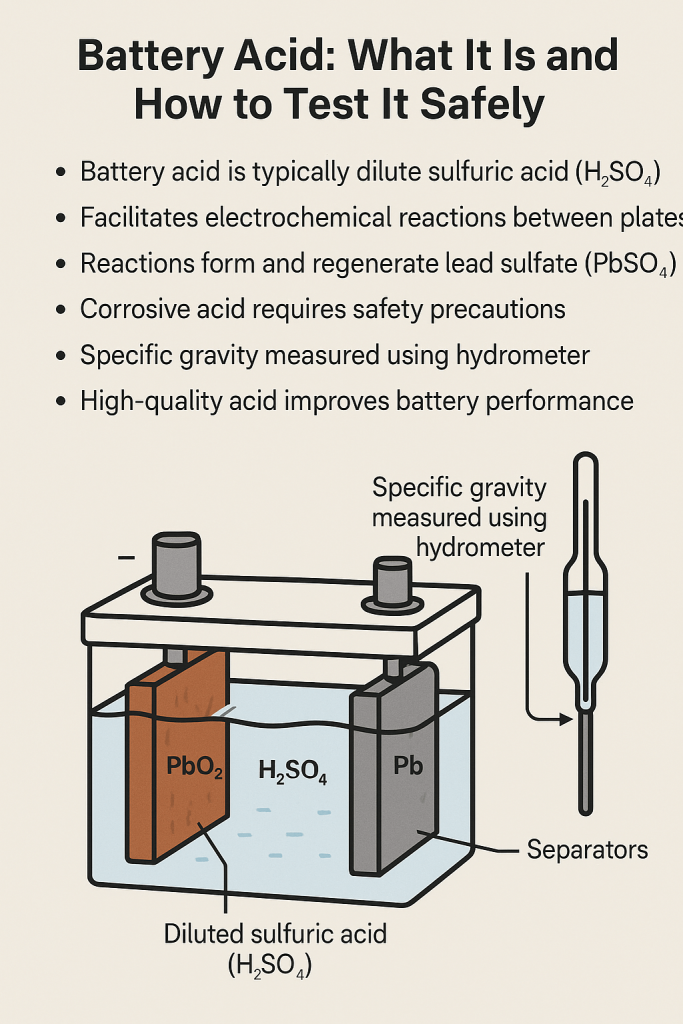
The acid found in traditional car batteries is sulfuric acid ($H2SO4$) diluted with purified water. This mixture is known as the electrolyte. In a standard 12-volt lead-acid battery, the electrolyte typically consists of approximately 35% to 50% sulfuric acid and 50% to 65% water.
The Chemical Reaction: Electrolytes and Electron Flow
From a technological standpoint, the battery is not merely a storage tank for electricity; it is a chemical energy conversion device. The sulfuric acid serves as the medium that facilitates the movement of ions between the positive and negative plates. When the battery is discharging, the acid reacts with the lead dioxide ($PbO_2$) on the positive plate and the sponge lead ($Pb$) on the negative plate.
This reaction produces lead sulfate ($PbSO_4$) and releases electrons, creating the electrical current required to power the vehicle’s starter motor and onboard computers. Without the specific reactive properties of sulfuric acid, the rapid discharge of high-amperage current needed to crank an internal combustion engine would be impossible within such a compact form factor.
Concentration and Specific Gravity
In the world of battery diagnostics, the “tech” used to measure the health of the acid is often a hydrometer or a refractometer. These tools measure the specific gravity of the electrolyte. As a battery discharges, the sulfate ions move out of the acid and onto the plates, leaving the liquid mostly as water. This reduces the density (specific gravity) of the fluid. Modern smart chargers use algorithms to detect these changes in internal resistance, allowing the hardware to adjust charging curves to prevent “stratification”—a condition where the acid and water separate, leading to premature hardware failure.
Evolution of Automotive Power: From Flooded to AGM and Gel
While the base chemistry remains sulfuric acid, the way this acid is packaged has seen significant technological leaps. We have moved far beyond the “flooded” batteries that required manual water top-ups, entering an era of Maintenance-Free (MF) and VRLA (Valve Regulated Lead Acid) technologies.
Absorbent Glass Mat (AGM) Advancements
AGM technology represents a major shift in how the acid is handled. Instead of the acid sloshing freely between plates, it is absorbed into a very fine fiberglass mat. This makes the battery spill-proof and highly resistant to vibration—a key requirement for off-road vehicles and high-performance gadgets.
From an engineering perspective, AGM batteries allow for a faster delivery of power and a quicker recharge rate. This tech is particularly crucial for modern “Stop-Start” systems. When your car’s engine shuts off at a red light, the AGM battery must keep the infotainment, GPS, and climate control running, then deliver a massive burst of energy to restart the engine instantly. The “acid” here is still sulfuric, but the glass mat delivery system is a triumph of materials science.
Gel Batteries and Deep Cycle Innovation
Gel batteries are another variation where silica is added to the sulfuric acid to turn it into a thick, jelly-like substance. This technology is less common in automotive starting batteries but is a staple in the “tech-adjacent” world of renewable energy storage and electric wheelchairs. The gelled electrolyte prevents the evaporation of the acid and provides superior performance in extreme temperature fluctuations, showcasing how chemical modifications can adapt old tech for new specialized gadgets.
The Digital Integration: Smart Charging and Battery Management Systems (BMS)
In contemporary automotive tech, the battery is no longer a “dumb” component. It is now part of an integrated digital ecosystem. The interaction between the sulfuric acid and the lead plates is now monitored by sophisticated Battery Management Systems (BMS).

Sensors and Monitoring Hardware
Modern vehicles are equipped with Intelligent Battery Sensors (IBS) usually located on the negative terminal. These sensors monitor the voltage, current, and temperature of the electrolyte. Because the chemical activity of sulfuric acid is highly temperature-dependent (it becomes less active in the cold), the BMS uses this data to communicate with the vehicle’s ECU (Engine Control Unit).
If the sensor detects that the acid is too cold to provide a full charge, the alternator’s output is digitally adjusted. This level of hardware-software integration ensures that the chemical lifecycle of the acid is maximized, preventing the “sulfation” that occurs when a battery remains in a low-state of charge for too long.
The Role of AI in Predicting Battery Failure
The next frontier in battery tech is the application of machine learning to predict the “State of Health” (SoH). By analyzing thousands of data points regarding how the sulfuric acid reacts to different load cycles, AI tools can now predict when a battery is likely to fail months before it actually dies. This predictive maintenance is a cornerstone of fleet management software and is becoming a standard feature in high-end electric and hybrid vehicles that still utilize a 12V lead-acid system for their low-voltage electronics.
Safety, Tech-Driven Maintenance, and Environmental Impact
Working with sulfuric acid requires a high degree of technical caution. It is highly corrosive and can produce flammable hydrogen gas during charging. However, modern technology has made the handling of these chemicals safer than ever before.
Thermal Management Systems
One of the primary enemies of battery acid is heat. High temperatures accelerate the chemical reaction, leading to “thermal runaway” where the battery expands and potentially leaks. Tech-heavy EVs and high-performance internal combustion cars now utilize dedicated thermal management systems—sometimes even liquid cooling—to keep the battery acid within an optimal temperature range (usually around 25°C or 77°F). This engineering keeps the chemical reactions stable and extends the hardware’s lifespan.
Circular Economy: Tech in Lead-Acid Recycling
Perhaps the most impressive “tech” story involving car battery acid is the recycling process. Lead-acid batteries are the most recycled consumer product in the world, with a nearly 99% recycle rate in many developed nations.
Specialized industrial tech allows for the recovery of the sulfuric acid, which is either neutralized and turned into sodium sulfate (used in laundry detergents) or purified and reused in new batteries. This closed-loop system is a benchmark for the “Green Tech” industry, proving that even “old” chemical technologies can be part of a sustainable digital and industrial future.
The Future Landscape: Moving Beyond Liquid Electrolytes?
As we look toward the future of technology, the role of sulfuric acid is being challenged by new materials. The tech industry is currently in a race to find more energy-dense, lighter, and faster-charging alternatives.
Solid-State Developments
The buzzword in the tech world today is “Solid-State.” While currently focused on lithium-ion replacements, the research into solid electrolytes aims to remove the “liquid” (the acid) from the equation entirely. By replacing liquid sulfuric acid with a solid ceramic or polymer electrolyte, engineers can create batteries that are non-flammable and significantly more energy-dense. However, the cost of this technology remains high, keeping the reliable, acid-based battery at the top of the automotive food chain for the time being.
Lithium-Ion vs. Traditional Lead-Acid
We are seeing a trend where high-end gadgets and even some performance cars (like Porsches) are replacing the heavy lead-acid battery with 12V Lithium-Ion (LiFePO4) units. These batteries don’t use sulfuric acid; they use lithium salts in an organic solvent. While they offer a weight saving of up to 70%, they require even more complex digital protection circuits to prevent damage. The “tech” here is a trade-off: the simplicity and robustness of sulfuric acid versus the high-performance but delicate nature of lithium chemistry.
Conclusion
The “acid” in a car battery—sulfuric acid—is a masterclass in functional chemistry that has survived the transition from the analog to the digital age. While it may seem like a relic of the 19th century, its integration with modern sensors, AGM delivery systems, and AI-driven management tools makes it a vital piece of 21st-century hardware.
As we move deeper into the era of electric mobility and smart cities, the way we manage, monitor, and recycle this potent chemical will continue to evolve. Whether it’s through the glass mats of an AGM battery or the smart sensors of a BMS, the relationship between this powerful acid and the technology that harnesses it remains one of the most successful partnerships in the history of engineering.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.