In the realm of technology, understanding the fundamental forces that govern the behavior of matter at the molecular level is crucial for innovation. From the development of advanced materials to the design of sophisticated electronic components, the subtle interplay between molecules can have profound implications. One such fundamental interaction, often overlooked in broader technological discussions, is the dipole-dipole interaction. While seemingly abstract, this intermolecular force plays a surprisingly significant role in various technological applications, influencing everything from the efficiency of your smartphone screen to the performance of specialized sensors.

At its core, a dipole-dipole interaction is a weak, attractive force that arises between molecules that possess a permanent electric dipole moment. This means that the electrical charges within the molecule are not evenly distributed, creating a slight positive and a slight negative end. When these polar molecules come into close proximity, the positive end of one molecule is attracted to the negative end of another, leading to a temporary, transient bond. This interaction is distinct from stronger forces like covalent or ionic bonds, and also from weaker, induced dipole forces like London dispersion forces. It’s a middle ground, yet its influence is pervasive.
Understanding dipole-dipole interactions is not merely an academic exercise for the tech industry. It’s a practical necessity that drives advancements in materials science, chemistry, and engineering. By manipulating and understanding these forces, engineers can design materials with specific properties, develop more efficient chemical processes, and create novel electronic devices. This article will delve into the nature of dipole-dipole interactions and explore their impactful applications within the technological landscape.
The Molecular Basis: Understanding Dipoles and Their Interactions
To truly grasp the technological relevance of dipole-dipole interactions, we must first understand their molecular underpinnings. This involves dissecting what constitutes a dipole and how these dipoles interact to create an attractive force.
Electronegativity and Molecular Polarity
The foundation of a permanent dipole moment lies in the concept of electronegativity. Electronegativity is a measure of an atom’s ability to attract shared electrons in a chemical bond. When two atoms with significantly different electronegativities form a covalent bond, the electrons in the bond will be pulled closer to the more electronegative atom. This unequal sharing of electrons results in a partial negative charge on the more electronegative atom and a partial positive charge on the less electronegative atom.
Consider a molecule like water (H₂O). Oxygen is significantly more electronegative than hydrogen. In a water molecule, the oxygen atom pulls the shared electrons from both hydrogen atoms towards itself. This creates a partial negative charge (δ-) around the oxygen atom and partial positive charges (δ+) around the hydrogen atoms. Because the water molecule is bent, these partial charges are not symmetrical. The molecule as a whole has a distinct positive end and a negative end, making it a polar molecule, or a dipole.
The Nature of the Dipole-Dipole Attraction
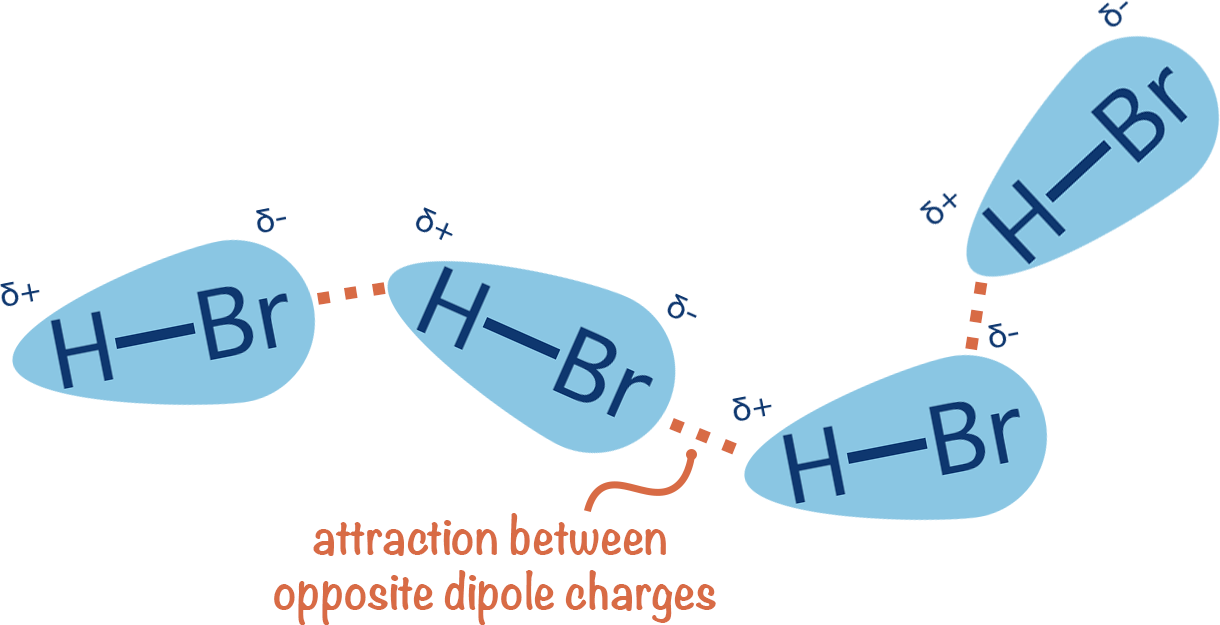
When two polar molecules, each acting as a dipole, encounter each other, they tend to orient themselves in a specific way to minimize their potential energy. The partially positive end of one molecule will be attracted to the partially negative end of another. This electrostatic attraction between the oppositely charged ends of adjacent polar molecules is the dipole-dipole interaction.
This attraction is weaker than the forces within an ionic or covalent bond, as it involves partial charges rather than full charges. However, it is stronger than the London dispersion forces, which arise from temporary fluctuations in electron distribution and are present in all molecules, polar or nonpolar. The strength of the dipole-dipole interaction depends on the magnitude of the dipole moments of the interacting molecules. Molecules with larger dipole moments will experience stronger dipole-dipole attractions. These interactions are crucial in determining the physical properties of substances, such as boiling point and solubility, which in turn have direct technological implications.
Technological Applications: Where Dipole-Dipole Interactions Make a Difference
The fundamental understanding of dipole-dipole interactions translates into tangible advancements across a spectrum of technologies. By leveraging these forces, engineers and scientists can design and improve materials, processes, and devices.
Materials Science and Engineering: Tailoring Properties with Polarity
The properties of many materials are intimately linked to the intermolecular forces present between their constituent molecules. Dipole-dipole interactions play a pivotal role in dictating the physical and chemical characteristics of a vast array of technologically relevant materials.
One prominent example is the development of specialty polymers. Many polymers used in advanced applications, such as high-performance plastics, adhesives, and coatings, are designed with polar functional groups. These groups introduce permanent dipole moments into the polymer chains. The resulting dipole-dipole interactions between adjacent polymer chains can significantly influence the material’s strength, flexibility, thermal stability, and adhesion properties. For instance, stronger dipole-dipole interactions can lead to materials with higher tensile strength and melting points, making them suitable for demanding engineering applications. Conversely, carefully controlled dipole-dipole interactions can enhance the solubility of polymers in specific solvents, which is critical for manufacturing processes like solution casting or spin coating.
Furthermore, in the field of liquid crystals, dipole-dipole interactions are fundamental to their unique electro-optical properties. Liquid crystals are substances that exhibit properties between those of conventional liquids and solid crystals. Their molecules often possess permanent dipole moments. When an electric field is applied, these polar molecules align themselves due to the dipole-dipole interactions and their response to the electric field. This controlled alignment is the very basis of how liquid crystal displays (LCDs) in our smartphones, televisions, and computer monitors work, allowing us to control the passage of light and create images.
Chemical Processes and Separation Technologies
Beyond materials, dipole-dipole interactions are instrumental in optimizing various chemical processes and enabling sophisticated separation technologies that are vital for industrial manufacturing and scientific research.
In solvent selection, the principle of “like dissolves like” is heavily influenced by dipole-dipole interactions. Polar solvents, characterized by molecules with permanent dipole moments, are effective at dissolving other polar solutes. This is because the dipole-dipole attractions between the solvent and solute molecules are strong enough to overcome the attractions between solute molecules themselves and between solvent molecules themselves. This understanding is critical in chemical synthesis, where choosing the right solvent can dramatically affect reaction rates, yields, and product purity. For instance, in the pharmaceutical industry, precise control over solubility is essential for drug formulation and delivery.
Chromatographic separation techniques, widely used for analyzing and purifying complex mixtures, also rely on differential dipole-dipole interactions. In techniques like liquid chromatography, stationary phases and mobile phases are chosen such that molecules with varying degrees of polarity will interact differently. Molecules with stronger dipole moments will interact more strongly with polar stationary phases, leading to slower elution, while nonpolar molecules will pass through more quickly. This ability to separate compounds based on their polarity, influenced by dipole-dipole interactions, is indispensable in fields ranging from environmental monitoring to quality control in food production.
Advanced Technologies: The Subtle Influence of Molecular Forces
As technology progresses into increasingly intricate and miniaturized domains, the subtle influence of intermolecular forces, including dipole-dipole interactions, becomes even more pronounced. These forces are not just background players; they are actively engineered and exploited to achieve novel functionalities.
Nanotechnology and Surface Science
In the realm of nanotechnology, where materials are manipulated at the atomic and molecular scale, dipole-dipole interactions become a dominant factor. When fabricating nanoscale structures, the forces between individual atoms, molecules, and surfaces are paramount. Dipole-dipole interactions can influence the self-assembly of nanoparticles, dictating how they arrange themselves into ordered structures. This is crucial for creating functional nanomaterials with tailored electronic, optical, or catalytic properties. For instance, controlled dipole-dipole interactions can guide the formation of nanowires or molecular monolayers on surfaces, which are building blocks for future electronic devices and sensors.
Furthermore, surface functionalization often involves introducing polar groups onto a material’s surface to modify its properties. This can enhance its adhesion to other materials, alter its wettability (how it interacts with liquids), or facilitate the binding of specific molecules for sensing applications. These modifications often rely on creating or exploiting dipole-dipole interactions between the surface and the surrounding environment or other adsorbed molecules.
Biosensors and Medical Devices
The application of dipole-dipole interactions extends into the sensitive domain of biosensors and medical devices. Biological molecules, such as proteins and DNA, are inherently polar due to the presence of charged and polar functional groups. These molecules interact with their environment and with each other through a complex interplay of forces, including dipole-dipole interactions.
In the design of biosensors, understanding these interactions is key to achieving specific and sensitive detection of target molecules. For example, a biosensor might immobilize a specific protein onto a surface. The surface chemistry is carefully designed to promote dipole-dipole interactions with the target protein, ensuring its binding. This selective binding then triggers a measurable signal, indicating the presence and concentration of the target molecule. This principle is applied in diagnostic tests for diseases, environmental monitoring for pollutants, and in research laboratories for understanding biological processes. The precise control over molecular recognition, driven in part by dipole-dipole forces, is what enables the high specificity and sensitivity of modern biosensing technologies.
Conclusion: The Enduring Significance of Intermolecular Forces in Tech
In conclusion, while “dipole-dipole interaction” might sound like a term confined to academic chemistry labs, its influence permeates the technological landscape in profound and often unseen ways. From the vibrant displays on our screens to the advanced materials that form the backbone of modern engineering, and the precise sensing mechanisms that drive scientific discovery, these seemingly subtle molecular forces are critical enablers of innovation.
The ability to understand, predict, and engineer dipole-dipole interactions allows technologists to tailor material properties, optimize chemical processes, and create sophisticated devices with unprecedented capabilities. As we push the boundaries of what’s possible in areas like nanotechnology, materials science, and biotechnology, a deep appreciation for these fundamental intermolecular forces will only become more crucial. They are the silent architects behind many of the conveniences and advancements that define our technological age, and their study continues to unlock new frontiers for future innovation.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.