In the rapidly evolving landscape of technology, we often focus on the “macro” achievements: the speed of a processor, the resolution of an OLED screen, or the capacity of a solid-state drive. However, as we push the boundaries of Moore’s Law and venture into the realms of nanotechnology and quantum computing, the focus is shifting toward the “micro.” At the heart of these advancements lies a fundamental physical interaction known as the dipole-dipole force.
While traditionally a concept relegated to chemistry textbooks, the dipole-dipole force has become a cornerstone of modern material science and technological engineering. Understanding these forces is no longer just for academics; it is essential for the engineers and tech visionaries designing the hardware of tomorrow. From the way your smartphone screen displays color to the development of high-density memory storage, dipole-dipole interactions are the invisible threads holding our digital world together.

Understanding the Mechanism: The Tech Behind the Attraction
To understand why tech giants are investing billions into molecular engineering, one must first grasp the technical mechanism of the dipole-dipole force. In the world of technology, efficiency is dictated by how well we can manipulate matter at the atomic level.
Polar Molecules and Charge Distribution
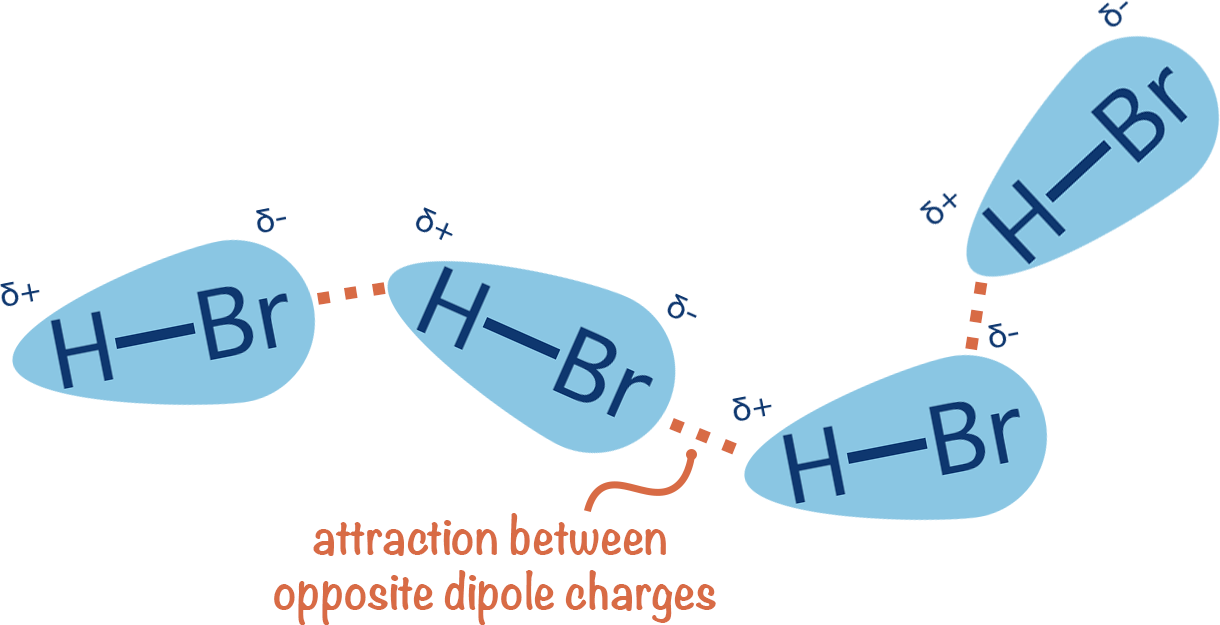
At its core, a dipole-dipole force is an intermolecular attraction between two polar molecules. In tech manufacturing—specifically in the creation of polymers and semiconductors—engineers look for molecules where electrons are not shared equally. This creates a “dipole moment,” where one end of the molecule is slightly negative and the other is slightly positive. In a technological context, these molecules act like tiny, microscopic magnets. When these molecules are aligned correctly in a material, they create a stable structure that can be used to conduct signals or insulate sensitive components.
The Physics of Electrostatic Interaction
The force itself is an electrostatic interaction. In high-end hardware design, controlling this interaction is vital. The positive end of one polar molecule is attracted to the negative end of its neighbor. While weaker than ionic or covalent bonds, these forces are cumulative. In the production of advanced lubricants for hard drives or the development of specialized coatings for aerospace tech, the strength and orientation of these dipole-dipole forces determine the durability and heat resistance of the final product.
Dipole-Dipole Forces in Modern Hardware and Material Science
The application of dipole-dipole forces is most visible in the materials used to build our daily devices. As we demand thinner, faster, and more energy-efficient gadgets, material science must find ways to utilize these molecular “magnets” to achieve performance benchmarks.
Liquid Crystal Displays (LCDs) and Screen Tech
One of the most ubiquitous applications of dipole-dipole forces in technology is the Liquid Crystal Display (LCD). Liquid crystals are unique because they possess the properties of both liquids and solids. The molecules used in these displays are highly polar.
When you look at your smartphone or laptop, the device uses an electric field to manipulate the dipole-dipole interactions between these liquid crystal molecules. By applying a voltage, the tech forces the dipoles to align or rotate, which in turn changes how light passes through them. This precise control over molecular orientation—driven entirely by dipole mechanics—is what allows for the sharp images and vibrant colors we see on our screens today.
Nanotechnology and Semiconductor Design
As semiconductors shrink to the 3nm and 2nm scales, the “bulk” properties of materials begin to fail. Here, dipole-dipole forces become a primary concern for tech engineers. At this scale, the attraction between molecules can cause “stiction” (static friction at the nano-scale), which can ruin a microchip.
Conversely, engineers use these forces to their advantage in “bottom-up” manufacturing. By designing molecules that naturally assemble themselves through dipole-dipole attraction, tech companies are experimenting with self-assembling circuits. This could revolutionize the tech industry by reducing the need for expensive photolithography machines and allowing for even denser, more powerful processors.
Thermal Management in High-Performance Computing
Heat is the enemy of performance. In the world of high-performance computing (HPC) and gaming rigs, managing thermal energy is a constant battle. Dipole-dipole forces play a massive role in the development of advanced thermal interface materials (TIMs). These are the pastes and pads that sit between a CPU and its cooler. By utilizing polar molecules that maintain strong dipole-dipole interactions, scientists have created thermal pastes that can better bridge the microscopic gaps between surfaces, ensuring that heat is moved away from the silicon as efficiently as possible.
The Role of Molecular Forces in AI and Quantum Computing
As we move toward the era of Artificial Intelligence and Quantum Computing, the demand for “smart materials” is skyrocketing. These technologies require hardware that can store more data in less space and process information at speeds that exceed traditional binary logic.
Optimizing Memory Storage at the Atomic Level
Traditional hard drives and even modern NVMe SSDs are reaching physical limits. The next frontier in tech storage is molecular or atomic memory. Tech researchers are currently investigating “Molecular Spin Cross-Over” materials, where dipole-dipole interactions can be used to switch a molecule’s state (from 0 to 1) using very little energy.
Because dipole-dipole forces are sensitive to external electric fields, they provide a mechanism for high-speed, low-power data writing. If perfected, this could lead to storage devices that are 1,000 times denser than current technology, powered by the subtle “push and pull” of molecular dipoles.
Quantum Dot Technology
Quantum dots are a significant trend in the tech industry, particularly in high-end TV displays (QLED) and medical imaging sensors. These are tiny semiconductor particles only a few nanometers in size. The way these dots interact with their environment and each other is largely governed by dipole-dipole interactions (specifically, a subset known as Förster Resonance Energy Transfer or FRET).
In the tech sector, mastering these forces allows for the creation of displays with nearly 100% color accuracy and the development of solar cells that can capture more energy from the sun by transferring “excitons” through dipole interactions before they are lost as heat.
Future Trends: Molecular Engineering and Bio-Tech Integration
The future of tech isn’t just in silicon; it’s in the convergence of biology and digital hardware. The dipole-dipole force is the primary language of biological molecules like DNA and proteins. As we look toward the next decade, “Bio-Tech” will likely be the niche where dipole-dipole force manipulation yields the most disruptive results.
Advanced Sensors and Wearables
The next generation of tech wearables will move beyond simple heart rate monitors. We are looking at “lab-on-a-chip” devices that can monitor glucose levels, cortisol, and other biomarkers in real-time. These sensors often rely on “molecular recognition.”
A sensor is coated with a specific polar molecule designed to attract a target molecule (like glucose) through—you guessed it—dipole-dipole forces. When the target molecule binds to the sensor via these electrostatic attractions, it changes the electrical conductivity of the sensor, sending a signal to your smartwatch. This is a perfect example of how a fundamental chemical force is being translated into a high-tech digital insight.
Sustainable Hardware and “Green” Tech
The tech industry is under increasing pressure to become sustainable. This has led to a surge in research into biodegradable electronics. By utilizing organic molecules that are held together by dipole-dipole forces rather than permanent covalent bonds, companies are beginning to develop temporary sensors and components.
These devices perform their technological function and then, when exposed to a specific trigger (like a change in pH or temperature), the dipole-dipole bonds are disrupted, allowing the device to break down safely into the environment. This represents a massive shift in how we think about the lifecycle of tech gadgets.
Conclusion: Why the Dipole-Dipole Force is the Future of Tech
While the term “dipole-dipole force” might seem better suited for a laboratory than a tech keynote, its importance in the digital age cannot be overstated. We have reached a point in technological history where we can no longer ignore the molecular foundations of our hardware.
As we seek to make AI more powerful, our screens more vivid, and our gadgets more sustainable, we are increasingly relying on the precise manipulation of these intermolecular attractions. The tech professionals, developers, and enthusiasts who understand these forces will have a clearer vision of where the industry is heading.
The transition from traditional mechanical engineering to molecular-driven technology is well underway. The dipole-dipole force is not just a scientific curiosity; it is the invisible architecture of the next technological revolution. Whether it’s through the alignment of liquid crystals in your hand or the self-assembly of a future quantum processor, these molecular magnets are the forces that will define the next century of innovation.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.