In the vast and intricate world of medicine, precision in language is not merely a nicety; it is a fundamental pillar of patient safety, treatment efficacy, and ethical practice. Among the myriad terms that form the lexicon of healthcare, “indication” stands out as profoundly significant. Far from being a mere synonym for “reason,” in medical terms, an indication is a specific and scientifically validated reason for using a particular medical treatment, drug, procedure, or diagnostic test. It defines why a certain intervention is appropriate for a patient’s condition, grounding every clinical decision in evidence and established protocols.
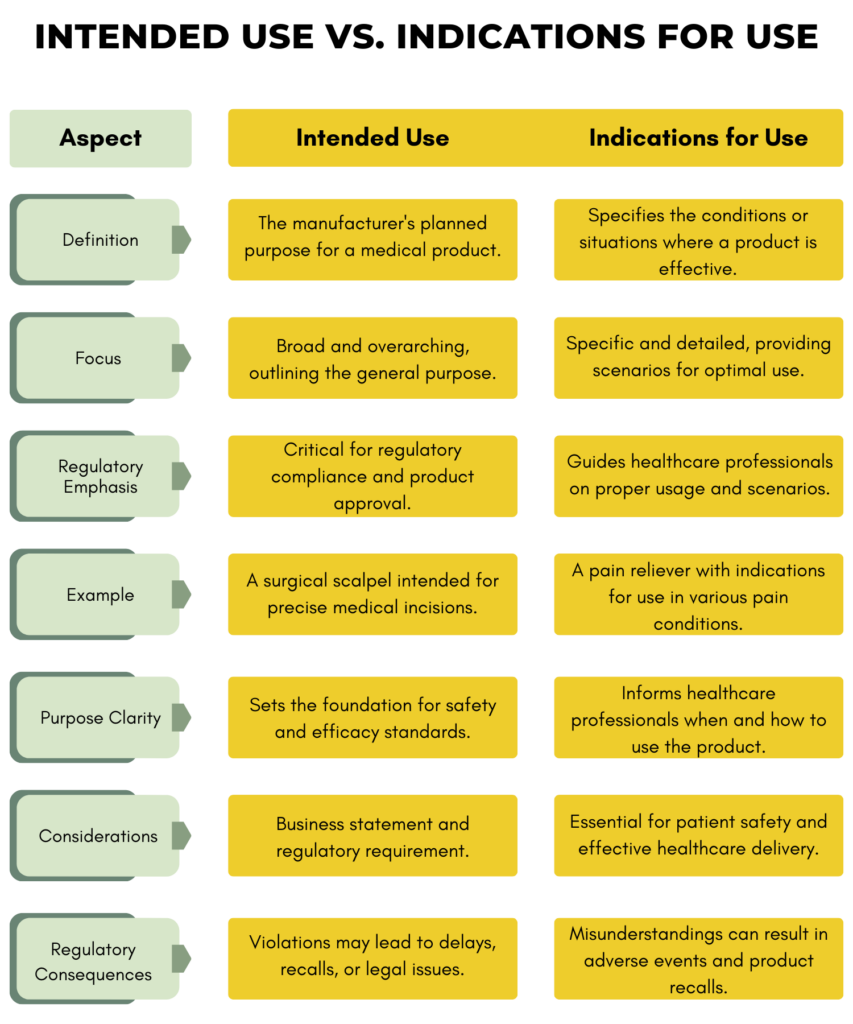
Understanding the concept of indication goes far beyond a simple dictionary definition. It underpins the entire healthcare ecosystem, influencing how new technologies are developed, how pharmaceutical brands are built and perceived, and how massive financial investments are made in research, development, and patient care. As we delve into what indication truly means, we will explore its critical role from foundational medical practice to its pervasive impact across the domains of technology, branding, and finance, revealing how this seemingly straightforward term is, in fact, a linchpin of modern healthcare innovation and economics.
The Foundation of Medical Decision-Making: Defining “Indication”
At its core, an indication is the bedrock upon which medical practitioners build their treatment plans. It is the demonstrable evidence that a specific therapy or intervention is likely to benefit a patient suffering from a particular condition. Without clear indications, medical practice would devolve into guesswork, jeopardizing patient well-being and undermining the scientific principles of healthcare.
The Literal Definition: Why Do We Use This Treatment?
An indication, in its most straightforward sense, is a valid reason to administer a specific medical treatment, intervention, or drug. For example, an “indication” for antibiotics is a bacterial infection, not a viral one. Insulin’s indication is diabetes mellitus. A surgical appendectomy’s indication is appendicitis. These are not arbitrary choices but are based on extensive research, clinical trials, and regulatory approval processes that confirm the treatment’s safety and effectiveness for that specific condition.
It’s crucial to differentiate “indication” from “contraindication.” While an indication tells us why to use a treatment, a contraindication tells us why not to use it. For instance, a severe allergy to penicillin is a contraindication for its use, even if the patient has a bacterial infection that penicillin would otherwise treat. Understanding both sides of this coin is vital for optimal patient care and avoiding harm.
Beyond the Basics: Primary, Secondary, and Off-Label Indications
The landscape of indications is often more complex than a single reason for a single drug. Treatments can have multiple layers of approved uses:
- Primary Indication: This is the main, most common, and often the first condition for which a drug or treatment receives approval. For example, the primary indication for metformin is Type 2 Diabetes.
- Secondary Indications: Many drugs, after their initial approval, are found through further research or clinical observation to be effective for other conditions. These become secondary indications, often requiring additional clinical trials and regulatory approval. For instance, some antidepressants might also have an approved secondary indication for generalized anxiety disorder. This expansion of indications can significantly broaden a drug’s utility and market reach.
- Off-Label Indications: This category presents a unique intersection of clinical judgment, regulatory frameworks, and ethical considerations. “Off-label” use refers to prescribing a medication for a condition, dosage, or route of administration that has not been specifically approved by regulatory bodies (like the FDA in the US). While it might sound concerning, off-label use is legal and widespread. Physicians can prescribe drugs off-label if, based on their clinical judgment and available scientific evidence (even if not yet formalized into an approved indication), they believe it is in the best interest of the patient. Common scenarios include using a drug for a rare disease where no approved treatment exists, or for pediatric populations where specific pediatric trials are often lacking. However, off-label use comes with increased responsibilities for the prescriber, and insurance reimbursement can be challenging, highlighting financial implications.
The Role of Indication in the Medical Ecosystem: Where Tech, Brand, and Money Intersect
The concept of “indication” is not confined to the clinician’s office. It reverberates throughout the entire healthcare ecosystem, serving as a critical nexus where technological innovation, brand strategy, and economic considerations converge.
Technology’s Influence on Identifying and Managing Indications
Technology plays an increasingly pivotal role in refining our understanding and management of medical indications, ushering in an era of greater precision and efficiency.
- Advanced Diagnostics & AI: The ability to accurately identify an indication is often dependent on sophisticated diagnostic tools. From high-resolution MRI scans and advanced laboratory tests to genomic sequencing and liquid biopsies, technology allows for ever-more precise identification of disease states and their underlying mechanisms. Artificial intelligence (AI) is revolutionizing this further, with algorithms capable of analyzing vast datasets from medical images, patient histories, and genetic profiles to identify subtle patterns indicative of specific conditions, often with greater speed and accuracy than human interpretation alone. AI tools can also predict a patient’s likelihood of responding to a particular treatment, essentially refining the “indication” to a personalized level. Electronic Health Records (EHRs), while foundational, provide the structured data necessary for these AI advancements.
- Drug Discovery & Development: The process of discovering new drugs and identifying new indications for existing ones is fundamentally driven by technological advancements. Bioinformatics and computational modeling allow researchers to screen millions of compounds virtually, predicting their potential therapeutic effects against specific disease targets. Big data analytics help identify patient cohorts that respond best to certain treatments, essentially uncovering more nuanced indications. Precision medicine, heavily reliant on genomics and proteomics, aims to tailor treatments based on an individual’s unique biological makeup, leading to ultra-specific indications where a drug might be indicated not just for “cancer,” but for “HER2-positive breast cancer with a specific genetic mutation.”
- Telemedicine & Remote Monitoring: Technology extends beyond discovery and diagnosis to ongoing patient management. Telemedicine platforms enable remote consultations, allowing healthcare providers to assess symptoms and monitor conditions, thereby identifying or confirming indications without the need for an in-person visit. Wearable devices and Internet of Things (IoT) sensors in healthcare collect real-time physiological data (heart rate, glucose levels, activity), providing continuous insights that can signal worsening conditions, confirm treatment efficacy, or even suggest new indications based on longitudinal data analysis. These tools not only enhance patient convenience but also generate invaluable data for understanding how treatments perform in real-world settings.

Branding and Reputation: Trust, Efficacy, and Market Positioning
For pharmaceutical companies, healthcare providers, and even individual medical specialists, the clarity and validity of indications are central to building brand trust and establishing market position.
- Pharmaceutical Branding: The approval of an indication by regulatory bodies like the FDA or EMA is a powerful endorsement of a drug’s efficacy and safety. This approval becomes a core component of a drug’s brand identity. A pharmaceutical company invests billions in clinical trials to prove its drug’s indications, knowing that these approvals will define its market, its marketing claims, and its perceived value. A drug with clear, compelling indications, backed by robust clinical evidence, commands trust among physicians and patients. Brand reputation is inextricably linked to the proven efficacy for its indications. Ethical marketing around approved indications is critical for maintaining this trust, while any deviation can lead to significant reputational and financial damage.
- Healthcare Provider Branding: Hospitals, clinics, and even individual physicians often build their brand around specific areas of expertise, which are essentially specialized indications. A hospital branded as a leading “cancer center” positions itself as having superior capabilities and outcomes for various oncology indications. A cardiologist specializes in indications related to heart disease. This specialization is a brand strategy, communicating expertise and attracting patients seeking care for those specific conditions. The reputation of these institutions hinges on their success in treating patients for these indicated conditions, reinforcing the link between medical efficacy and brand perception.
- Patient Education & Communication: For patients, understanding the indication for their medication or procedure is vital for adherence, engagement, and peace of mind. Pharmaceutical companies and healthcare providers invest in clear, accessible communication about indications to empower patients. This commitment to transparency and health literacy builds confidence in the treatment and, by extension, in the prescribing physician and the drug’s brand. Clear communication about indications helps manage patient expectations and fosters a collaborative approach to healthcare.
The Economics of Indications: Investment, Reimbursement, and Access
The financial implications surrounding medical indications are profound, influencing investment decisions, healthcare spending, and patient access to life-saving treatments.
- Drug Development Costs & Return on Investment (ROI): Bringing a new drug to market, or even securing a new indication for an existing drug, is an astronomically expensive undertaking, often costing billions of dollars. This massive investment is driven by the rigorous requirement to prove safety and efficacy for specific indications through extensive clinical trials. The clearer and more impactful the indication, the greater the potential market, and thus the higher the potential for return on investment (ROI) for pharmaceutical companies and their investors. Patent protection, which grants exclusivity for approved indications, is a critical mechanism for allowing companies to recoup these R&D costs and incentivize further innovation.
- Reimbursement Policies: Insurance companies and government healthcare systems heavily rely on approved indications when making reimbursement decisions. Generally, if a drug or procedure is prescribed for an approved indication, it is more likely to be covered. Off-label use, while medically permissible, often faces hurdles in reimbursement, leading to out-of-pocket costs for patients or administrative burdens for providers. This economic reality significantly influences prescribing patterns and patient access. Healthcare economics departments rigorously analyze the cost-effectiveness of treatments for their indications to inform policy and manage budgets.
- Market Access & Equity: The economic value placed on a drug’s indications directly impacts market access globally. For rare diseases, where the indication population is small, the financial model can be challenging, sometimes leading to “orphan drug” designations and incentives to encourage development. In contrast, blockbuster drugs with broad indications represent huge markets. Disparities in healthcare spending between nations, and even within regions, mean that access to drugs for specific indications can be uneven, highlighting issues of global health equity and the economic factors that dictate who receives what treatment, when, and where.
Challenges and Future Directions: Navigating the Evolving Landscape
The critical role of “indication” continues to evolve, presenting both challenges and exciting opportunities for the future of medicine.
Evolving Regulatory Frameworks and Ethical Considerations
As technology accelerates drug discovery and diagnostic capabilities, regulatory bodies face the complex task of adapting. How do we quickly validate new indications for AI-driven therapies or gene-editing techniques while maintaining stringent safety standards? Ethical dilemmas surrounding off-label use persist, particularly concerning informed consent and equitable access when an unapproved treatment is the only option. The balance between fostering innovation and protecting public health remains a constant tension, requiring flexible yet robust regulatory frameworks. From a brand perspective, companies must navigate increasingly complex global regulatory environments, ensuring their marketing claims align precisely with approved indications to maintain trust and avoid legal repercussions.
The Promise of Personalized Medicine and Precision Health
The future of medical indications is undeniably headed towards hyper-personalization. Driven by advances in genomics, proteomics, and AI, medicine will increasingly identify indications at an individual level. Treatments will be tailored not just for a disease, but for your disease, based on your unique genetic profile, lifestyle, and environmental factors. This means indications will become incredibly precise, targeting specific biomarkers or genetic mutations rather than broad disease categories. For example, a drug might be indicated for “patients with XYZ mutation who also exhibit ABC resistance profile.” This shift promises unprecedented efficacy and reduced side effects but also poses significant challenges for drug development, regulatory approval, and, crucially, the economic models for highly individualized therapies.

Conclusion
The term “indication” in medical language is far more than a technical definition; it is the cornerstone of responsible, evidence-based healthcare. It dictates when, why, and for whom medical interventions are appropriate, acting as a critical filter that ensures patient safety and treatment efficacy. From the foundational decision-making at the bedside to the sprawling complexities of the healthcare industry, the understanding and application of indications are paramount.
Its influence stretches across the three key domains we explored:
- Technology continuously refines our ability to identify, diagnose, and manage indications with unparalleled precision, driving innovation from AI diagnostics to personalized medicine.
- Branding hinges on the clear communication and proven efficacy of indications, building trust for pharmaceutical products and healthcare providers alike.
- Money dictates the massive investments in proving indications, influences reimbursement policies, and ultimately shapes equitable access to life-saving treatments globally.
As medicine continues its rapid evolution, driven by technological breakthroughs and a deeper understanding of human biology, the concept of “indication” will remain central. It is the compass guiding us towards a future where treatments are ever more targeted, effective, and ethically administered, ensuring that every medical decision is rooted in sound scientific reasoning and the best interests of the patient.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.