In the realm of medical technology, few innovations have had as profound an impact on daily life as the home pregnancy test. What was once a complex laboratory procedure requiring days of processing has been miniaturized into a hand-held diagnostic tool that delivers results in minutes. However, as with any piece of hardware or biochemical sensor, the technology is not infallible. When we discuss a “false positive,” we are essentially discussing a failure in the diagnostic interface—a moment where the biochemical “software” or the mechanical “hardware” of the test misinterprets biological data.
In the contemporary tech landscape, the pregnancy test has evolved from simple paper strips to sophisticated digital devices integrated with Bluetooth and mobile applications. Understanding what causes a false positive requires a deep dive into the engineering of these biosensors and the variables that can interfere with their data processing.
The Mechanics of Detection: How Bio-Tech Sensors Work
To understand why a technological system fails, one must first understand how it is designed to succeed. Modern pregnancy tests are essentially lateral flow immunoassays—a specialized form of biotechnology that uses antibodies to detect the presence of Human Chorionic Gonadotropin (hCG).
Lateral Flow Immunoassays: The Analog Foundation
The “analog” version of this technology relies on a multi-stage fluidic pathway. When a sample is introduced, it travels via capillary action across a strip of nitrocellulose membrane. This membrane is embedded with mobile antibodies tagged with a dye (usually gold nanoparticles or colored latex). If hCG is present, these antibodies bind to it, and the complex is then trapped by a second set of stationary antibodies on the “test line.”
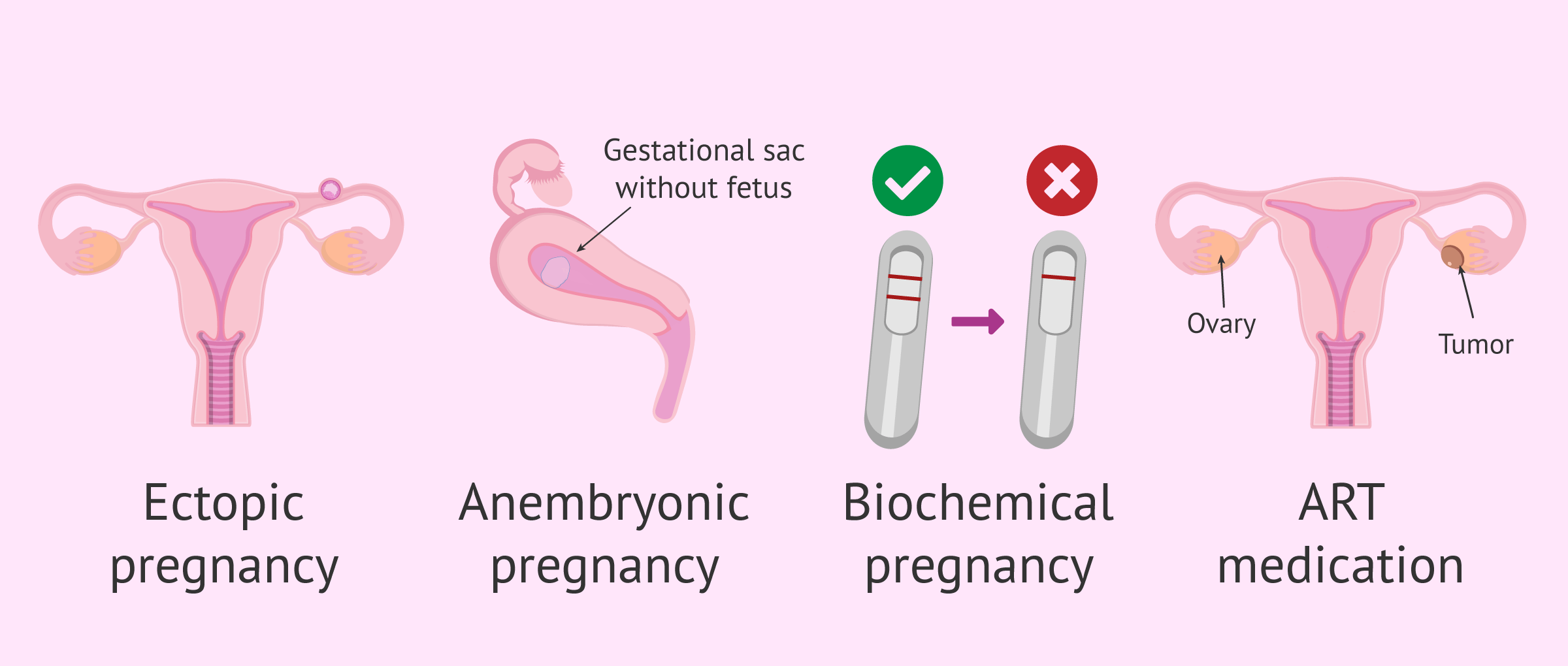
A false positive in this analog tech often stems from a “read error.” For instance, “evaporation lines” are a common mechanical byproduct. As the urine evaporates from the nitrocellulose strip, the structural integrity of the membrane changes, sometimes leaving a faint shadow where the antibodies are concentrated. To the human eye, this looks like a positive result, but in technical terms, it is a visual artifact—a ghost in the machine caused by the physical degradation of the testing medium.
Digital Transducers: Translating Chemistry into Data
Digital pregnancy tests represent a significant leap in consumer medical hardware. These devices do not rely on the human eye to interpret a faint line; instead, they use optical sensors or “transducers.” Inside the plastic casing of a digital test sits a small microprocessor and a light-emitting diode (LED) paired with a photo-detector.
The sensor measures the intensity of the color change on the internal wick. If the light reflection hits a specific threshold programmed into the device’s firmware, the LCD screen displays “Pregnant.” A false positive here is often an algorithmic error. If there is a “signal noise”—perhaps due to a minor chemical fluctuation or a hardware glitch in the optical sensor—the microprocessor may incorrectly categorize the data. Unlike analog tests, where a user might see a faint, ambiguous line, a digital test forces a binary output (Yes/No), which can occasionally mask a “low-confidence” sensor reading.
Chemical and Digital Interference: Why the Tech Fails
In any data-driven system, the quality of the output is only as good as the purity of the input. In the context of pregnancy diagnostics, several factors can introduce “noise” into the system, leading the biosensors to trigger a false positive.
Medication and System Input Errors
The most common cause of a technologically accurate but clinically “false” positive is the presence of exogenous hormones. Patients undergoing fertility treatments often receive “trigger shots” containing synthetic hCG (such as Ovidrel). Because the diagnostic kit is engineered to detect hCG molecules regardless of their source, the sensor is technically functioning perfectly—it detects the molecule—but the context of the data is wrong.
From a systems-engineering perspective, this is a classic input error. The hardware is programmed to recognize “X,” and when “X” is introduced via medication rather than pregnancy, the system triggers. Other medications, such as certain anti-seizure drugs or anti-anxiety medications, can occasionally interfere with the chemical binding process, causing the “test” antibodies to clump together even in the absence of the target hormone, effectively “hacking” the sensor.
Software Lag and Interpretive Algorithms
In the latest generation of “Smart” tests, the data is often sent via Bluetooth to a smartphone application. These apps use proprietary algorithms to track cycles and predict outcomes. A false positive can occur if the software’s interpretive logic is flawed. For example, if an app is programmed to “weight” a faint digital signal more heavily based on the user’s previously logged menstrual data, it might present a high-confidence “Positive” notification. If the underlying biochemical sensor was actually reporting a marginal, low-confidence signal (perhaps due to a chemical imbalance or a faulty batch of reactive dye), the software’s “predictive” layer can create a false sense of certainty.

The Role of Data and AI in Reproductive Health Technology
As we move toward “Health 4.0,” the integration of Artificial Intelligence (AI) and Big Data into pregnancy diagnostics is changing how we view false positives. We are seeing a shift from “reactive” hardware (tests that just react to a sample) to “proactive” systems.
Smart Tests and Bluetooth Integration
The new frontier of pregnancy tech involves devices that sync with an ecosystem of wearable data. These devices don’t just look for hCG; they correlate that data with basal body temperature, heart rate variability, and sleep patterns collected from smartwatches.
The risk of a false positive in these integrated systems is often found in the “data fusion” process. If a user has a viral infection that raises their body temperature and heart rate, an AI-driven fertility app might misinterpret these biological telemetry signals as signs of early pregnancy. When the user then takes a physical test that happens to have a faint evaporation line, the “Confirmation Bias” of the AI system can lead to a documented false positive in the user’s digital health record.
Predictive Analytics vs. Biochemical Reality
Tech companies are increasingly using machine learning to refine the sensitivity of their tests. By analyzing millions of anonymized data points from users worldwide, companies like Clearblue or Ava can calibrate their sensors to detect minute amounts of hCG—sometimes even before a missed period.
However, high sensitivity often comes at the cost of specificity. In the tech world, this is the “Sensitivity vs. Specificity” trade-off. By lowering the threshold for what the sensor considers a “Positive,” the hardware becomes more prone to “False Alarms.” In early pregnancy diagnostics, this may result in detecting “chemical pregnancies”—conceptions that never implant or progress. While technically a “positive” from a biochemical standpoint, from a clinical and user-experience perspective, it is often categorized as a false positive because it does not result in a viable pregnancy.
Improving Diagnostic Accuracy through Advanced Engineering
To combat the frequency of false positives, the diagnostic tech industry is turning to high-end engineering solutions that move beyond the simple nitrocellulose strip.
Nanotechnology in Hormone Detection
The next generation of biosensors is utilizing nanotechnology to increase “Signal-to-Noise” ratios. By using carbon nanotubes or graphene-based sensors, engineers can create diagnostic surfaces that only respond to the exact molecular weight and shape of hCG. These “nano-gates” are much less likely to be triggered by the “noise” of other hormones, such as Luteinizing Hormone (LH) or Follicle-Stimulating Hormone (FSH), which are structurally similar to hCG and can sometimes cause cross-reactivity in cheaper, lower-tech tests.
The Future of Wearable Diagnostic Tech
We are seeing the emergence of “Lab-on-a-Chip” (LoC) technology. These are microfluidic devices that can perform multiple laboratory functions on a single chip only millimeters in size. Future pregnancy diagnostics may not be “one-and-done” plastic sticks, but rather semi-permanent wearable patches that monitor hormone fluctuations in interstitial fluid over several days.
This longitudinal data collection would virtually eliminate the “false positive” caused by a single faulty reading or a momentary chemical spike. By analyzing the trend of the data rather than a single data point, the technology can provide a much higher “Confidence Score.” In this model, a positive result is only confirmed if the hCG signal follows a specific logarithmic growth curve programmed into the patch’s firmware.
Conclusion: The Interface of Biology and Hardware
The “false positive” is a fascinating intersection of human biology and technological limitation. Whether it is an evaporation line on an analog strip, a firmware glitch in a digital LCD test, or an algorithmic miscalculation in a fertility app, the failure usually lies in the interpretation of complex biological signals.
As we continue to digitize our health, the technology behind pregnancy tests will become more sophisticated, moving from simple chemical reactions to complex data-processing ecosystems. While no sensor is 100% accurate, the move toward nanotechnology and AI-driven longitudinal monitoring promises a future where the “false positive” is a relic of the analog past. For now, understanding that these devices are sophisticated pieces of biotech hardware helps users recognize that a result is not just a biological fact, but the output of a complex, and occasionally fallible, technological process.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.