In the relentless march of technological progress, certain scientific discoveries don’t just expand our understanding of the natural world; they fundamentally reshape our capabilities, paving the way for entirely new technological paradigms. Pluripotent cells represent one such monumental discovery. Far from being a mere biological curiosity, these remarkable cells are foundational to an explosion of innovation within the biotechnology sector, driving advancements in regenerative medicine, drug discovery, and personalized healthcare. Understanding what pluripotent cells are isn’t just about grasping a biological concept; it’s about recognizing a pivotal technological enabler that promises to redefine the future of health, research, and even our very interaction with human biology.
At their core, pluripotent cells possess the extraordinary ability to differentiate into almost any cell type in the body—from a nerve cell to a heart cell, a skin cell to a bone cell. This incredible versatility places them at the heart of what many consider to be the next major wave in health technology, offering solutions to challenges that have long seemed insurmountable. By delving into the essence of pluripotent cells, we uncover not just their biological marvel but their profound technological significance, revealing how they are powering a new generation of tools, therapies, and diagnostic platforms that are actively shaping the biotech landscape.
The Core Science: A Technological Breakthrough
The journey to harness pluripotent cells has been a testament to human ingenuity, transforming a complex biological phenomenon into a powerful technological asset. This exploration begins with defining their unique properties and understanding the landmark discovery that democratized their use for research and development.
Defining Pluripotency: Beyond Basic Biology
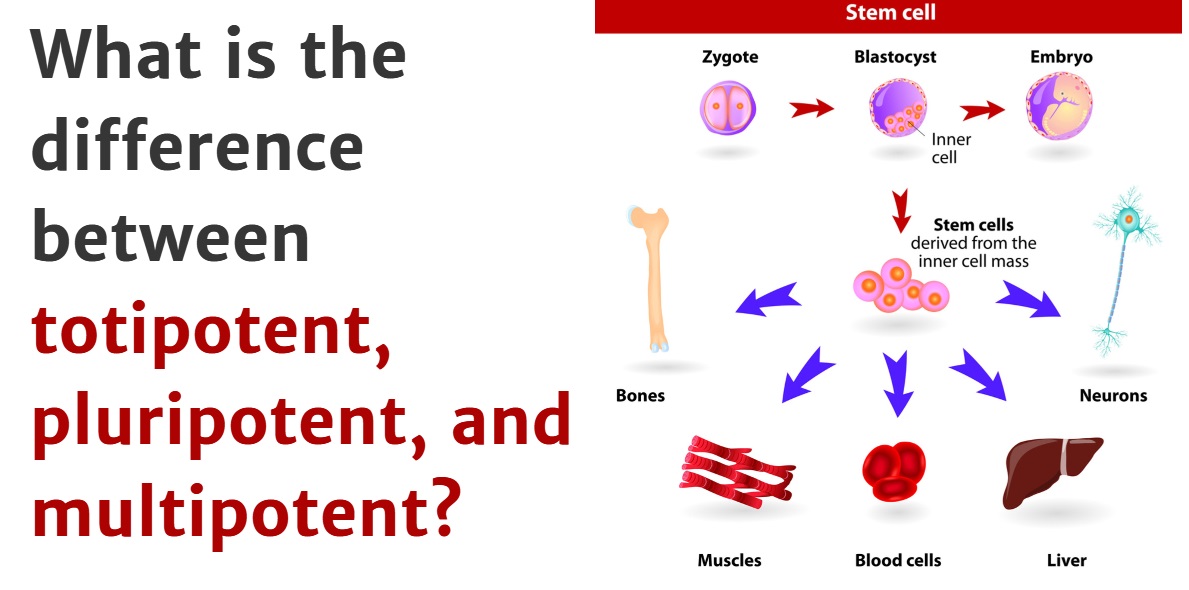
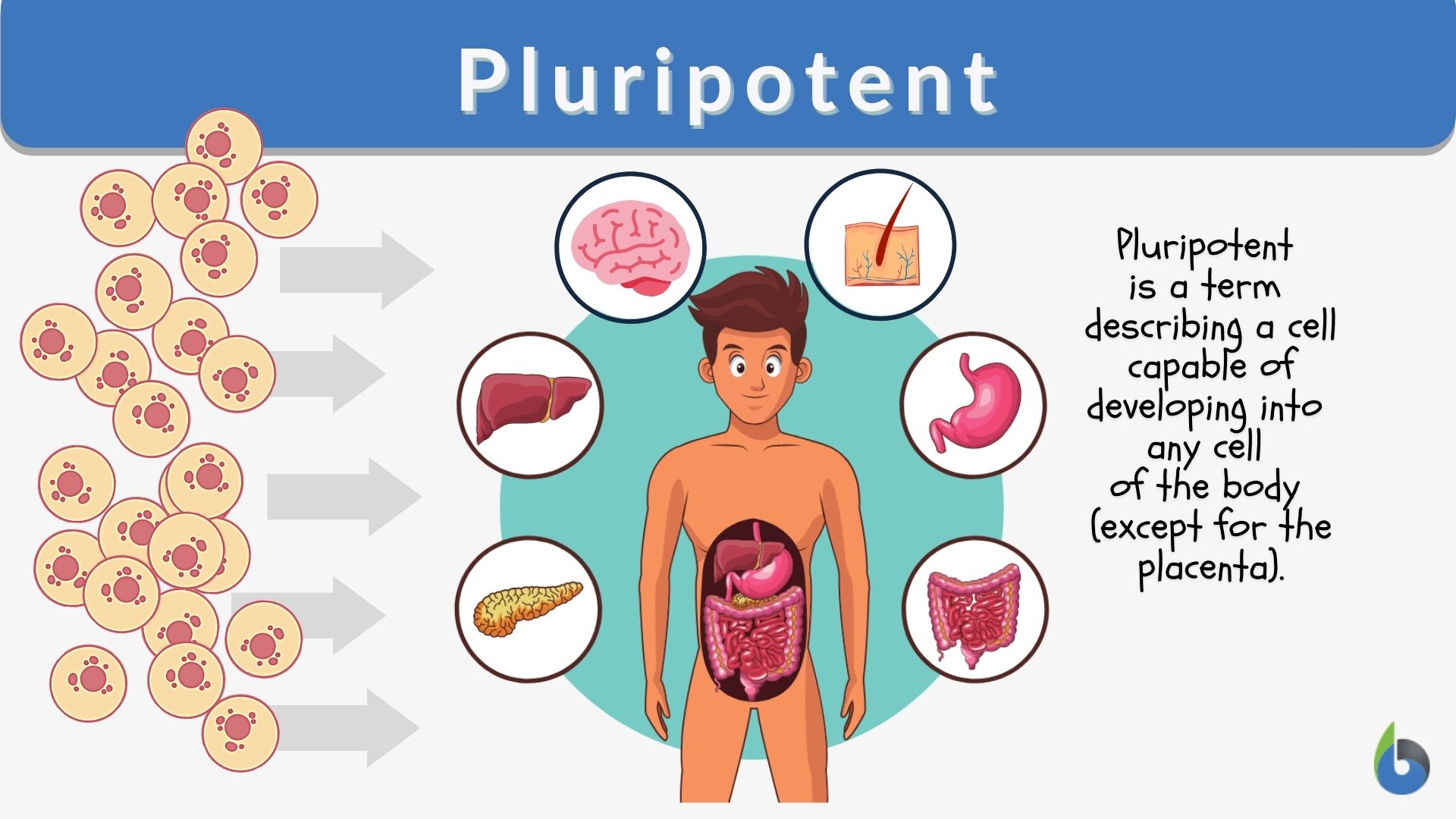
Pluripotency stands distinct from other cellular states. Unlike totipotent cells (like a zygote), which can form an entire organism including extra-embryonic tissues, pluripotent cells can form all cell types of the three germ layers (ectoderm, mesoderm, and endoderm) that make up an embryo, but cannot by themselves form a complete organism. This distinction is crucial because it gives them immense therapeutic potential without the ethical complexities associated with totipotency.
For biotechnologists, this intrinsic property is akin to having a universal cellular “building block.” Imagine a sophisticated 3D printer that can take a single type of raw material and, with the right programming, produce an almost endless variety of functional components. Pluripotent cells offer a similar promise at the biological level. Their consistent and predictable capacity for differentiation, when guided by precise molecular signals and cellular engineering techniques, transforms them into a versatile platform for generating specific tissues or organs in vitro, providing unprecedented control over biological systems. This controlled differentiation is a sophisticated technological process, requiring advanced media formulations, growth factors, and often automated cell culture systems to ensure purity and functionality. The ability to control cell fate is a hallmark of modern biotechnology, and pluripotent cells offer the ultimate canvas.
The Discovery of iPSCs: A Game-Changer in Cellular Engineering
While embryonic stem cells (ESCs) were the first type of pluripotent cells identified, their use raised significant ethical and immunological challenges. The true technological revolution began in 2006 with the groundbreaking work of Shinya Yamanaka, who discovered how to reprogram adult somatic cells (like skin cells) back into an embryonic-like pluripotent state. These cells are known as induced Pluripotent Stem Cells (iPSCs).
The creation of iPSCs was not just a biological discovery; it was a profound engineering feat. It demonstrated that cell identity is not immutable but can be reprogrammed using a specific set of genetic factors. This discovery circumvented many of the ethical concerns surrounding ESCs and, more importantly for technology, opened the door to generating patient-specific pluripotent cells. This means that a patient’s own cells can be used to create iPSCs, which can then be differentiated into various cell types for therapeutic purposes, virtually eliminating the risk of immune rejection. This personalized aspect is a cornerstone of future precision medicine and gene therapy technologies. The technical precision required to introduce and express the reprogramming factors, verify pluripotency, and then guide differentiation into a desired lineage involves sophisticated viral or non-viral gene delivery systems, advanced microscopy, flow cytometry, and genomic analysis tools—all key components of modern biotech.
Enabling Technologies: How Pluripotent Cells Drive Innovation
The true power of pluripotent cells lies in their ability to serve as a foundational technology, giving rise to entirely new fields and accelerating existing ones within the health tech sector.
Regenerative Medicine and Tissue Engineering: Building the Future of Health Tech
Perhaps the most intuitive application of pluripotent cells is in regenerative medicine. The ability to generate specific cell types on demand offers revolutionary solutions for repairing or replacing damaged tissues and organs. Imagine a future where heart attack victims could receive injections of lab-grown heart muscle cells, or individuals with spinal cord injuries could regain function through implanted neural cells. These aren’t distant dreams; they are active areas of research and technological development powered by pluripotent cells.
Tissue engineering, in particular, has been transformed. Biotechnologists are now working to grow complex tissues, and even rudimentary organs, in vitro using pluripotent cells as the starting material. This involves sophisticated scaffolding technologies (biomaterials), 3D bioprinting techniques to precisely arrange cells, and bioreactors that mimic the body’s physiological environment. Companies are investing heavily in automated systems for culturing and differentiating these cells at scale, moving from laboratory curiosities to potential industrial-level therapeutic production. The integration of robotics, AI-driven image analysis for quality control, and advanced sensor technologies within these bioreactors represents the cutting edge of health tech.
Disease Modeling and Drug Discovery: Accelerating Pharmaceutical R&D
Before iPSCs, studying human diseases often relied on animal models, which don’t always accurately reflect human physiology, or on limited post-mortem tissue samples. Pluripotent cells have revolutionized disease modeling by allowing scientists to create “disease in a dish.” By taking cells from a patient with a genetic disease, reprogramming them into iPSCs, and then differentiating them into the affected cell type (e.g., neurons for Parkinson’s disease, cardiomyocytes for heart conditions), researchers can study the disease mechanisms in a human cellular context.
This capability has a massive technological impact on drug discovery. Pharmaceutical companies can now use these patient-specific disease models to screen thousands of potential drug compounds in a highly relevant setting, identifying promising candidates more efficiently and accurately. This high-throughput screening involves advanced robotics, liquid handling systems, and sophisticated data analytics powered by machine learning to process vast amounts of experimental data. By reducing reliance on less predictive animal models and accelerating the identification of viable drug candidates, pluripotent cell technology is directly improving the efficiency and success rates of pharmaceutical research and development—a critical driver of innovation in medical technology.

Personalized Medicine: Tailoring Treatments with Cellular Tech
The concept of personalized medicine—treatments tailored to an individual’s unique genetic makeup and physiological responses—is no longer futuristic science fiction but a rapidly advancing technological reality, largely thanks to pluripotent cells. As mentioned, iPSCs derived from a patient can be used to create specific cell types that mirror their disease, enabling researchers to test the efficacy and toxicity of various drugs before administering them to the patient.
This “drug-on-a-chip” or “patient-in-a-dish” approach minimizes adverse drug reactions and maximizes therapeutic benefit. Imagine a cancer patient whose tumor cells are reprogrammed into iPSCs, then differentiated into specific cancer cells to test which chemotherapy agent is most effective for their particular cancer type. This level of precision is unprecedented. The underlying technology involves genomic sequencing, bioinformatics for genetic analysis, microfluidic devices for culturing cells in a controlled environment, and AI algorithms to predict drug responses. This integration of genomics, cellular biology, and advanced computational tools exemplifies the technological convergence driving personalized healthcare solutions.
The Technological Toolbox: Working with Pluripotent Cells
Effectively leveraging pluripotent cells requires an array of sophisticated technologies that push the boundaries of cellular manipulation, analysis, and production.
Gene Editing Technologies (CRISPR): Precision Cellular Modification
The advent of CRISPR-Cas9 and other gene editing technologies has formed a powerful synergy with pluripotent cell research. Gene editing allows scientists to precisely alter the DNA of pluripotent cells, correcting disease-causing mutations, inserting reporter genes for tracking cell fate, or even creating specific disease models by introducing mutations. This level of precise genetic engineering is critical for both research and therapeutic applications. For instance, a patient’s iPSCs could be genetically corrected ex vivo before differentiation and transplantation, offering a curative approach for genetic disorders. The technological finesse required for accurate gene delivery, off-target detection, and validation is immense, involving advanced molecular biology techniques, high-throughput sequencing, and computational tools for guide RNA design.
Advanced Imaging and AI: Visualizing and Analyzing Cellular Systems
Working with pluripotent cells necessitates meticulous monitoring and analysis. Advanced imaging techniques, including high-resolution microscopy, live-cell imaging, and 3D imaging, allow researchers to observe cell growth, differentiation, and morphology in real-time. These imaging platforms are increasingly integrated with artificial intelligence and machine learning algorithms. AI can automatically identify pluripotent colonies, assess their quality, track differentiation patterns, and even predict cell fate based on subtle morphological cues. This automation significantly enhances throughput, reduces human error, and extracts deeper insights from complex biological data, accelerating the discovery process and ensuring the quality of cell-based products.
Bioreactors and Automation: Scaling Production for Therapeutic Applications
Moving from laboratory-scale experiments to clinical-grade production of pluripotent cell-derived therapies requires industrial-level automation and control. Bioreactors are closed systems designed to grow and differentiate cells under precisely controlled conditions (temperature, pH, oxygen levels, nutrient supply). Modern bioreactors for pluripotent cells are highly sophisticated, often incorporating real-time sensors, automated nutrient feeding, and waste removal systems. Robotics are used for cell passaging, media changes, and quality control sampling, ensuring consistency and sterility. The development of scalable, GMP (Good Manufacturing Practice)-compliant bioreactor technology is a crucial bottleneck in bringing pluripotent cell therapies to market, demanding advanced engineering, process optimization, and digital control systems. These automated platforms are not just lab equipment; they are sophisticated biotechnological manufacturing plants.
Future Horizons: Pluripotent Cells at the Forefront of Biotech
The trajectory of pluripotent cell technology suggests an even more transformative future, with continued innovation at the intersection of biology and engineering.
Organoids and “Organs-on-a-Chip”: Next-Gen Research Platforms
Building on the success of 2D disease models, pluripotent cells are now being used to create 3D organoids—miniature, self-organized tissues that mimic the structure and function of actual organs (e.g., brain organoids, gut organoids). Even more advanced are “organs-on-a-chip” technologies, which combine microfluidics with engineered human tissues derived from pluripotent cells to create functional models of organs within a microchip. These systems can replicate complex physiological functions, including blood flow and mechanical stimuli. They offer unparalleled platforms for drug testing, toxicology studies, and understanding human development and disease, potentially revolutionizing preclinical research and reducing the need for animal testing. This convergence of microelectronics, biomaterials, and cellular biology epitomizes future health tech.
Ethical and Regulatory Tech Challenges
As with any powerful technology, pluripotent cells present ethical and regulatory challenges. Ensuring equitable access to these potentially life-saving therapies, addressing concerns about genetic manipulation, and establishing robust regulatory frameworks for their development and deployment are critical. The technology itself needs to be developed responsibly, with built-in safeguards and transparency. This involves not only scientific and medical expertise but also significant input from legal, ethical, and public policy domains to guide the responsible evolution of this transformative technology.
The Economic Impact: Fueling the Biotech Sector
The profound technological implications of pluripotent cells have not gone unnoticed by the financial world. The biotech sector is witnessing significant investment in companies leveraging these cells for therapeutic development, drug screening services, and research tools. Venture capital flows into startups focused on regenerative medicine, cellular agriculture, and advanced disease modeling. This economic activity underscores the recognition that pluripotent cells are not just a scientific curiosity but a key driver of innovation and growth in the global health technology market, promising new industries, job creation, and substantial returns on investment for those pioneering their applications.
In conclusion, pluripotent cells are far more than just a biological phenomenon; they are a cornerstone of modern biotechnology, an enabling technology that is fundamentally reshaping our approach to health and disease. From engineering new tissues for regenerative therapies to accelerating drug discovery and personalizing medicine, these remarkable cells, coupled with an ever-expanding array of supporting technologies, are unlocking capabilities that were once confined to the realm of science fiction. As research continues to advance and technological hurdles are overcome, pluripotent cells stand poised to drive the next wave of innovation in the tech sector, delivering transformative solutions that promise to enhance human health and longevity on an unprecedented scale.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.