The universe, in all its intricate complexity, is built upon fundamental building blocks. While we might marvel at towering skyscrapers, the vastness of the cosmos, or the subtle intricacies of artificial intelligence, at their very core, all matter is composed of atoms. And these atoms, in turn, are not indivisible entities but are themselves made up of even smaller, fundamental particles. Understanding these atomic constituents is not just an exercise in scientific curiosity; it has profound implications across the technological landscape, shapes the very brands we interact with, and underpins the financial systems that drive our economies.

For decades, the foundational understanding of atomic structure has centered on three primary subatomic particles: protons, neutrons, and electrons. While modern physics delves into a more complex zoo of even smaller, more exotic particles, the proton, neutron, and electron remain the cornerstones of elemental properties and chemical behavior, making them essential to grasp for anyone seeking a deeper understanding of the physical world.
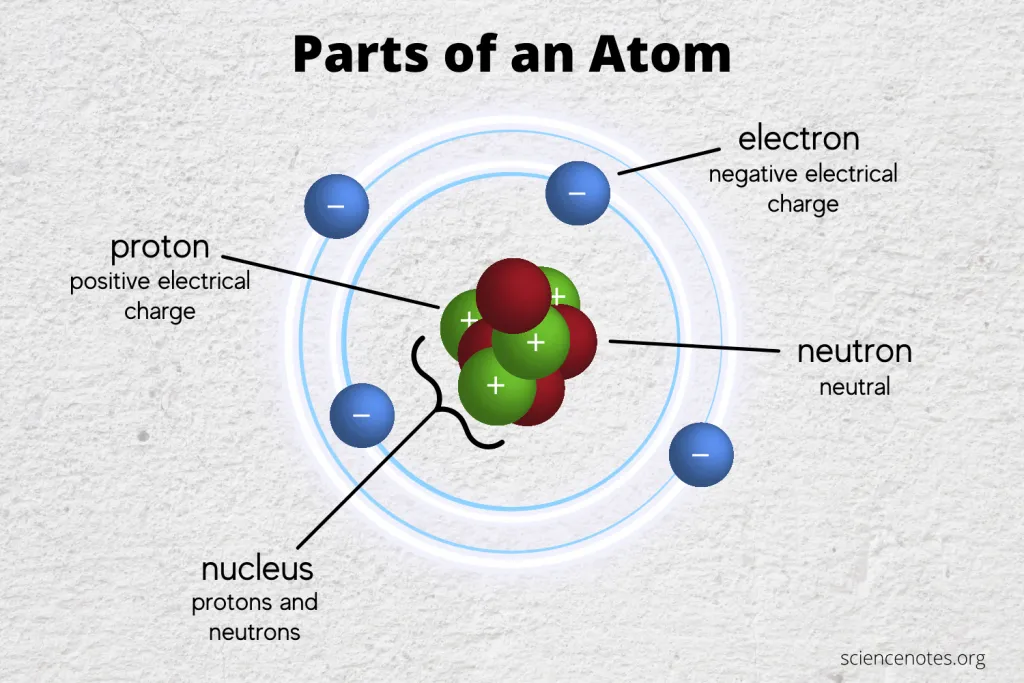
The Nucleus: The Atom’s Dense Core
At the heart of every atom lies the nucleus, a remarkably dense and positively charged region. This central hub is where the bulk of the atom’s mass is concentrated, and it’s composed of two of the three fundamental particles: protons and neutrons.
Protons: The Positive Identifiers
Protons are subatomic particles that carry a positive electrical charge. This charge is equal in magnitude but opposite in sign to the charge of an electron. Crucially, the number of protons in an atom’s nucleus is what defines the element. This is known as the atomic number. For instance, every atom with one proton is hydrogen, every atom with two protons is helium, and so on, up to the heaviest naturally occurring elements. This fundamental property is the basis of the periodic table, a cornerstone of chemistry and materials science, which in turn fuels innovation in technology.
The positive charge of protons plays a vital role in holding the atom together. It attracts the negatively charged electrons, preventing them from flying off into space. The electrostatic force between the protons and electrons is a key factor in chemical bonding, the process by which atoms combine to form molecules. These molecular structures are the basis of everything from the silicon chips that power our digital devices to the advanced polymers used in cutting-edge gadgets and the complex organic compounds that form the basis of new pharmaceuticals.
From a branding perspective, the intrinsic properties of elements, determined by their proton count, directly influence the materials used in product design and manufacturing. The conductivity of copper (defined by its 29 protons), the strength of titanium (22 protons), or the reactivity of lithium (3 protons) all contribute to a product’s performance, durability, and aesthetic appeal, ultimately impacting consumer perception and brand identity.
Neutrons: The Neutral Stabilizers
Neutrons, as their name suggests, are subatomic particles that carry no electrical charge; they are neutral. Like protons, neutrons reside within the nucleus. While the number of protons dictates the element, the number of neutrons can vary within atoms of the same element. These variations are called isotopes. For example, carbon-12, the most common isotope of carbon, has 6 protons and 6 neutrons, while carbon-14, famous for its use in radiocarbon dating, has 6 protons and 8 neutrons.
The primary role of neutrons in the nucleus is to act as a sort of “glue.” The strong nuclear force, a fundamental interaction in physics, binds protons and neutrons together, overcoming the electrostatic repulsion between the positively charged protons. Without neutrons, the nucleus of most elements would be unstable and would fly apart.
The existence of isotopes has significant implications. In the realm of technology, certain isotopes are crucial for nuclear power generation, medical imaging, and industrial processes. The precise control and understanding of isotopic properties are vital for developing new technologies and ensuring the safety and efficiency of existing ones. For instance, the development of nuclear fusion reactors, a frontier in energy technology, relies heavily on understanding the behavior of specific isotopes of hydrogen.
In the financial sector, the understanding and manipulation of materials with specific isotopic properties can lead to the development of specialized components for high-tech industries, which can then be leveraged for investment and market growth. The rarity and specific applications of certain isotopes can also influence their market value.
Electrons: The Orbiting Architects of Interaction
Orbiting the nucleus are the electrons, the third fundamental particle that makes up an atom. Electrons are significantly smaller and lighter than protons and neutrons, and they carry a negative electrical charge. This negative charge is exactly equal in magnitude to the positive charge of a proton.
The Electron Cloud and Chemical Behavior
Electrons do not orbit the nucleus in fixed paths like planets around a star. Instead, they exist in a region of probability around the nucleus, often visualized as an “electron cloud.” These clouds are organized into energy levels or shells. The arrangement of electrons, particularly those in the outermost shell (valence electrons), dictates an atom’s chemical properties. It is these valence electrons that participate in chemical reactions, forming bonds with other atoms to create molecules and compounds.
The way atoms interact through their electrons is the foundation of all chemistry. This understanding is critical for developing new materials with specific properties, from semiconductors that enable our digital world to advanced catalysts that drive industrial processes and new drugs that revolutionize healthcare. The digital security of our online transactions and the algorithms that power AI tools are all built upon the predictable behavior of electrons within matter.
In the context of branding and marketing, the materials used in products directly influence their perceived quality and functionality. A smartphone with a high-quality display, enabled by advanced semiconductor technology driven by electron behavior, conveys a message of premium quality. The tactile feel of a product, the way it conducts heat or resists corrosion, are all tangible manifestations of its atomic and molecular structure, heavily influenced by electron interactions.
The Significance of Electron Configuration for Technology and Brands
The specific arrangement of electrons in an atom’s shells determines its reactivity and its ability to form different types of chemical bonds (covalent, ionic, etc.). This electron configuration is what allows for the vast diversity of substances we see in the world.
- Semiconductors: The unique properties of silicon and germanium, elements with specific electron configurations, are the bedrock of the semiconductor industry. By carefully controlling the addition of impurities (doping), engineers can manipulate the flow of electrons to create transistors, diodes, and integrated circuits – the essential components of all modern electronics. This technological advancement has reshaped communication, entertainment, and virtually every aspect of our lives, directly impacting the brands that leverage these technologies.
- Advanced Materials: Understanding electron behavior allows scientists and engineers to design and synthesize new materials with tailor-made properties. This includes high-strength alloys for aerospace, supercapacitors for energy storage, and biocompatible materials for medical implants. These innovations are often the driving force behind new product categories and are central to a brand’s ability to differentiate itself in the market.
- Catalysis: Many industrial processes, from the production of plastics to the refining of fuels, rely on catalysts – substances that speed up chemical reactions without being consumed themselves. The effectiveness of these catalysts is intimately linked to the electron configurations of the atoms involved and their ability to interact with reacting molecules. This impacts efficiency and cost in manufacturing, ultimately affecting the pricing and accessibility of goods.
From a brand perspective, the performance and reliability of technology are directly tied to the underlying atomic and molecular structures. A brand that consistently delivers innovative and high-performing electronics, for instance, builds a reputation for quality and technological prowess. This reputation is built on the fundamental science of how electrons behave within the materials used to create those devices.
The Interplay of Particles: Shaping Our World
The simple yet profound existence of protons, neutrons, and electrons, and their specific arrangements within atoms, is the foundation upon which our entire physical reality is built. This understanding is not confined to academic laboratories; it has tangible and far-reaching implications across technology, branding, and finance.
Technological Advancements
The relentless pursuit of innovation in technology is, in many ways, a continuous exploration of atomic and subatomic behavior. From the development of quantum computing, which harnesses the peculiar properties of quantum mechanics at the subatomic level, to the design of more efficient batteries, the understanding of particle physics is paramount. Digital security relies on complex cryptographic algorithms, which are ultimately grounded in the mathematical principles that govern the interactions of these fundamental particles. AI tools, while appearing abstract, are powered by hardware whose very existence and functionality are dictated by the predictable yet controllable behavior of electrons.
Brand Identity and Perception
Brands are often built on the promise of performance, reliability, and innovation. These promises are intrinsically linked to the materials and technologies used in their products. A brand that pioneers a new lightweight alloy for a sports car is leveraging advancements in materials science, which in turn are based on an understanding of atomic structure. The aesthetic appeal of a product, its durability, and its user experience are all influenced by the chemical and physical properties of the materials, all originating from the interplay of protons, neutrons, and electrons. Consumers often associate certain brands with specific qualities – a reputation built on the tangible manifestations of underlying atomic principles.

Financial Implications
The economic impact of our understanding of atomic particles is immense. The development of new technologies, fueled by advancements in materials science and electronics, creates new industries and investment opportunities. The efficient production of essential goods relies on industrial processes optimized through chemical engineering, a field deeply rooted in atomic interactions. The value of rare earth elements, crucial for many advanced technologies, is directly tied to their unique atomic properties and their scarcity. Furthermore, the financial tools and services that enable global commerce are themselves often built on the digital infrastructure powered by technologies that owe their existence to our understanding of subatomic particles. The efficient allocation of capital towards research and development in areas like materials science and renewable energy can lead to significant economic growth and new financial markets.
In conclusion, while the title “what are three particles that make up an atom” might seem like a simple question from a basic science class, the answer – protons, neutrons, and electrons – unlocks a universe of understanding. This fundamental knowledge is not merely academic; it is the bedrock upon which our technological advancements are built, the silent architect of brand identity and consumer perception, and a crucial driver of economic prosperity. By comprehending these fundamental building blocks, we gain a deeper appreciation for the intricate workings of our world and the endless possibilities that lie ahead.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.