In the realm of modern technology, the intersection of chemistry and computational science has birthed a field that is revolutionizing how we understand the physical world. While the question “what is the bond order of $N_2$?” might seem like a simple relic of a high school chemistry textbook, in the context of advanced technology, it represents a fundamental benchmark for molecular modeling, simulation software, and the development of high-performance materials.
In the tech sector, specifically within material science and computational chemistry, the bond order of Nitrogen ($N2$)—which is numerically 3—serves as a critical data point. It informs the development of everything from aerospace alloys to the sustainable production of ammonia through digital twin simulations. To understand the bond order of $N2$ is to understand the stability and energy potential that drives some of the most sophisticated software algorithms in use today.

The Fundamentals of Molecular Modeling and Nitrogen Stability
To the software engineer or the material scientist, a molecule is more than a set of atoms; it is a complex system of electromagnetic interactions that can be modeled via code. Nitrogen ($N_2$) is the most abundant gas in our atmosphere, and its technological significance stems from its immense stability.
Defining Bond Order in a Technical Context
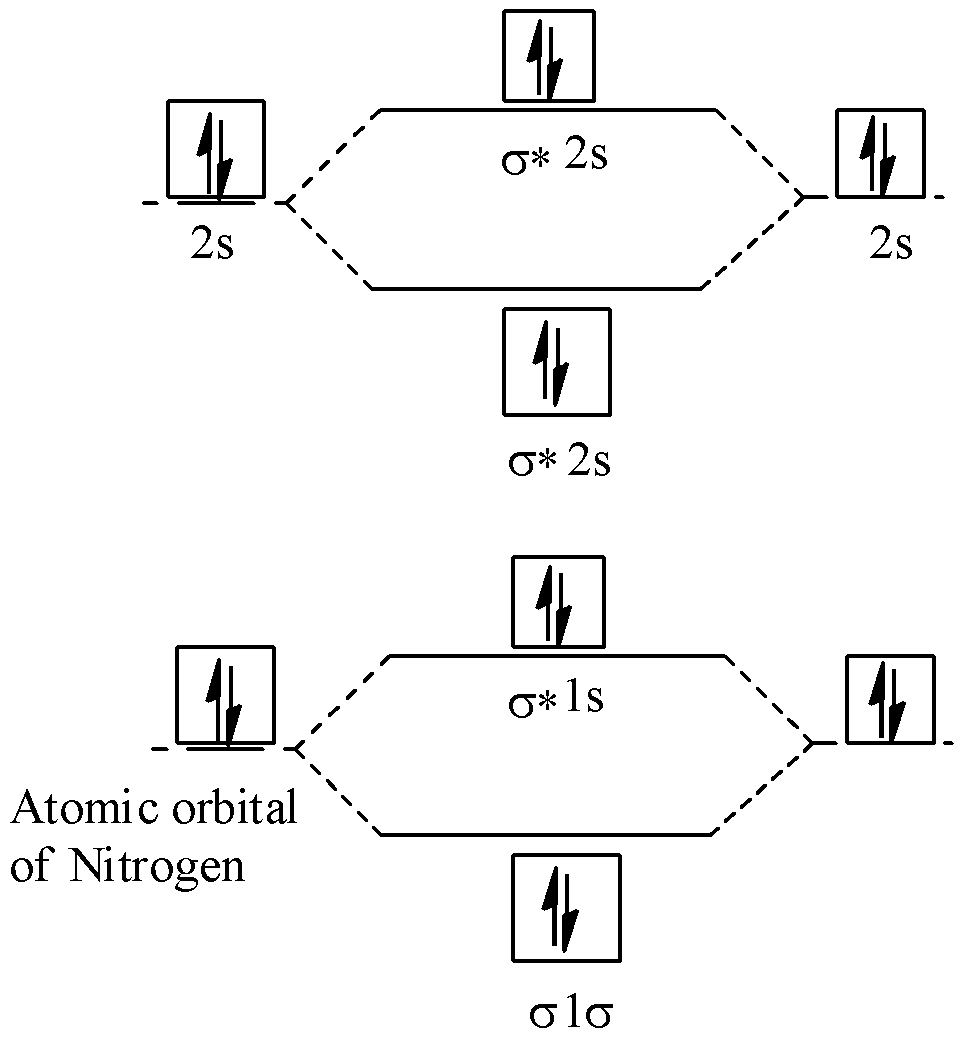
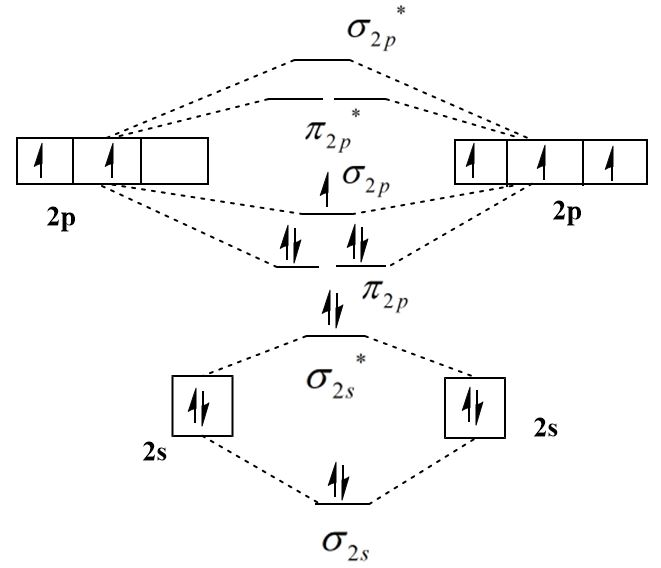
Bond order is a technical metric used in molecular orbital theory to quantify the stability of a chemical bond. Mathematically, it is calculated as half the difference between the number of electrons in bonding orbitals and those in antibonding orbitals. For $N2$, the calculation results in a bond order of 3. In the tech industry, this “triple bond” is synonymous with extreme structural integrity. When developers build simulations for explosive detection or high-pressure pneumatic systems, the triple bond of $N2$ represents a high-energy threshold that must be overcome, making it a “gold standard” for testing the accuracy of chemical simulation software.
Why $N_2$ is the Benchmark for Stability
The bond order of 3 implies that Nitrogen atoms share six electrons, creating one sigma bond and two pi bonds. From a digital modeling perspective, this requires high-fidelity calculations of electron density. Because the bond is so strong, $N2$ is chemically inert under standard conditions. This makes it an ideal substance for “blanketing” sensitive tech manufacturing processes, such as semiconductor fabrication, where preventing oxidation is critical. Software designed to manage the environments of cleanrooms relies heavily on the known physical constants of the $N2$ triple bond.
Technological Approaches to Calculating Bond Order
The determination of molecular structures has transitioned from manual calculations to sophisticated software suites. Today, identifying the bond order of complex molecules starts with the foundational data derived from simpler molecules like $N_2$.
Molecular Orbital (MO) Theory and Software Simulations
Modern computational tools like Gaussian, ORCA, and NWChem use Molecular Orbital Theory to visualize and calculate the electron distribution of $N_2$. These software packages allow researchers to solve the Schrödinger equation for many-electron systems. By inputting the atomic coordinates of two Nitrogen atoms, the software uses basis sets (mathematical functions) to predict where electrons reside. The resulting “output file” confirms a bond order of 3, validating the software’s ability to handle electron correlation—a key metric for any computational chemistry tool.
Density Functional Theory (DFT) in Modern Engineering
While MO theory provides a conceptual framework, Density Functional Theory (DFT) is the “workhorse” of the materials science industry. DFT software calculates the electronic structure of atoms and molecules by focusing on electron density rather than individual wavefunctions. For tech companies developing new battery technologies or carbon capture systems, using DFT to analyze the bond order of $N_2$ and its derivatives is a daily task. The efficiency of these algorithms determines how quickly a company can move from a digital concept to a physical prototype.
The Role of AI and Machine Learning in Molecular Analysis
We are currently witnessing a shift where classical simulation is being augmented—and in some cases replaced—by Artificial Intelligence (AI) and Machine Learning (ML).
Predictive Modeling for Chemical Bonds
Deep learning models are now being trained on massive datasets of molecular structures. Instead of solving grueling partial differential equations to find the bond order of a new nitrogen-based compound, an AI can predict the bond order and bond length in milliseconds. By using $N_2$ as a training baseline, these models learn the characteristics of a triple bond. This technological leap is essential for “High-Throughput Screening,” where AI explores millions of potential molecular combinations to find the next breakthrough in flexible electronics or high-strength polymers.
High-Throughput Screening of Nitrogen-Based Materials
In the tech world, “speed to market” is everything. High-throughput screening (HTS) uses cloud computing clusters to run thousands of simulations simultaneously. When engineers look for new refrigerants or propellants, they use HTS to filter molecules based on their bond orders. A higher bond order generally correlates with higher dissociation energy. By automating the calculation of bond orders, tech firms can quickly identify which molecules will remain stable under the extreme temperatures of a data center or the vacuum of space.
Quantum Computing: The Future of Bond Order Calculations
While classical supercomputers are efficient, they struggle with the “n-body problem” in complex molecules. This is where quantum computing enters the fray, promising to solve molecular structures with 100% accuracy.
Overcoming Classical Limitations with Quantum Algorithms
Classical computers approximate the behavior of electrons in $N2$. However, quantum computers use qubits, which can exist in multiple states simultaneously, to perfectly mirror the quantum nature of the Nitrogen triple bond. Algorithms such as the Variational Quantum Eigensolver (VQE) are currently being tested on hardware from companies like IBM and Google. These experiments often use the $N2$ bond order calculation as a “unit test” to prove that the quantum processor can handle electron entanglement better than a binary silicon chip.
Real-World Applications: From Aerospace to Sustainable Energy
The ability to perfectly calculate and manipulate bond orders through quantum tech has massive implications. For example, the “Haber-Bosch” process, which creates fertilizer from $N2$, consumes about 1% of the world’s energy because it is so difficult to break the $N2$ triple bond. Tech startups are now using quantum-inspired algorithms to design catalysts that can “weak” the bond order of $N_2$ at lower temperatures. If successful, this software-driven innovation could lead to a monumental shift in global energy consumption and green technology.
The Digital Transformation of Chemical Engineering
As we look toward the future, the boundary between “chemistry” and “tech” continues to blur. The digital transformation of the laboratory means that the bond order of $N_2$ is no longer just a theoretical value; it is a parameter in a much larger digital ecosystem.
Integrating Lab Data with Digital Twins
The concept of the “Digital Twin” involves creating a virtual replica of a physical process. In chemical manufacturing plants, sensors provide real-time data to a digital model that monitors the molecular state of gases. If the system detects that Nitrogen is reacting in an unexpected way, the digital twin uses the known bond order and dissociation energy of $N_2$ to troubleshoot the anomaly. This integration of IoT (Internet of Things) and molecular physics is a prime example of how fundamental science is embedded into modern tech infrastructure.
The Evolution of Computational Chemistry Tools
From the early days of Fortran-based chemistry scripts to modern, Python-integrated libraries like RDKit and PySCF, the tools used to calculate molecular properties have become more accessible. Today’s developers can use open-source APIs to pull data on bond orders, dipole moments, and ionization energies. This democratization of data allows even small tech startups to engage in sophisticated material design, using the stable triple bond of $N_2$ as a foundational building block for their innovations.
Conclusion
The question “what is the bond order of $N_2$?” leads us down a path that spans the entirety of the modern tech landscape. While the answer remains a constant “3,” the methods we use to arrive at that answer—and the ways we apply that knowledge—are evolving at a lightning pace. From the high-performance computing clusters running DFT simulations to the burgeoning field of quantum supremacy, the Nitrogen molecule remains a central figure in the story of technological progress.
As we continue to refine our AI models and quantum algorithms, our understanding of these fundamental molecular bonds will be the key to unlocking the next generation of materials, energy solutions, and digital tools. In the world of technology, even the smallest bond order is a gateway to a massive world of infinite possibility.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.