The human body is an intricate marvel of biological engineering, a complex ecosystem where each organ and system plays a vital role in maintaining life. Among the most crucial processes is the continuous production of red blood cells (RBCs), also known as erythrocytes. These microscopic powerhouses are responsible for transporting oxygen from our lungs to every cell in our body, a function absolutely indispensable for survival. But where does this constant supply of RBCs originate? While the answer might seem straightforward to some, a deeper dive into the technological and scientific understanding of this process reveals a fascinating interplay of biological mechanisms, advanced imaging, and molecular diagnostics, firmly placing it within the purview of cutting-edge Tech.

The primary site of red blood cell production, or erythropoiesis, is located deep within the intricate architecture of our skeletal system. This is not a casual, haphazard process but rather a highly regulated and sophisticated biological manufacturing plant. Over the centuries, our understanding of this organ has evolved dramatically, moving from rudimentary observations to the sophisticated technological insights we possess today.
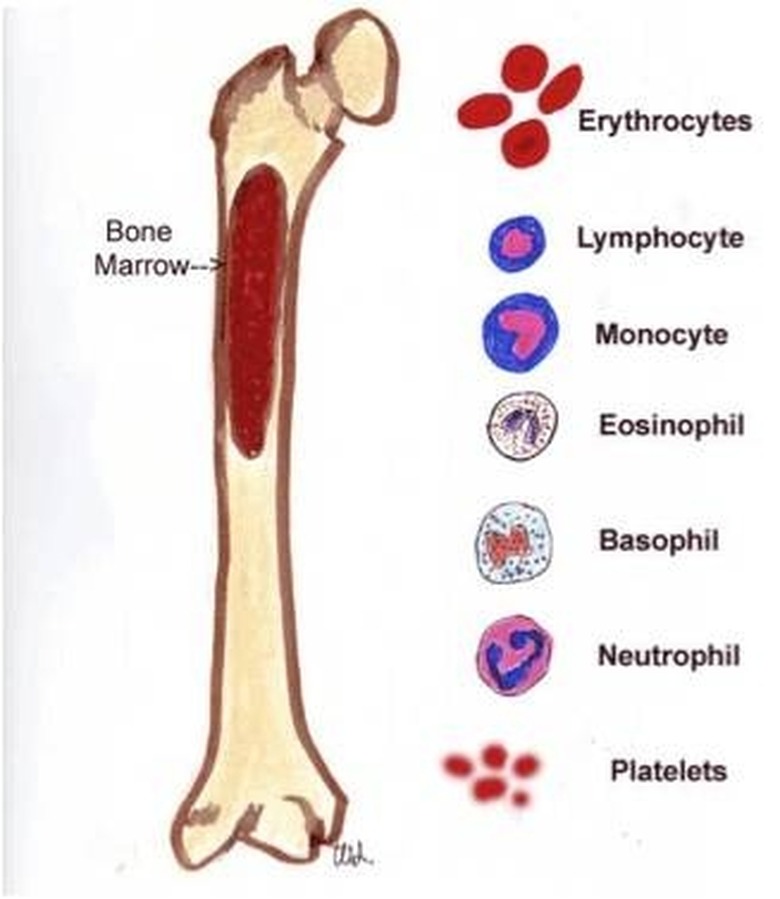
The Bone Marrow: A Sophisticated Biological Foundry
The undisputed champion of RBC production is the bone marrow, specifically the red bone marrow found within the spongy, porous inner core of certain bones. This is not just inert tissue; it’s a dynamic, living organ teeming with cellular activity and governed by complex signaling pathways. The transition from understanding bone marrow as a mere component of the skeleton to recognizing it as a highly specialized biological foundry is a testament to technological advancements in microscopy, cell biology, and molecular genetics.
The Microenvironment: A Crucial Technological Insight
Early scientific investigations, relying on visual examination, identified bone marrow as the source. However, modern technological tools have allowed us to dissect the crucial microenvironment within the bone marrow responsible for erythropoiesis. This microenvironment is not a single entity but a complex ecosystem comprising stromal cells, adipocytes, endothelial cells, and extracellular matrix components. These cells act as supportive scaffolding, providing essential growth factors and signaling molecules that nurture hematopoietic stem cells (HSCs) and guide their differentiation into RBC precursors.
Techniques like advanced confocal microscopy allow researchers to visualize the intricate spatial relationships between these cells and HSCs in real-time. Flow cytometry, a cornerstone of modern hematology, enables the precise identification and quantification of different cell populations within the bone marrow, including the stem cells and progenitor cells involved in RBC production. By analyzing the surface markers expressed by these cells, scientists can track their development and understand the signals that trigger their proliferation and maturation.
Hematopoietic Stem Cells: The Master Cells of Production
At the apex of the erythropoietic hierarchy are the hematopoietic stem cells (HSCs). These are multipotent stem cells residing in the bone marrow that possess the remarkable ability to self-renew and differentiate into all types of blood cells, including RBCs, white blood cells, and platelets. The identification and characterization of HSCs, a monumental achievement in biological research, relied heavily on technological innovations in cell sorting and immunological techniques.
The ability to isolate HSCs using fluorescence-activated cell sorting (FACS) has been transformative. This technology allows for the selection of rare cell populations based on their unique molecular fingerprints. Once isolated, these HSCs can be studied in vitro, allowing researchers to unravel the genetic programs and signaling cascades that govern their commitment to the erythroid lineage. Gene sequencing technologies further contribute by identifying the specific genes that are activated or silenced during this differentiation process, providing a molecular blueprint for RBC production.
The Role of Hormonal and Molecular Signaling: A Technologically Driven Understanding
The production of RBCs is not a spontaneous event but is meticulously regulated by a complex interplay of hormones and molecular signals. Understanding these intricate communication pathways has been significantly advanced through sophisticated biotechnological tools.
Erythropoietin (EPO): The Master Regulator
The most prominent regulator of erythropoiesis is the hormone erythropoietin (EPO). Primarily produced by the kidneys, EPO acts on the bone marrow, stimulating HSCs and their progeny to differentiate into RBCs. The discovery and characterization of EPO were landmark achievements, enabled by advances in radioimmunoassay (RIA) and later, enzyme-linked immunosorbent assay (ELISA) technologies, which allowed for the accurate measurement of hormone levels in the blood.
The subsequent technological leap came with the advent of recombinant DNA technology. This allowed for the production of synthetic EPO, which has revolutionized the treatment of anemia in patients with chronic kidney disease and other conditions where natural EPO production is insufficient. The development and refinement of these biotechnological processes for producing therapeutic EPO exemplify how understanding biological mechanisms directly translates into life-saving technological applications.
Growth Factors and Cytokines: The Symphony of Cellular Communication
Beyond EPO, a host of other growth factors and cytokines orchestrate the complex process of erythropoiesis within the bone marrow microenvironment. These signaling molecules, such as interleukins and stem cell factor, bind to specific receptors on developing RBC precursors, influencing their survival, proliferation, and maturation.
Techniques like quantitative polymerase chain reaction (qPCR) and RNA sequencing allow scientists to measure the expression levels of genes encoding these signaling molecules and their receptors. This provides a detailed map of the molecular conversations happening within the bone marrow. Furthermore, in vitro cell culture systems, coupled with precisely controlled delivery of these factors, allow researchers to mimic and manipulate these signaling pathways to understand their specific roles and to potentially develop novel therapeutic strategies for blood disorders.
Beyond the Bone Marrow: Technological Interventions and Future Horizons
While the bone marrow remains the primary site of RBC production throughout adult life, our technological understanding has opened up avenues for intervention and the exploration of alternative production sites, particularly in developmental stages and in pathological conditions.
Fetal Erythropoiesis: A Developmental Technological Insight
During fetal development, RBC production initially occurs in the yolk sac and then shifts to the liver before finally establishing in the bone marrow. This sequential transition is governed by a complex cascade of genetic and molecular events. Researchers utilize advanced imaging techniques like ultrasound and fetal blood sampling, coupled with sophisticated genetic analysis, to study these developmental stages. Understanding these early processes is crucial for diagnosing and treating congenital blood disorders.

Therapeutic Interventions: Leveraging Technological Advancements
The ability to understand and manipulate RBC production has led to significant technological advancements in treating various anemias. Beyond recombinant EPO, research is actively exploring gene therapy and stem cell transplantation as potential cures for inherited blood disorders that affect RBC production, such as sickle cell anemia and thalassemia.
CRISPR-Cas9 gene editing technology holds immense promise in correcting the genetic defects responsible for these conditions. While still largely in the experimental phase for widespread clinical application, the precision and efficiency of this gene-editing tool represent a paradigm shift in our ability to address the root causes of RBC production disorders. Furthermore, ongoing research into artificial blood substitutes and methods for ex vivo RBC production leverages bioengineering and cell culture technologies to address blood shortages and offer alternatives for transfusion-dependent patients.
In conclusion, the question “what organ produces RBC” leads us on a journey into the sophisticated biological machinery of the bone marrow. However, a truly comprehensive understanding, especially from a Tech perspective, reveals that this process is underpinned by a wealth of advanced technologies. From visualizing the intricate microenvironment and isolating master stem cells to deciphering hormonal signals and developing life-saving biotechnological interventions, technology has been and continues to be the key to unlocking the secrets of red blood cell production. This ongoing technological evolution promises even more groundbreaking discoveries and therapeutic possibilities in the years to come.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.