While the title “What Happens If Mounjaro Is Not Refrigerated” might initially suggest a focus on healthcare or pharmaceutical storage, when viewed through the lens of Brand, it opens a fascinating discussion about the critical role of brand integrity, regulatory compliance, and the downstream impact on consumer trust when a product’s handling instructions are violated. Mounjaro, a significant brand in the pharmaceutical space, exemplifies how a deviation from prescribed storage conditions can cascade into complex brand perception issues, impacting not only the efficacy of the drug but also the reputation of the company behind it. This exploration will delve into the brand implications of improper Mounjaro storage, focusing on how such incidents can affect brand perception, the importance of regulatory adherence in maintaining brand value, and the strategic measures companies must implement to safeguard their brands against the consequences of mishandled products.
The Erosion of Brand Trust: Beyond Pharmaceutical Efficacy
The primary concern when a medication like Mounjaro is not refrigerated is, of course, its potential loss of efficacy and the risk to patient health. However, from a Brand perspective, the repercussions extend far beyond the immediate medical implications. The integrity of a pharmaceutical brand is built on a foundation of trust, scientific rigor, and consistent quality. When a product fails to meet its intended performance due to improper handling, it directly challenges this trust.
Direct Impact on Patient Experience and Perception
For patients prescribed Mounjaro, the expectation is a reliable and effective treatment. If they receive or administer a product that has been compromised due to lack of refrigeration, their experience can be severely negative. This not only leads to a potentially ineffective treatment but also fosters doubt about the overall quality control and reliability of the brand.
- Perceived Ineffectiveness: Patients may attribute their lack of improvement to the drug itself, rather than the storage conditions. This can lead to frustration and a belief that the brand is not living up to its promises, even if the fault lies elsewhere.
- Safety Concerns: While the primary risk is reduced efficacy, consumers may also harbor anxieties about potential safety issues arising from temperature excursions. These fears, even if not medically substantiated in every case, can contribute to negative brand sentiment.
- Word-of-Mouth and Social Media: In today’s interconnected world, negative patient experiences spread rapidly. A single instance of a compromised Mounjaro pen, shared on social media or discussed within patient communities, can create a ripple effect, damaging the brand’s reputation among a wider audience. This can be particularly potent for a brand associated with significant health outcomes.
The Ripple Effect on Brand Reputation
A brand’s reputation is an intangible asset, meticulously cultivated over years through marketing, clinical trials, and positive patient outcomes. Improper handling of a product like Mounjaro can, directly and indirectly, tarnish this hard-won reputation.
- Undermining Brand Promise: Pharmaceutical brands often position themselves on pillars of innovation, reliability, and improved quality of life. A compromised product directly contradicts these core brand promises, creating a dissonance between what the brand claims and what the consumer experiences.
- Competitor Advantage: In a competitive market, any lapse in a competitor’s brand integrity can be an opportunity for rivals. Negative publicity surrounding Mounjaro’s storage issues could lead patients and healthcare providers to consider alternative medications, thereby benefiting competing brands.
- Investor Confidence: For publicly traded pharmaceutical companies, brand reputation is intrinsically linked to investor confidence. A significant incident related to product integrity can signal operational weaknesses, leading to a decline in stock value and affecting the company’s ability to attract future investment.
Regulatory Adherence: The Bedrock of Pharmaceutical Brand Value
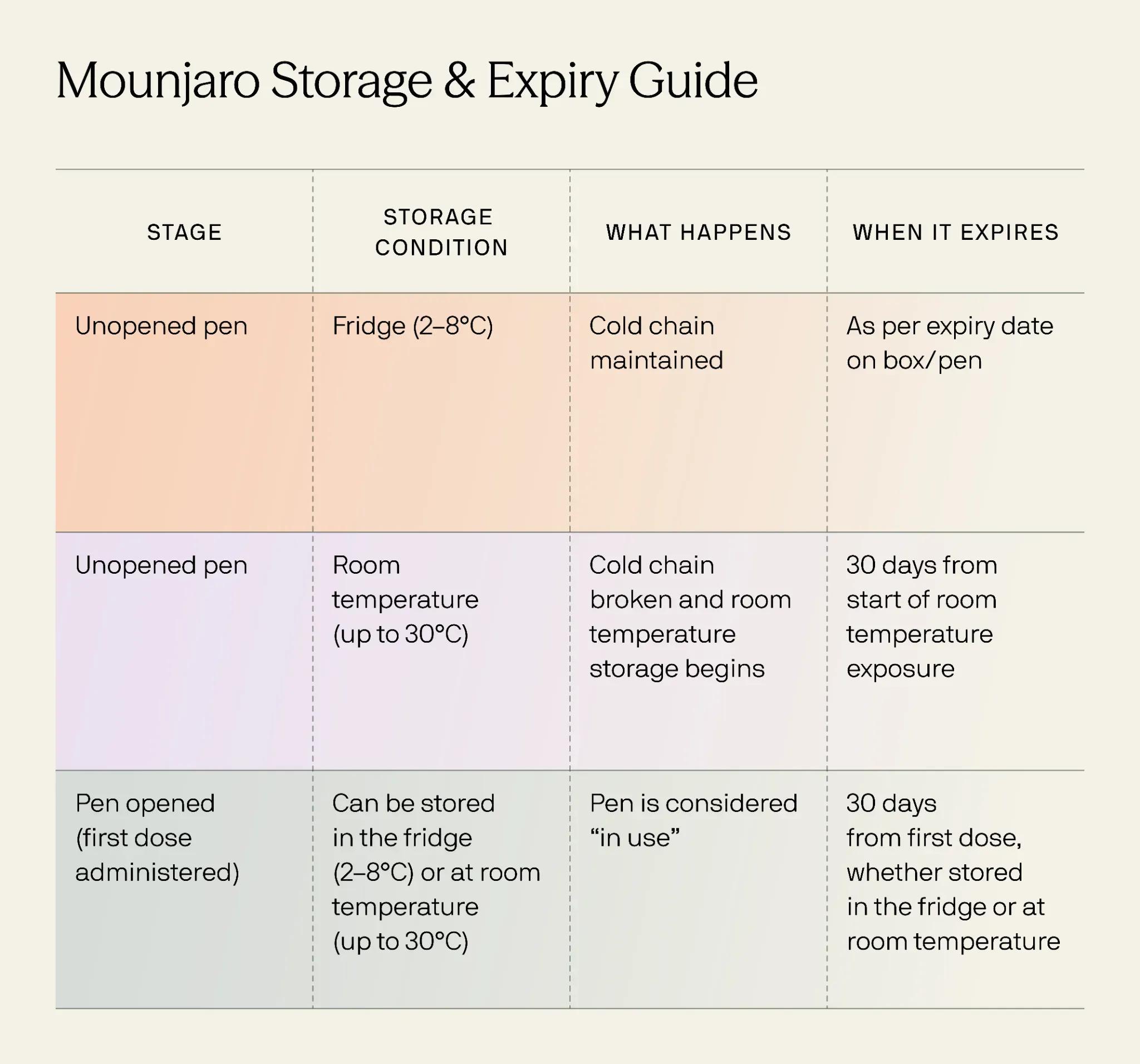
The stringent regulations governing pharmaceutical products are not merely bureaucratic hurdles; they are fundamental to ensuring product safety, efficacy, and ultimately, the sustained value of a brand. For a drug like Mounjaro, which is administered via injection and relies on precise chemical stability, adherence to cold chain requirements is paramount and directly tied to the brand’s credibility in the eyes of regulators and the public.
The Role of Regulatory Bodies in Brand Protection
Organizations like the FDA (in the United States) and other global health authorities set the standards for drug manufacturing, packaging, and distribution. Their oversight plays a crucial role in safeguarding the integrity of pharmaceutical brands.
- Mandatory Storage Conditions: The requirement for refrigeration for Mounjaro is not arbitrary. It is based on scientific data demonstrating the drug’s stability and efficacy within specific temperature ranges. Regulatory bodies mandate these conditions to ensure that the product reaching the patient is as intended by the manufacturer.
- Compliance as a Brand Differentiator: For pharmaceutical companies, strict adherence to regulatory guidelines is not just about avoiding penalties; it’s a key differentiator. Brands that consistently demonstrate compliance signal a commitment to quality and patient safety, enhancing their overall trustworthiness.
- Consequences of Non-Compliance: Failure to adhere to storage instructions can lead to regulatory action, including product recalls, warning letters, and even manufacturing facility shutdowns. Such actions have severe implications for a brand, signaling a fundamental breakdown in operations and a disregard for patient well-being.
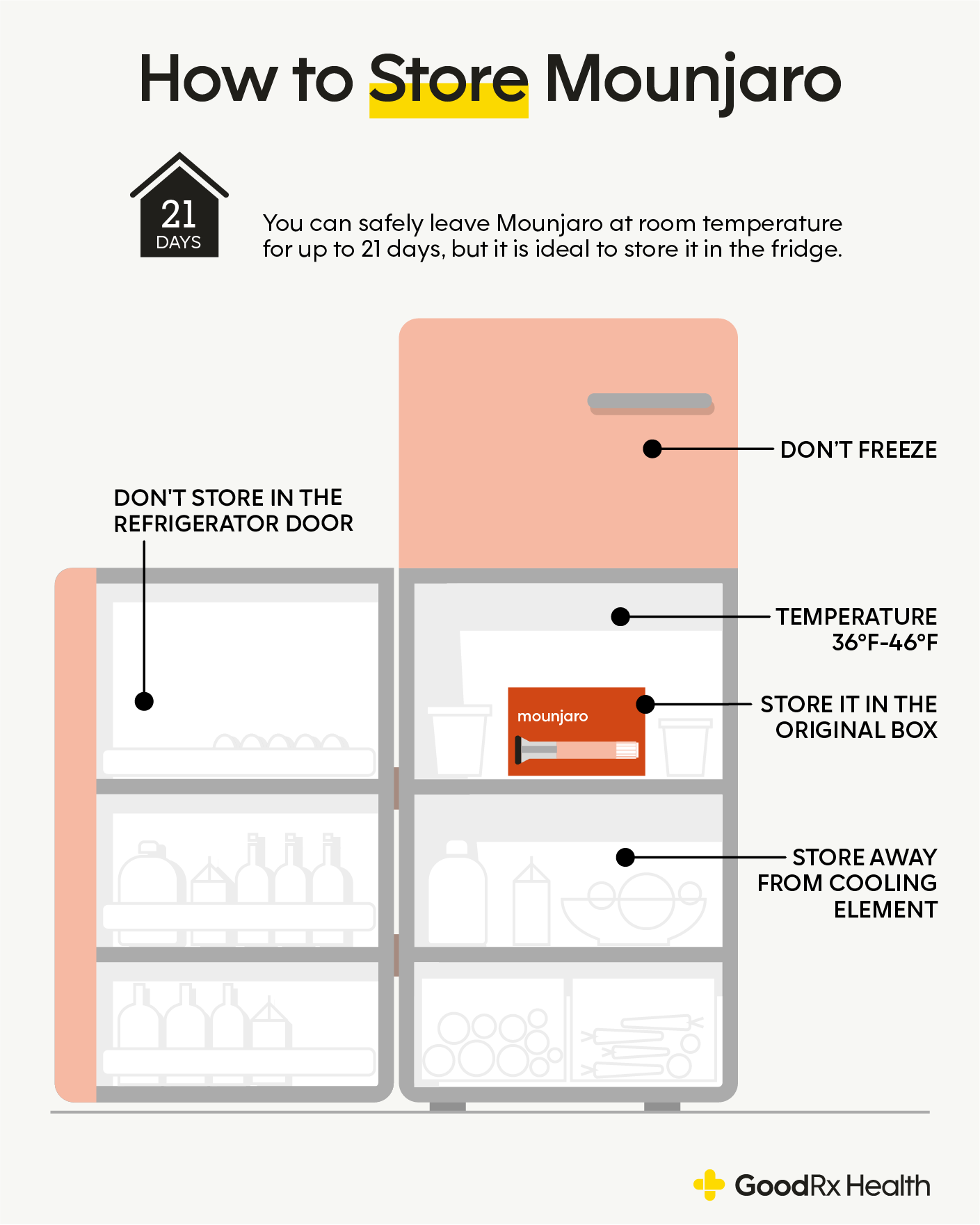
Maintaining the Cold Chain: A Brand Responsibility
The “cold chain” refers to the temperature-controlled supply chain required for pharmaceuticals, vaccines, and other sensitive products. For Mounjaro, maintaining this unbroken chain from manufacturing to the patient is a critical aspect of brand stewardship.
- Supply Chain Management as a Brand Pillar: A robust and reliable supply chain is a critical component of any pharmaceutical brand’s strategy. It ensures that the product is delivered safely and effectively, reinforcing the brand’s promise of quality and reliability.
- Educating Stakeholders: The responsibility for maintaining the cold chain extends beyond the manufacturer. It involves educating distributors, pharmacies, and most importantly, patients, on the proper handling and storage of the medication. Clear, accessible instructions are vital for brand success.
- Impact on Market Access and Trust: If a brand is perceived to have weak cold chain management, it can impact its ability to gain market access and foster trust among healthcare providers and payers, who are increasingly scrutinized for their own supply chain practices.
Strategic Brand Safeguards: Mitigating the Risks of Improper Storage
Protecting a brand like Mounjaro from the negative consequences of improper storage requires a proactive and multi-faceted strategic approach. This involves not only robust internal processes but also effective external communication and education initiatives.
Robust Packaging and Delivery Systems
The initial design and delivery of the product play a significant role in mitigating temperature-related risks.
- Innovative Packaging Solutions: Pharmaceutical companies are increasingly investing in innovative packaging that can help maintain temperature stability for a limited period, providing a buffer against minor temperature excursions during transit or brief delays. This includes insulated packaging and temperature-monitoring indicators.
- Logistical Partnerships: Selecting reliable logistics partners with proven expertise in cold chain management is crucial. These partners must adhere to strict protocols and possess the necessary infrastructure to ensure the integrity of the product throughout its journey.
- Real-time Temperature Monitoring: The implementation of advanced tracking and monitoring systems that provide real-time temperature data throughout the supply chain allows for early detection of any deviations and enables prompt corrective action. This data can also serve as an audit trail, reinforcing brand accountability.
Comprehensive Patient and Healthcare Provider Education
Empowering patients and healthcare professionals with the knowledge and tools to properly store and handle Mounjaro is a critical brand defense mechanism.
- Clear and Accessible Instructions: Product packaging and accompanying leaflets must provide clear, concise, and easily understandable instructions on refrigeration requirements. This includes visual aids and multilingual options.
- Digital Support and Reminders: Leveraging digital platforms, such as mobile apps or online portals, can offer additional support, including reminders for refrigeration, storage tips, and easy access to customer service for any queries.
- Healthcare Provider Training and Resources: Educating healthcare providers—doctors, nurses, and pharmacists—about the importance of Mounjaro’s storage conditions and how to effectively communicate these to patients is paramount. Providing them with resources and talking points reinforces the brand’s commitment to patient safety and efficacy.

Proactive Crisis Management and Communication
Despite best efforts, incidents of improper storage can occur. A well-defined crisis management plan is essential to minimize brand damage.
- Rapid Response Protocols: Having clear protocols in place to address reports of compromised products, including investigation, recall procedures (if necessary), and communication strategies, is vital.
- Transparent Communication: When issues arise, transparent and timely communication with patients, healthcare providers, and regulatory bodies is crucial. Acknowledging the problem, explaining the steps being taken, and demonstrating a commitment to resolution can help mitigate reputational damage.
- Learning and Improvement: Every incident, regardless of its scale, presents an opportunity to learn and improve. Analyzing the root cause of temperature excursions and implementing corrective actions ensures continuous enhancement of brand protection strategies.
In conclusion, while the immediate concern regarding Mounjaro not being refrigerated centers on its pharmaceutical efficacy and patient safety, the Brand implications are profound and far-reaching. The integrity of a pharmaceutical brand is a delicate construct, built on trust, quality, and regulatory adherence. Deviations from prescribed storage conditions, like failure to refrigerate, directly challenge this trust, erode brand reputation, and can have significant economic consequences. By implementing robust packaging, ensuring stringent supply chain management, prioritizing comprehensive education, and maintaining proactive crisis communication, pharmaceutical companies can effectively safeguard their brands against the potentially damaging effects of improper product handling, reinforcing their commitment to delivering reliable and effective treatments. The story of what happens when Mounjaro is not refrigerated is, at its core, a testament to the critical importance of upholding brand promise through meticulous attention to every detail, from the laboratory to the patient’s medicine cabinet.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.