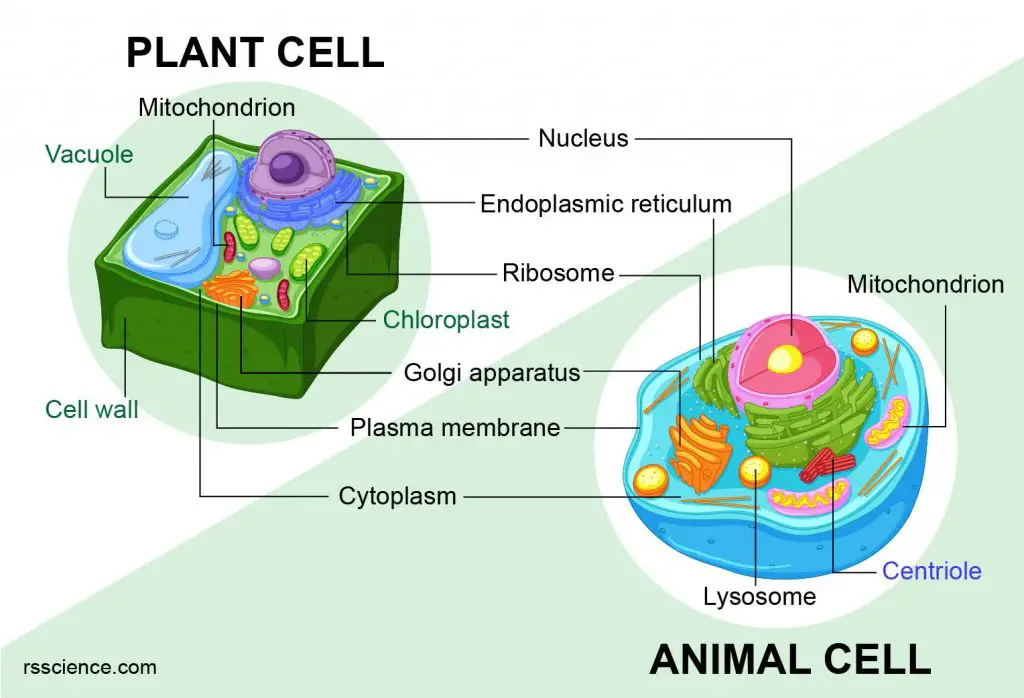
The fundamental differences between animal and plant cells represent more than just biological classification; they delineate distinct opportunities and challenges in technological innovation. While both share eukaryotic characteristics, a suite of unique components and processes found exclusively in animal cells forms the bedrock for pioneering advancements in medicine, diagnostics, and bioengineering. Understanding these distinctions is not merely an academic exercise but a critical pathway to developing highly targeted technological solutions.
Unique Cellular Components Driving Biomedical Innovation
Animal cells possess several distinguishing organelles and structures that are absent in plant cells, or present in a highly specialized, functionally divergent manner. These exclusive features are not mere curiosities but serve as critical targets and enablers for a vast array of modern biotechnologies, influencing drug design, regenerative medicine, and diagnostics.
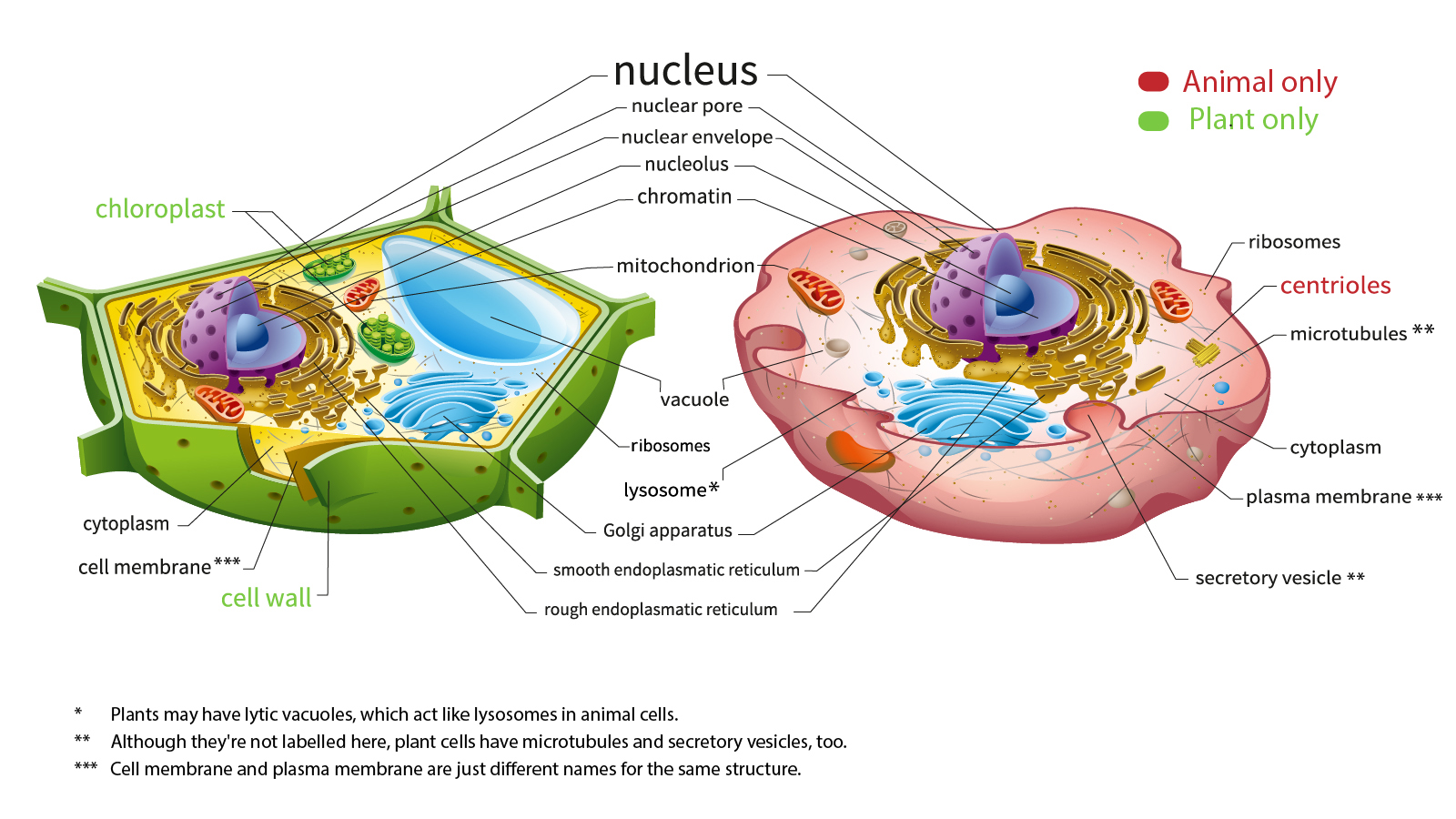
Centrioles and Precision Cell Division Engineering
One of the most defining characteristics of animal cells is the presence of centrioles, cylindrical structures composed of microtubules that play a pivotal role in cell division, forming the poles of the spindle fibers during mitosis and meiosis. Plant cells achieve similar spindle formation without centrioles. This animal-specific organelle is not just a structural difference but a key element in understanding and manipulating cell proliferation.
Technologically, the unique mechanism of centriole-driven cell division in animal cells is a prime target in oncology. Precision anti-cancer therapies often aim to disrupt the cell cycle by interfering with spindle formation or centriole function, exploiting their exclusivity to animal cells (and thus cancerous human cells) to minimize off-target effects on plant-based food sources or environmental systems. Furthermore, in vitro fertilization (IVF) and reproductive technologies heavily rely on understanding centriole behavior in oocytes and early embryos. Advanced imaging techniques and AI-driven image analysis are increasingly used to monitor centriole dynamics, providing insights into chromosomal segregation errors and contributing to the development of more effective reproductive interventions. Bioengineers are also exploring synthetic centriole analogues or inhibitors to precisely control the proliferation of animal cells in tissue culture for regenerative medicine applications, ensuring controlled growth without neoplastic transformation.
Lysosomes and Targeted Drug Delivery Systems
While both animal and plant cells contain vesicles with digestive enzymes, lysosomes—the primary acidic organelles responsible for cellular waste degradation and recycling—are far more prominent, numerous, and functionally diverse in animal cells. Plant cells primarily rely on a large central vacuole for similar functions. This distinction makes lysosomes a crucial component in animal cell biology and, consequently, in drug delivery and therapeutic strategies.
The acidic internal environment and hydrolytic enzyme repertoire of lysosomes are leveraged in advanced drug delivery systems. For instance, nanocarriers are engineered to specifically target and release their therapeutic cargo within lysosomes, a strategy particularly effective for treating lysosomal storage diseases through enzyme replacement therapy. Moreover, the understanding of lysosomal autophagy, an animal-cell-specific process where cellular components are broken down and recycled, has opened avenues for developing drugs that modulate autophagy to combat neurodegenerative diseases, infections, and certain cancers. AI algorithms are now employed to predict the optimal chemical structures for compounds that can either enhance or inhibit lysosomal activity, accelerating the discovery of novel therapeutic agents that precisely interact with these animal-specific degradative pathways.
Cholesterol and Membrane Engineering for Pharmaceuticals
Cholesterol is a vital lipid component of the animal cell plasma membrane, absent in plant cell membranes, which instead rely on phytosterols. Cholesterol contributes significantly to membrane fluidity, permeability, and the function of membrane-bound proteins. This fundamental difference has profound implications for pharmaceutical development and membrane-based technologies.
In drug discovery, the specific composition of animal cell membranes, particularly the presence of cholesterol, dictates how drugs interact with cell surface receptors and channels. Pharmaceutical companies invest heavily in understanding these interactions, using computational models and molecular dynamics simulations to predict drug efficacy and potential side effects based on membrane integration. Membrane engineering, a burgeoning field, utilizes animal cell membrane models (e.g., liposomes, nanodiscs) as sophisticated drug delivery vehicles that mimic natural cellular recognition and fusion processes, enhancing drug bioavailability and targeting specific tissues. Furthermore, cell culture technologies, which are foundational for vaccine production and therapeutic protein manufacturing, must meticulously replicate the cholesterol environment necessary for optimal animal cell growth and function, often involving specialized lipid supplements in bioreactor media.

Specialized Communication and Motility Enabling Advanced Bioengineering
Beyond individual organelles, animal cells exhibit unique capabilities in intercellular communication and active movement that are not found in plant cells, largely due to their lack of a rigid cell wall and their diverse tissue structures. These attributes are critical for organismal development, immune response, and wound healing, making them focal points for advanced bioengineering and therapeutic interventions.
Cell Motility and Microfluidic Device Development
Animal cells possess diverse mechanisms for active locomotion, such as pseudopods, flagella, and cilia, facilitating processes like immune cell migration, tissue repair, and embryonic development. Plant cells, encased in their cell walls, generally exhibit immotility. This inherent mobility of animal cells is a powerful tool in microfluidics and diagnostics.
Microfluidic devices, often termed “labs-on-a-chip,” exploit animal cell motility for high-throughput screening, cell sorting, and analysis. Researchers design intricate microchannels to observe cell migration patterns in response to chemical gradients (chemotaxis), mimicking in vivo environments. This technology is invaluable for studying cancer metastasis, immune cell activation, and drug efficacy, allowing for the rapid identification of compounds that modulate cell movement. Robotics and automation are integrated into these systems, enabling precise manipulation and analysis of individual motile animal cells, which is impossible with static plant cells. The ability to precisely control and analyze animal cell movement underpins the development of more accurate diagnostic tools for diseases involving cellular dysfunction, such as autoimmune disorders and infectious diseases.
Specialized Cell Junctions and Organ-on-a-Chip Models
Animal cells form various types of specialized intercellular junctions—tight junctions, gap junctions, desmosomes, and adherens junctions—that facilitate direct cell-to-cell communication, adhesion, and tissue integrity. These complex structures are largely absent in plant cells, which instead rely on plasmodesmata for cytoplasmic continuity. The intricate architecture and dynamic regulation of animal cell junctions are central to advanced tissue engineering and sophisticated in vitro models.
Organ-on-a-chip platforms are revolutionary bioengineering tools that leverage these animal-specific junctions to mimic the physiological functions of human organs. By culturing different animal cell types on microfabricated scaffolds, researchers can recreate the complex tissue architectures and intercellular communication networks found in vivo. For example, gut-on-a-chip models utilize tight junctions to simulate the intestinal barrier, allowing for precise studies of drug absorption and microbial interactions. Brain-on-a-chip and lung-on-a-chip models similarly capitalize on the unique junctional complexes to replicate complex physiological barriers and functional units. These advanced models significantly reduce reliance on animal testing and accelerate drug discovery by providing more accurate predictions of drug efficacy and toxicity in a human-relevant context. Digital twins of these organ-on-a-chip models, built using computational biology, further enhance their utility by allowing predictive simulations of drug responses and disease progression, all reliant on the unique communication structures of animal cells.
Leveraging AI and Computational Biology for Animal Cell-Specific Research
The complexities arising from these unique animal cell features present an enormous challenge and opportunity for artificial intelligence (AI) and computational biology. AI excels at identifying subtle patterns, making predictions, and automating processes that are crucial for harnessing the full potential of animal cell technologies.
AI-driven image analysis is transforming cellular research. Machine learning algorithms are trained to recognize and quantify specific animal cell structures, such as centrioles, lysosomes, or junctional complexes, from high-resolution microscopy images. This automation accelerates phenotypic screening for drug discovery, allowing researchers to rapidly assess the impact of thousands of compounds on animal cell morphology, proliferation, and function. Predictive modeling, leveraging AI, is used to simulate complex animal cell signaling pathways and predict drug interactions with unique animal cell receptors or enzymes, significantly de-risking pharmaceutical development. Furthermore, computational genomics and proteomics focus on identifying gene regulatory networks and protein interactions unique to animal cells, providing targets for gene editing and personalized medicine approaches that would be irrelevant in a plant cell context.
Ethical and Digital Security Dimensions of Animal Cell Technologies
The burgeoning field of animal cell-based technologies also raises critical ethical considerations and necessitates robust digital security frameworks. When dealing with human or animal cell lines, genetic data, and proprietary research, the stakes are exceptionally high.
Biosecurity protocols are paramount for managing animal cell cultures, especially those used in vaccine production or advanced therapies. The potential for contamination or accidental release of genetically modified animal cells requires stringent digital tracking and access control systems for laboratory equipment and data. Data privacy for genomic sequencing of human and animal cells is a significant concern, demanding advanced encryption and secure cloud storage solutions to protect sensitive biological information. Ethical AI in animal experimentation simulations ensures that algorithms are developed responsibly, minimizing bias and upholding animal welfare standards when creating digital models to replace live animal testing. Moreover, preventing biopiracy and intellectual property theft related to unique animal cell lines or biotechnological processes requires sophisticated digital rights management and secure data sharing platforms, safeguarding innovative developments that capitalize on the intrinsic differences between animal and plant biology.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.