The question “what’s the pH of distilled water?” seems straightforward, almost elementary. Yet, like many seemingly simple scientific inquiries, the answer is nuanced and reveals a fascinating interplay between purity, environment, and measurement. For those navigating the worlds of technology, branding, and finance, understanding the fundamentals of something as basic as water can offer surprising insights, from optimizing digital processes to understanding the meticulous nature of branding and the essentiality of purity in certain financial instruments.
While the commonly cited pH of pure water is 7, often labeled as neutral, distilled water, due to its extreme purity, presents a unique case. This article will explore the scientific reasons behind its pH, how external factors can influence it, and why this seemingly minor detail holds significance across various domains, even those as far removed as technology trends and corporate identity.
Understanding pH and the Ideal of Pure Water
Before we can definitively answer the pH of distilled water, it’s crucial to establish a foundational understanding of what pH is and what constitutes “pure” water.
The pH Scale: A Measure of Acidity and Alkalinity
The pH scale is a logarithmic measure of the hydrogen ion (H+) concentration in a solution. It ranges from 0 to 14:
- pH < 7: Acidic (higher concentration of H+ ions)
- pH = 7: Neutral (equal concentration of H+ ions and hydroxide ions, OH-)
- pH > 7: Alkaline or basic (lower concentration of H+ ions and higher concentration of OH- ions)
At 25 degrees Celsius, pure water undergoes a process called autoionization, where a small fraction of water molecules dissociate into H+ and OH- ions. In an ideal, perfectly pure water sample at this temperature, the concentration of H+ ions is 1 x 10⁻⁷ moles per liter. This concentration gives pure water a neutral pH of 7.
Distilled Water: The Pursuit of Purity
Distilled water is produced by boiling water and then condensing the steam back into liquid form. This process effectively removes most impurities, including minerals, salts, and other dissolved solids. The goal is to create a highly pure, demineralized water. Because these dissolved substances are typically neutral or slightly alkaline in their pure form, their removal brings the water closer to the theoretical ideal of pure H₂O.
However, this very purity makes distilled water highly susceptible to absorbing substances from its surroundings. Unlike tap water, which contains dissolved minerals that buffer it against pH changes, distilled water has very little buffering capacity. This is a key factor in understanding its pH.
The Nuances of Distilled Water’s pH: Why it’s Not Always Exactly 7
While we strive for a pH of 7 with distilled water, achieving and maintaining it can be more complex than it appears. Several factors can influence the measured pH, leading to values that might deviate slightly from the ideal neutral point.
The Influence of Dissolved Carbon Dioxide
One of the most significant factors affecting the pH of distilled water is the absorption of carbon dioxide (CO₂) from the atmosphere. Carbon dioxide readily dissolves in water to form carbonic acid (H₂CO₃), a weak acid.
The reaction proceeds as follows:
CO₂ (g) + H₂O (l) ⇌ H₂CO₃ (aq)
Carbonic acid then dissociates to release hydrogen ions (H+):
H₂CO₃ (aq) ⇌ H⁺ (aq) + HCO₃⁻ (aq)
This increase in H+ ions lowers the pH of the water, making it slightly acidic. Therefore, distilled water that has been exposed to the air for a period will often have a pH slightly below 7, typically ranging from 5.5 to 6.5. This is a natural process and a testament to the water’s purity and lack of buffering agents.
Implications for Technology and Digital Processes:
In high-tech manufacturing, particularly in the semiconductor industry, ultra-pure water is essential. Even minute impurities can contaminate delicate circuitry. While the pH of distilled water might be slightly acidic due to dissolved CO₂, the absence of dissolved ions is often a greater concern. Processes requiring precise pH control might necessitate the use of deionized water with carefully adjusted pH levels, often achieved through the addition of specific buffering agents or the use of highly controlled water purification systems. Understanding these subtle pH shifts is critical for maintaining the integrity of sensitive technological processes, akin to optimizing algorithms for speed and efficiency.
The Role of Container Materials
The material of the container in which distilled water is stored can also influence its pH.
- Glass: Generally considered inert and the best choice for storing distilled water to maintain its purity and pH. However, even glass can leach trace amounts of silica over very long periods.
- Plastic: Many plastics can leach chemicals into the water, potentially altering its pH and introducing contaminants. The type of plastic and the duration of contact are critical factors. Some plastics are specifically designed for laboratory use and are less prone to leaching.
- Metal: Metal containers, especially those not made of inert materials like stainless steel, can react with the water and introduce metallic ions, which can affect pH and purity.
Branding and Corporate Identity:

The choice of packaging material for products, especially those associated with health, wellness, or high quality, is a crucial branding decision. Just as distilled water’s purity can be compromised by its container, a brand’s integrity can be undermined by poor-quality packaging. A premium product housed in cheap, flimsy packaging signals a disconnect in brand strategy. Companies invest heavily in material science and design to ensure their packaging not only protects the product but also reinforces the brand’s message of quality, sustainability, or luxury. The meticulous attention to detail in selecting packaging materials mirrors the careful control needed to maintain the intended pH of distilled water.
Temperature Variations
The autoionization of water is temperature-dependent. As temperature increases, water autoionizes more, meaning there are more H+ and OH- ions. This leads to a decrease in the pH value, even in pure water. So, while pure water is neutral at 25°C (pH 7), it might have a pH of around 7.4 at 0°C and a pH of around 6.14 at 100°C.
Relevance to Financial Markets and Investment:
While seemingly unrelated, understanding dynamic systems and how environmental factors influence outcomes can be broadly applied. In financial markets, algorithms and trading strategies are constantly recalibrated to account for fluctuating market conditions, economic indicators, and even geopolitical events. Just as temperature affects water’s pH, external stimuli can significantly alter market behavior. Investors and tech developers in the finance sector must be aware of these dynamic shifts and build systems that can adapt. The concept of neutrality being a point in a spectrum, influenced by external forces, is a valuable lesson in risk management and strategic planning, whether one is dealing with chemical solutions or volatile stock prices.
Practical Applications and Why pH Matters
The pH of distilled water might seem like a niche scientific detail, but its understanding has practical implications across various fields, including those touched upon by the website’s core topics.
In Scientific Research and Laboratories
In many scientific experiments, precise control of chemical conditions is paramount. Distilled or deionized water is often used as a solvent or a base for solutions because it lacks the dissolved ions that could interfere with experimental results.
- Chemical Reactions: The pH of the water can affect the rate and outcome of chemical reactions.
- Biological Studies: In cell culture or experiments involving sensitive biological samples, maintaining a specific pH environment is crucial for cell viability and experimental integrity.
- Calibration: pH meters and other sensitive equipment are often calibrated using solutions made with distilled water to ensure accurate readings.
Tech and AI Tools:
The precision required in scientific labs mirrors the demand for accuracy in technological development. When developing AI models for scientific research or complex simulations, the underlying data and the environments in which these models are trained and tested need to be meticulously controlled. The use of high-purity water in scientific equipment, like mass spectrometers or chromatography systems, is analogous to the need for clean, well-defined datasets in AI development. Errors or impurities in either can lead to flawed results and unreliable insights. Developers creating specialized software for laboratory automation or data analysis must account for the precise chemical requirements of these environments.
In Personal Health and Wellness
While drinking distilled water is generally safe, its lack of minerals can be a point of discussion. Our bodies rely on a delicate balance of electrolytes, and prolonged consumption of demineralized water might, in theory, lead to a slight depletion of essential minerals if not balanced by diet.
- Hydration: Distilled water effectively hydrates the body.
- Taste: Many people find the taste of distilled water to be flat or unappealing due to the absence of dissolved minerals.
Personal Finance and Lifestyle Choices:
The discussion around distilled water’s mineral content can draw parallels to personal finance and lifestyle choices. Consumers are often presented with choices that involve perceived purity, added benefits, or cost-effectiveness. For instance, choosing between filtered tap water, bottled mineral water, or even distilled water for drinking involves weighing factors like cost, perceived health benefits, and environmental impact. In personal finance, individuals might choose between investing in low-cost index funds (analogous to tap water – functional, readily available) versus more specialized, potentially higher-fee investment vehicles (akin to premium bottled water with added “benefits”). Understanding what truly matters – in this case, hydration and safety – versus marketing claims or perceived premium qualities is a key financial literacy skill.
In Industrial Applications
Beyond scientific labs, distilled and deionized water are vital in numerous industrial processes:
- Boilers and Steam Generation: In power plants and industrial facilities, highly purified water is used in boilers to prevent scale buildup and corrosion, improving efficiency and lifespan.
- Pharmaceuticals and Cosmetics: The production of medicines and cosmetic products demands ultra-pure water to avoid contamination and ensure product safety and efficacy.
- Automotive Industry: Used in car batteries, cooling systems, and paint shops.
Business Finance and Operational Efficiency:
The consistent and reliable supply of high-purity water is crucial for industrial operations, directly impacting production quality, equipment maintenance costs, and overall business efficiency. This is analogous to the importance of robust financial management and clear operational processes in business finance. A company that neglects its supply chain for critical resources like water, or fails to maintain its machinery properly (which purified water helps with), will eventually face higher costs and reduced output. Sound business finance involves proactively managing such operational necessities to ensure long-term profitability and sustainability.
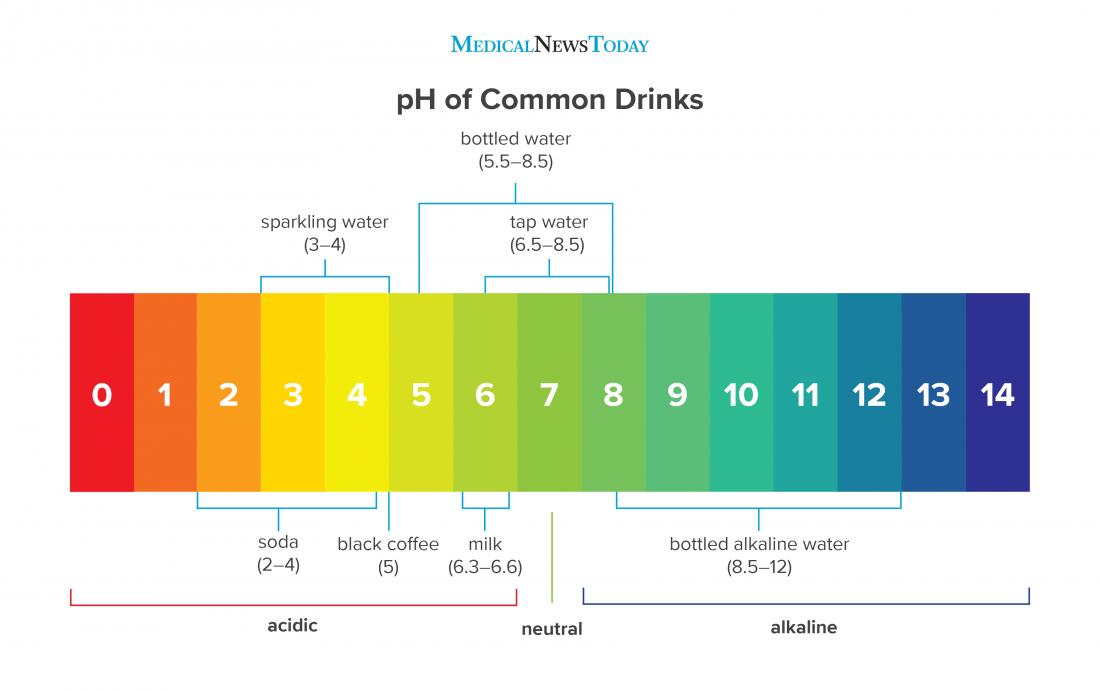
Conclusion: The pH of Distilled Water is More Than Just a Number
The question of “what’s the pH of distilled water” is a gateway to understanding the subtle yet significant influence of purity, environment, and measurement. While often cited as neutral (pH 7), distilled water is in a constant state of interaction with its surroundings, most notably absorbing CO₂ from the air to become slightly acidic.
For professionals in technology, branding, and finance, this seemingly simple scientific concept offers valuable lessons:
- Technology: Emphasizes the critical importance of precision, purity, and controlled environments in achieving desired outcomes, whether in semiconductor manufacturing or AI development.
- Branding: Highlights how seemingly minor details, like packaging materials, can significantly impact perception and brand integrity, requiring meticulous attention to every element.
- Money: Illustrates that true value often lies beneath the surface, requiring an understanding of fundamental principles and dynamic forces to make sound decisions, whether managing personal finances or navigating complex market conditions.
Ultimately, the pH of distilled water serves as a reminder that even the most fundamental elements of our world are dynamic, interconnected, and deserve a closer look to unlock their full significance.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.