In the rapidly evolving landscape of health technology, the focus has long remained on blood-based diagnostics and heart rate variability. However, as we push toward the era of personalized medicine and continuous physiological monitoring, a new medium is emerging as a critical data source for tech innovators: serous fluid. While traditionally a term relegated to the halls of biology and clinical pathology, “serous fluid” is becoming a buzzword in the development of next-generation biosensors, lab-on-a-chip (LOC) devices, and wearable health tech.
Understanding what serous fluid is—and more importantly, how technology can capture and interpret its data—is essential for anyone tracking the intersection of biotechnology and digital health. This article explores the technical nuances of serous fluid and the hardware-software ecosystems being built to decode it.
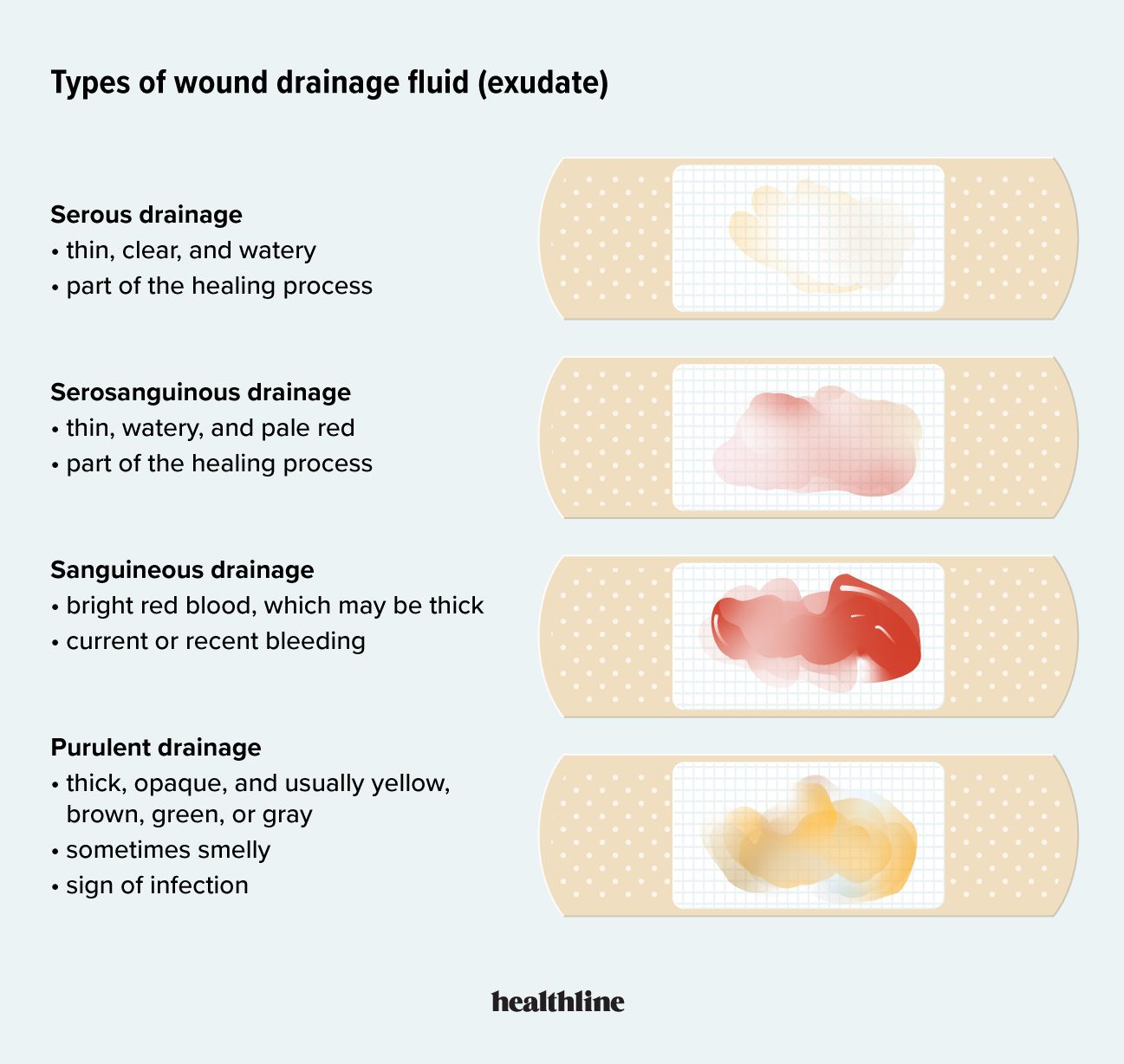
The Biological Signal: Understanding Serous Fluid in the Digital Age
Before diving into the hardware, we must establish the technical parameters of the medium. Serous fluid is a clear, pale yellow, or transparent fluid found within the various body cavities—specifically the pleural, pericardial, and peritoneal cavities. Its primary biological function is to reduce friction between organs, but from a data-science perspective, it is a rich reservoir of biochemical information.
What is Serous Fluid? A Technical Overview
Serous fluid is an ultrafiltrate of blood plasma. It consists of water, electrolytes, and proteins such as albumin. Unlike blood, which is dense with red blood cells and prone to clotting, serous fluid provides a relatively “cleaner” substrate for certain types of chemical analysis. In medical technology, the concentration of specific enzymes or proteins within this fluid serves as a high-fidelity signal for internal inflammation, infection, or organ dysfunction.
The Role of Interstitial and Serous Fluids in Diagnostic Data
In the tech world, we often group serous fluid with interstitial fluid (the fluid between cells). These fluids represent a frontier for “minimally invasive” tech. Because these fluids are governed by osmotic pressure and hydrostatic filtration, they reflect the body’s internal state in real-time. For developers of AI-driven diagnostic tools, serous fluid represents a “low-noise” environment compared to the complex cellular matrix of whole blood, making it an ideal candidate for automated micro-sampling.
Wearable Tech and Non-Invasive Monitoring
The wearable market is currently dominated by optical sensors (PPG) that track blood flow. However, the next leap in gadgetry—often referred to as “Wearables 2.0″—is moving toward biochemical sensing. This shift involves moving from tracking movement to tracking molecules.
Moving Beyond Blood: Why Serous Fluid Matters for Wearables
One of the greatest hurdles in tech-enabled health monitoring is the “needle barrier.” Most users are unwilling to use devices that require frequent blood draws. Serous and interstitial fluids can be accessed through the skin barrier via transdermal extraction techniques that do not trigger pain receptors. Companies like Dexcom and Abbott have already pioneered this with continuous glucose monitors (CGMs), but the next generation of sensors aims to target the specific markers found in serous-type fluids to monitor more than just sugar.
Smart Patches and Micro-Needle Sensors
The hardware currently under development involves “smart patches” equipped with arrays of microneedles. These needles are often less than 1mm in length, designed to reach the fluid-filled layers of the dermis without hitting nerves.
- Sensor Integration: These patches utilize electrochemical sensors to convert chemical concentrations in the serous fluid into digital signals.
- Data Transmission: Using Low-Energy Bluetooth (BLE) or Near-Field Communication (NFC), the patch transmits this bio-data to a smartphone or a cloud-based AI for processing.
- Use Cases: Tech startups are currently prototyping patches that detect lactic acid, cortisol (the stress hormone), and even specific biomarkers for heart failure that accumulate in serous fluid before they manifest as physical symptoms.

Lab-on-a-Chip: Microfluidics and Serous Analysis
The miniaturization of laboratory equipment is perhaps the most significant tech trend of the decade. Lab-on-a-chip (LOC) technology integrates one or several laboratory functions on a single integrated circuit (only millimeters to a few square centimeters in size). Serous fluid is the perfect medium for these microfluidic systems.
Precision Medicine through Microfluidic Technology
Microfluidics deals with the behavior, precise control, and manipulation of fluids that are geometrically constrained to a small scale. Because serous fluid is thin and consistent, it can be moved through micro-channels using electro-osmotic flow.
- Automated Sorting: Advanced LOC devices can sort cells within a serous sample, identifying malignant cells in cases of pleural effusion (fluid buildup in the lungs) using high-speed imaging and machine learning.
- Point-of-Care (POC) Devices: Tech firms are developing handheld devices that allow a technician to take a tiny sample of serous fluid and receive a full proteomic profile in minutes. This removes the need for expensive, time-consuming “wet labs.”
AI-Driven Diagnostics: Processing Serous Data at the Edge
The “Edge Computing” revolution is also playing a role here. Instead of sending raw biological data to a central server, new bio-chips are being designed with enough processing power to analyze the fluid data locally. By applying neural networks to the biochemical signatures found in serous fluid, these devices can detect patterns—such as the specific protein “fingerprint” of a bacterial infection—instantly. This synthesis of microfluidics and on-chip AI is the pinnacle of current med-tech innovation.
The Future of Health-Tech: Predictive Analytics and Real-Time Bio-Feedback
As we look toward the 2030s, the integration of serous fluid analysis into the broader Internet of Medical Things (IoMT) will transition from reactive care to predictive maintenance of the human body.
Integrating Serous Biomarkers into the Internet of Medical Things (IoMT)
The IoMT is an ecosystem of connected medical devices and software applications that can communicate with healthcare IT systems. By constantly monitoring the volume and composition of serous fluid in high-risk patients (such as those with chronic heart or kidney issues), the IoMT can trigger automated alerts.
- Cloud Synchronization: Imagine a scenario where a patient’s internal “smart stent” detects a change in the serous fluid surrounding the heart. This data is instantly synced to the cloud, compared against millions of other data points, and an alert is sent to a cardiologist before the patient even feels a chest pain.
- Digital Twins: Tech companies are using data derived from serous fluid to create “digital twins”—virtual models of a patient’s biology. These models allow doctors to simulate how a specific drug might react with the patient’s unique biochemical makeup.
Cybersecurity Challenges in Bio-Data Transmission
With the rise of fluid-sensing technology comes a significant tech challenge: digital security. Serous fluid data is the ultimate form of “Personally Identifiable Information” (PII).
- Encryption at the Source: Emerging tech standards require that bio-data be encrypted at the sensor level.
- Blockchain for Bio-Data: Some developers are looking at decentralized ledgers (blockchain) to store serous fluid diagnostic history. This ensures that while the data is accessible for emergency medical response, it remains immutable and owned solely by the patient, preventing unauthorized access by insurance companies or third-party bad actors.
Conclusion: The Siliconization of Biology
The question “what is serous fluid?” no longer has a purely medical answer. In the context of modern technology, serous fluid is a high-bandwidth data stream. It is the liquid interface between our biological systems and the digital tools we use to understand them.
From the development of painless micro-needle patches to the deployment of AI-powered microfluidic chips, the tech industry is successfully “siliconizing” biology. By mastering the collection and analysis of serous fluid, we are moving toward a future where our gadgets don’t just tell us how many steps we’ve taken, but provide a deep, real-time diagnostic map of our internal health. The fluid is old, but the technology we are using to read it is the absolute cutting edge of the digital revolution.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.