In the vast and intricate tapestry of the periodic table, elements are categorized into distinct groups: metals, nonmetals, and a fascinating, often overlooked class known as metalloids. These enigmatic elements occupy a unique interstitial space, exhibiting properties that blur the lines between their more definitive neighbors. Far from being mere curiosities, metalloids are, in fact, the unsung heroes of the digital age. Their peculiar electrical conductivity—a characteristic falling precisely between that of conductors and insulators—is the foundational principle upon which the entire edifice of modern technology is built. From the microprocessors powering our smartphones and AI systems to the advanced sensors in autonomous vehicles, the unique capabilities of metalloids, particularly silicon and germanium, have catalyzed a technological revolution that continues to shape every facet of human existence. This exploration delves into the nature of metalloids, their defining characteristics, and their indispensable role in propelling technological innovation forward.

The Unique Nature of Metalloids: Bridging the Divide
The periodic table is elegantly organized, revealing patterns in atomic structure and chemical behavior. While metals are known for their high electrical and thermal conductivity, malleability, and lustrous appearance, nonmetals typically exhibit the opposite traits—poor conductivity, brittleness, and dullness. Metalloids exist in a fascinating “Goldilocks zone,” possessing a hybrid set of properties that make them invaluable for specific technological applications.
Defining Metalloids: A Chemical Ambiguity
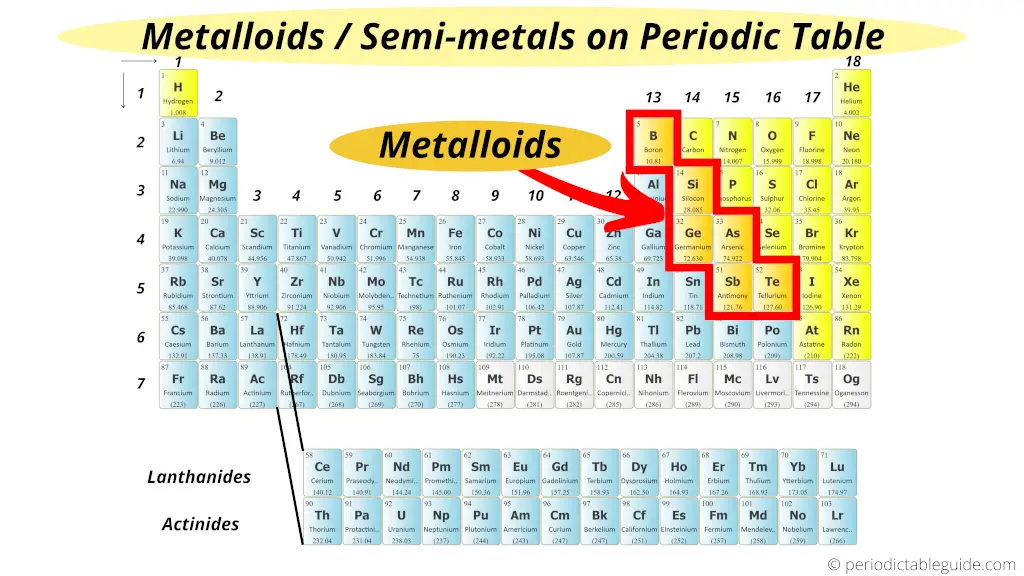
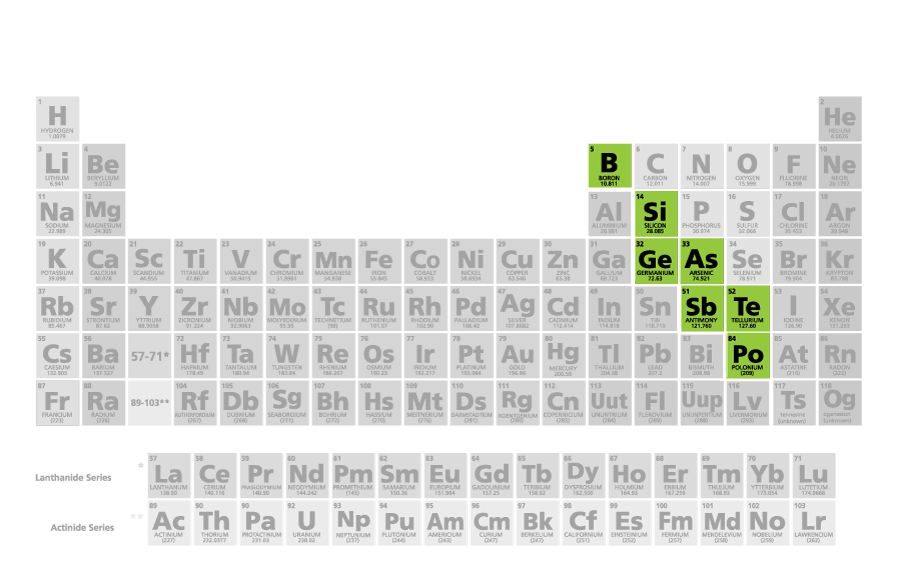
Metalloids, sometimes referred to as semimetals, are found along the stair-step line that separates metals from nonmetals on the periodic table. The six commonly recognized metalloids are Boron (B), Silicon (Si), Germanium (Ge), Arsenic (As), Antimony (Sb), and Tellurium (Te). Polonium (Po) and Astatine (At) are sometimes also classified as metalloids, though their properties are less clear due to their high radioactivity and scarcity.
Chemically, metalloids exhibit an intriguing duality. They often have the physical appearance of metals—possessing a metallic luster—yet they are typically brittle like nonmetals. Their atomic structures allow them to form covalent bonds readily, similar to nonmetals, but they can also exhibit more metallic bonding characteristics under certain conditions. This chemical versatility allows them to engage in a wide array of reactions and form compounds with unique properties, many of which are directly leveraged in advanced materials and electronics.
Electrical Conductivity: The Goldilocks Zone
The most distinguishing and technologically significant property of metalloids is their semiconducting nature. Unlike metals, which conduct electricity readily due due to their delocalized electrons, and nonmetals, which act as insulators due to tightly bound electrons, metalloids offer a controllable middle ground. Their electrical conductivity lies between these two extremes and, crucially, can be precisely manipulated.
This manipulability is achieved primarily through a process called “doping,” where small, controlled amounts of impurity elements are introduced into the metalloid’s crystal lattice. For instance, adding elements with an extra valence electron (like phosphorus) to silicon creates an n-type semiconductor (negative charge carriers), while adding elements with fewer valence electrons (like boron) creates a p-type semiconductor (positive “holes” as charge carriers). The ability to tune their conductivity, switching them between an insulating and a conducting state with precision, is what makes metalloids the cornerstone of modern electronics, enabling the binary logic (on/off states) that defines digital computing.
Silicon and Germanium: The Pillars of Modern Electronics
Among the metalloids, Silicon and Germanium stand out as titans of the tech world. Their unique semiconductor properties have been harnessed to create the fundamental building blocks of virtually every electronic device we use today.
Silicon: The Digital Age’s Foundation
Silicon (Si), the second most abundant element in the Earth’s crust, is the undisputed king of semiconductors. Its abundance, cost-effectiveness, and excellent intrinsic properties have made it the material of choice for the vast majority of microchips, transistors, and integrated circuits. The entire “Silicon Valley” is named after its foundational role in the tech industry.
The marvel of silicon lies in its crystalline structure and the precision with which its electrical properties can be engineered. A perfectly pure silicon crystal is an insulator at room temperature. However, by introducing minute quantities of dopants—typically Group 13 elements like boron (creating p-type silicon) or Group 15 elements like phosphorus or arsenic (creating n-type silicon)—its conductivity can be precisely controlled. The junction formed between p-type and n-type silicon creates a diode, the simplest semiconductor device, which allows current to flow in only one direction. Stacking these junctions, often with a thin layer of intrinsic silicon, creates transistors, the fundamental switch that powers all digital logic. Millions, even billions, of these tiny transistors are etched onto a single silicon wafer, forming the integrated circuits that are the “brains” of computers, smartphones, AI accelerators, and countless other intelligent systems. Silicon’s stable oxide (silicon dioxide, or glass) also serves as an excellent insulator in microchip fabrication, providing isolation between components and forming the dielectric layer in capacitors within circuits.
Germanium: A Niche, Yet Potent Player
While silicon dominates the semiconductor landscape, Germanium (Ge) holds a significant and historically important place. Germanium was the material used in the very first transistor invented in 1947 at Bell Labs, marking the dawn of the electronic age. Although largely supplanted by silicon due to silicon’s higher temperature tolerance and the stability of its oxide, germanium continues to be a vital component in various specialized high-tech applications.
Germanium possesses higher electron mobility than silicon, meaning electrons can move through it faster. This property makes it desirable for high-frequency applications, such as in radio frequency identification (RFID) tags, microwave circuits, and certain types of high-speed transistors where silicon’s performance might be insufficient. Germanium is also crucial in fiber optics, where germanium-doped silica glass enhances the refractive index of the fiber core, guiding light more efficiently. Its transparency to infrared light makes it ideal for infrared optics, thermal imaging cameras, and night vision devices. Furthermore, germanium is increasingly used in advanced solar cells, particularly in multi-junction photovoltaic cells, where it forms one of the light-absorbing layers, capturing a broader spectrum of sunlight and achieving higher efficiencies than traditional silicon cells alone. The ongoing research into silicon-germanium (SiGe) alloys also promises to combine the best properties of both elements, leading to even faster and more efficient electronic components.
Beyond Silicon and Germanium: Diverse Applications in Tech
While silicon and germanium are the most prominent, other metalloids also contribute significantly to the tech landscape, enabling various specialized and advanced applications.
Boron: From Hardness to High-Tech Materials
Boron (B) is a metalloid with unique characteristics that make it valuable in several high-tech sectors. It is exceptionally hard, second only to diamond among elemental solids, leading to its use in abrasive materials and protective coatings. However, its semiconductor properties are also exploited. Boron is a common p-type dopant for silicon semiconductors, a critical step in creating the p-n junctions essential for transistors.
Beyond traditional semiconductors, boron finds applications in advanced materials. Boron fibers are exceptionally strong and lightweight, making them ideal for aerospace components, sporting goods, and high-performance composites. Boron nitride, a compound of boron and nitrogen, exists in various forms, including hexagonal boron nitride (h-BN), which is an excellent electrical insulator and thermal conductor, and cubic boron nitride (c-BN), which is nearly as hard as diamond and used in cutting tools. Boron’s role in creating strong, heat-resistant, and chemically stable materials underscores its versatility in pushing material science boundaries for technological advancement.
Arsenic and Antimony: Essential Dopants and Alloys
Arsenic (As) and Antimony (Sb) are perhaps more infamous for their toxicity but are nevertheless critical metalloids in specific high-performance electronic applications. Both are used as n-type dopants for silicon and germanium, providing the necessary free electrons to create n-type semiconductors.
However, their most significant contribution comes in compound semiconductors. Gallium Arsenide (GaAs) is a prime example. Unlike silicon, GaAs is a direct bandgap semiconductor, meaning it can efficiently emit and absorb light. This property makes it indispensable for light-emitting diodes (LEDs), laser diodes (used in optical fiber communication and CD/DVD/Blu-ray players), and high-frequency microwave components. GaAs transistors can operate at much higher frequencies and speeds than silicon transistors, making them vital for satellite communications, radar systems, and supercomputers where speed is paramount. Antimony, on the other hand, is a common alloying agent. When alloyed with lead, it increases the hardness and strength of lead for battery grids. It’s also used in solders, particularly lead-free solders, and in thermoelectric alloys which can convert heat directly into electrical energy and vice-versa.
Tellurium: Thermoelectrics and Advanced Photovoltaics
Tellurium (Te) is another critical metalloid, primarily known for its role in advanced energy technologies and specialized electronics. Its most prominent application is in cadmium telluride (CdTe) thin-film solar cells. These solar cells are a significant alternative to traditional silicon cells, offering advantages in manufacturing costs and flexibility, making them suitable for large-scale solar power installations.
Beyond photovoltaics, tellurium compounds are central to thermoelectric devices. These devices, based on the Seebeck and Peltier effects, can convert temperature differences directly into electrical voltage (for power generation) or use electrical current to create a temperature difference (for cooling). Bismuth telluride (Bi₂Te₃) and lead telluride (PbTe) are common materials for thermoelectric coolers used in electronics to dissipate heat from sensitive components (e.g., in spacecraft, medical equipment, and high-performance CPUs) or in niche power generators that convert waste heat into usable electricity. Tellurium also finds use in phase-change memory, a promising non-volatile memory technology, and as an alloying agent to improve the machinability of metals.
The Future of Metalloids in Technology: Pushing Boundaries
The journey of metalloids from chemical curiosities to the bedrock of global technology is far from over. As the demands for faster, smaller, and more energy-efficient devices continue to grow, metalloids will remain at the forefront of innovation.
Miniaturization and Enhanced Performance
The constant drive towards miniaturization in electronics pushes the limits of material science. Metalloids, particularly silicon, have been instrumental in enabling Moore’s Law, allowing the density of transistors on integrated circuits to double approximately every two years. Future advancements will rely on further optimizing metalloid-based structures at the nanoscale. This includes developing novel architectures like FinFETs and Gate-All-Around (GAA) transistors, which maximize control over electron flow within ever-smaller dimensions. Researchers are also exploring strain engineering in silicon and silicon-germanium alloys to enhance carrier mobility, leading to faster transistors without further shrinking them. The continued ability to precisely manipulate the electrical properties of metalloids at the atomic level is crucial for extending the lifespan of silicon-based technologies and pushing performance boundaries.
Emerging Technologies and Novel Materials
Beyond refining existing applications, metalloids are poised to play critical roles in entirely new technological paradigms. In quantum computing, the precise control of individual electrons or nuclear spins, often within a silicon or germanium substrate, is fundamental. Understanding and exploiting the quantum properties of metalloids at ultralow temperatures is key to developing stable qubits.
Furthermore, the exploration of two-dimensional materials, such as silicene and germanene (analogues of graphene), derived from metalloids, promises revolutionary applications in flexible electronics, transparent conductors, and ultra-thin sensors. New compounds involving metalloids are being investigated for advanced photonics, neuromorphic computing (inspired by the human brain), and highly efficient energy harvesting. The unique electronic band structures and tunable properties of metalloids, when combined with innovative material engineering techniques, will undoubtedly unlock unforeseen capabilities, ensuring their continued indispensability in shaping the technological landscape of tomorrow.
Conclusion
Metalloids, a small but mighty group of elements bridging the divide between metals and nonmetals, are far more than a mere classification on the periodic table. They are the silent enablers of the modern world, the fundamental materials that allowed humanity to transition from vacuum tubes to microprocessors and beyond. From silicon, the ubiquitous foundation of our digital lives, to germanium’s role in high-speed and infrared technologies, and the specialized contributions of boron, arsenic, antimony, and tellurium, these elements underpin virtually every piece of technology we rely on. Their unique ability to be precisely controlled as semiconductors is not just a scientific curiosity; it is the very essence of digital logic and the driving force behind the ongoing technological revolution. As we continue to push the boundaries of miniaturization, performance, and entirely new computational paradigms, metalloids will undoubtedly remain at the heart of innovation, forever shaping the future of technology and human progress.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.