In the intricate landscape of the pharmaceutical industry, the brand name “Celexa” (citalopram) represents a well-established antidepressant medication. While the direct query about its side effects might initially seem purely clinical, from a brand perspective, the very concept of “side effects” is a profound and multi-faceted challenge that shapes perception, drives strategy, and underpins the ethical framework of pharmaceutical branding. This article will explore how the existence and communication of side effects—not just for Celexa, but as a universal truth for prescription medications—profoundly influence a pharmaceutical brand’s identity, its relationship with stakeholders, and its long-term market sustainability.

The Dual Challenge: Balancing Efficacy and Brand Trust
For any pharmaceutical brand, the primary objective is to highlight the efficacy and benefits of its product. However, this must always be balanced with the critical obligation to disclose potential adverse effects. This inherent tension forms the cornerstone of pharmaceutical branding strategy. How a brand manages this dual challenge directly impacts its trustworthiness and, ultimately, its success.
Navigating the Regulatory Landscape and Public Perception
Pharmaceutical brands operate within one of the most rigorously regulated environments globally. Agencies like the FDA in the United States or the EMA in Europe mandate comprehensive disclosure of all known side effects, from the common and mild to the rare and severe. This isn’t merely a legal requirement; it’s a fundamental aspect of brand integrity. For a brand like Celexa, ensuring that all marketing and patient information adheres strictly to these regulations is paramount. Any misstep can lead to severe penalties, loss of public trust, and irreparable damage to the brand’s reputation.
Public perception is heavily influenced by how transparent and proactive a brand is in communicating these details. In an age of instant information and social media, a single negative patient experience, if mismanaged, can quickly escalate into a widespread brand crisis. Therefore, brands must invest heavily in clear, accessible, and scientifically accurate communication channels to manage expectations and provide necessary context. The brand’s messaging must educate without alarming, and reassure without downplaying risks.
The Ethical Imperative in Pharmaceutical Branding
Beyond legal compliance, there is a strong ethical imperative that guides pharmaceutical branding, especially concerning side effects. The product directly impacts human health and well-being, placing a unique moral responsibility on the brand owner. This translates into a commitment to patient safety that must permeate every aspect of the brand’s existence, from product development and clinical trials to post-market surveillance and patient support programs.
For a brand, embracing this ethical imperative means going beyond mere disclosure. It means actively engaging with healthcare professionals, funding patient education initiatives, and ensuring that channels for reporting adverse events are robust and responsive. This builds a foundation of trust that is indispensable for long-term brand equity. When patients and healthcare providers perceive a brand as genuinely caring for patient safety, even in the face of potential side effects, it strengthens loyalty and positive association.
The Strategic Impact of Side Effect Profiles on Brand Identity
A drug’s side effect profile is not just a list of medical possibilities; it’s a critical strategic element that shapes the brand’s identity, its competitive positioning, and its marketing narratives. How a brand integrates this information into its core messaging is key to establishing its unique place in the market.
Shaping Corporate Identity and Brand Messaging
The side effect profile of a flagship drug often reflects on the overall corporate identity of the pharmaceutical company itself. A company known for a drug with a well-managed side effect profile, or one that has pioneered new standards in patient safety communication, builds a stronger corporate brand. Marketing teams for products like Celexa must skillfully craft messages that are both compliant and reassuring, highlighting the benefits while responsibly contextualizing potential drawbacks. This involves nuanced language, visual cues, and the careful selection of communication channels. The brand’s identity must convey competence, trustworthiness, and a deep understanding of patient needs.
Furthermore, the brand’s messaging often focuses on the management of side effects as much as the effects themselves. Offering resources, advice for coping, or emphasizing the transient nature of certain side effects can be part of the brand’s value proposition, positioning it as a supportive partner in patient care.
Differentiating in a Competitive Market
In a crowded therapeutic area with multiple drug options, a medication’s side effect profile can become a significant point of differentiation. If a drug offers a similar efficacy to competitors but with a demonstrably milder or more manageable side effect profile for certain patient populations, this can be a powerful marketing advantage. Conversely, a more challenging side effect profile necessitates a branding strategy that emphasizes unique benefits, specific patient populations for whom the drug is particularly suited, or innovative support mechanisms.
For brands facing generic competition, managing side effect perceptions becomes even more critical. Original brands often leverage their extensive clinical data and long-standing safety records to reinforce trust, differentiating themselves from newer, often cheaper, generic alternatives that may not have the same breadth of real-world post-market data. The brand narrative can focus on consistent quality, comprehensive patient support, and a history of reliable performance.
Communicating Adverse Events: A Brand’s Reputation on the Line
The way a pharmaceutical brand communicates about adverse events is a direct reflection of its commitment to transparency and patient well-being. This communication is not merely reactive; it’s a proactive strategy to build and maintain trust.
Transparency as a Pillar of Brand Loyalty
Proactive and clear communication about side effects is foundational to building brand loyalty in the pharmaceutical sector. Patients and healthcare providers are far more likely to trust a brand that is forthright and comprehensive in its disclosures, even when the information may not be entirely positive. Hiding or downplaying risks, even unintentionally, can severely erode trust and lead to a perception of corporate insincerity.
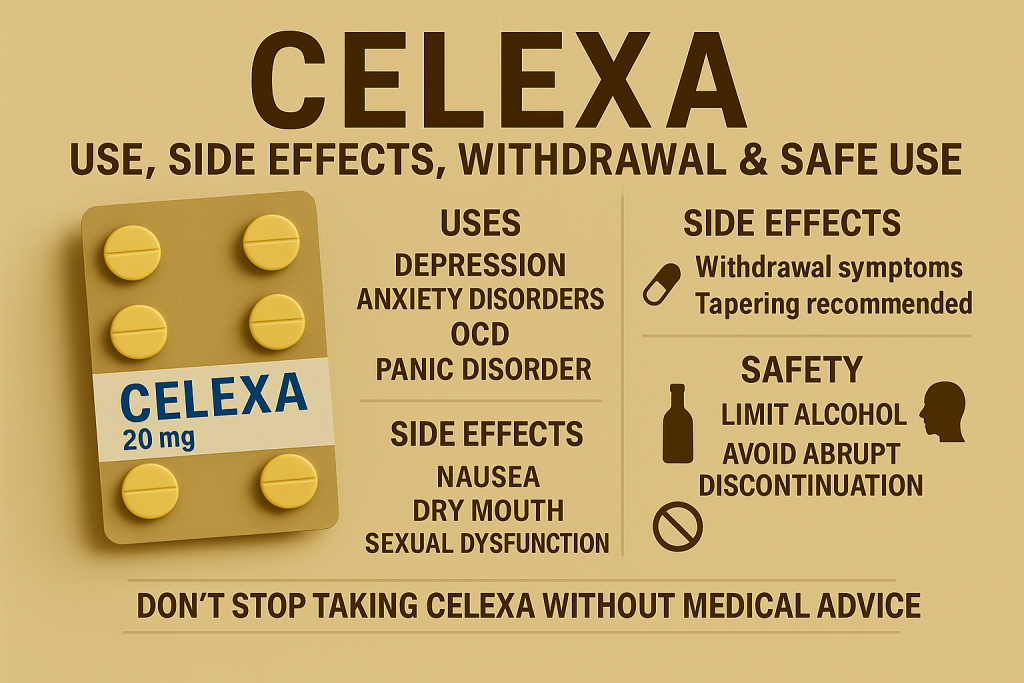
For a brand like Celexa, transparency means making patient information leaflets easy to understand, providing readily accessible resources for questions, and ensuring that healthcare professionals are fully informed. It involves a continuous dialogue, not just a one-time disclosure. This approach fosters a sense of partnership between the brand, patients, and prescribers, leading to greater adherence and better health outcomes, which in turn reinforces positive brand perception.
Crisis Management and Reputation Safeguarding
Despite best efforts, adverse events or public concerns can arise. Pharmaceutical brands must have robust crisis management plans in place to address these situations swiftly and effectively. This involves not only legal and medical teams but also a dedicated communications strategy that prioritizes transparency, empathy, and accuracy.
When a brand responds to a crisis by openly acknowledging concerns, providing clear data, and outlining steps for resolution, it can mitigate reputational damage. Conversely, a defensive or evasive stance can amplify negative sentiment and lead to lasting brand scars. The reputation of a brand is built over years but can be compromised in moments of crisis, making a well-rehearsed and ethically guided response critical for safeguarding its future.
From Clinical Data to Consumer Experience: The Brand Journey
The journey of a pharmaceutical brand, especially concerning side effects, extends far beyond initial clinical trials and market launch. It encompasses the entire patient experience, from prescription to ongoing management.
Patient Engagement and Education as Brand Touchpoints
Every interaction a patient has with information about a drug, its support materials, or healthcare providers discussing it, is a brand touchpoint. For managing side effects, patient engagement and education are paramount. Brands often develop comprehensive patient support programs, websites with detailed FAQs, and helplines. These resources become extensions of the brand’s commitment to patient well-being, providing practical advice on how to manage common side effects, when to seek medical attention, and what to expect during treatment.
By offering these robust support systems, a brand distinguishes itself not just as a provider of medication, but as a holistic partner in health management. This adds significant value, particularly for chronic conditions where long-term adherence and ongoing support are crucial. The brand builds an identity that is supportive, informative, and patient-centric.
Post-Market Surveillance and Brand Evolution
The lifecycle of a pharmaceutical brand continues long after market approval. Post-market surveillance, or pharmacovigilance, involves continuously monitoring the drug’s safety in real-world conditions, gathering data on previously unknown or rare side effects, and refining existing knowledge. This ongoing process is vital for the brand’s evolution.
New safety information can necessitate updates to labeling, adjustments to prescribing guidelines, or even targeted patient education campaigns. A brand that actively engages in and communicates about its pharmacovigilance efforts reinforces its commitment to patient safety and scientific rigor. This continuous learning and adaptation ensure the brand remains relevant, trusted, and compliant, allowing it to evolve with new scientific understanding and patient needs. It demonstrates a dynamic and responsible corporate identity that is prepared to adapt its product and messaging based on real-world evidence.
Future-Proofing Pharmaceutical Brands in an Information-Rich Era
In today’s digital age, information about side effects, both accurate and inaccurate, spreads rapidly. Future-proofing pharmaceutical brands requires a proactive and technologically informed approach to managing and communicating this crucial data.
Leveraging Digital Platforms for Responsible Information Dissemination
Digital platforms offer unprecedented opportunities for pharmaceutical brands to disseminate accurate and accessible information about their products and their side effects. Official websites, patient portals, and even curated social media content can serve as vital resources. Brands must ensure their digital presence is not only compliant with regulations but also user-friendly, engaging, and trustworthy.
By providing clear, concise, and multi-format information (e.g., videos, infographics, interactive tools), brands can empower patients to make informed decisions and better manage their treatment journey. This digital strategy reinforces the brand’s image as a reliable and modern health partner.
Building Brand Resilience Through Proactive Risk Management
Building brand resilience in the context of side effects means adopting a proactive risk management strategy. This involves anticipating potential concerns, conducting thorough research, and developing comprehensive communication plans before issues arise. It’s about having a strong internal culture of transparency and patient safety that permeates all levels of the organization.
A resilient brand is one that can withstand scrutiny, adapt to new information, and maintain trust even when faced with challenges related to product safety. By continuously investing in research, ethical communication, and patient support, pharmaceutical brands can safeguard their reputation and ensure long-term success in a complex and ever-evolving healthcare landscape.

Conclusion
The question “what are the side effects of Celexa?” transcends a simple medical inquiry when viewed through the lens of brand strategy. For pharmaceutical brands, side effects are not just clinical data points; they are pivotal elements that shape corporate identity, dictate marketing strategies, drive regulatory compliance, and fundamentally influence public trust and patient loyalty. Managing and communicating about side effects effectively is a cornerstone of responsible pharmaceutical branding. It requires a delicate balance of scientific accuracy, ethical responsibility, and strategic communication, ultimately determining a brand’s longevity and its perceived value in the lives of patients.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.