The pharmaceutical landscape is constantly evolving, with innovative treatments emerging to address a range of health concerns. Among these, Tirzepatide has garnered significant attention for its efficacy in managing type 2 diabetes and, more recently, for its potential in weight management. As individuals explore treatment options, a crucial question arises: what is the maximum dose of Tirzepatide? This inquiry is paramount for understanding the therapeutic window, optimizing treatment outcomes, and ensuring patient safety.
This article delves into the complexities surrounding Tirzepatide dosage, drawing parallels to principles found across technology, brand building, and financial management – domains where understanding limits, scaling, and strategic allocation are critical. While Tirzepatide is a medical treatment, the underlying concepts of optimization, responsible application, and achieving desired results resonate across these diverse fields.
Understanding Tirzepatide: A Dual-Action Approach
Tirzepatide operates as a novel dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist. This unique mechanism of action allows it to target multiple pathways involved in glucose regulation and appetite control. By stimulating insulin secretion, reducing glucagon secretion, slowing gastric emptying, and increasing satiety, Tirzepatide offers a comprehensive approach to managing type 2 diabetes and promoting weight loss.
The development and approval of Tirzepatide have been a significant technological advancement in metabolic disease treatment. Like a cutting-edge software update, its dual-action mechanism represents an evolution beyond single-receptor agonists, offering enhanced efficacy. The journey from initial research and development to clinical trials and eventual market approval is a testament to rigorous scientific methodology and technological innovation in pharmaceutical research. This process mirrors the iterative development cycles seen in the tech world, where constant refinement and testing lead to more robust and effective products.
Determining the Maximum Effective and Safe Dosage
The question of “what is the maximum dose of Tirzepatide” is not a static one. It’s a dynamic exploration aimed at identifying the upper limit of therapeutic benefit while minimizing the risk of adverse events. Clinical trials are instrumental in establishing these dosage ranges. These trials systematically evaluate different doses in diverse patient populations to identify the optimal balance between efficacy and safety.
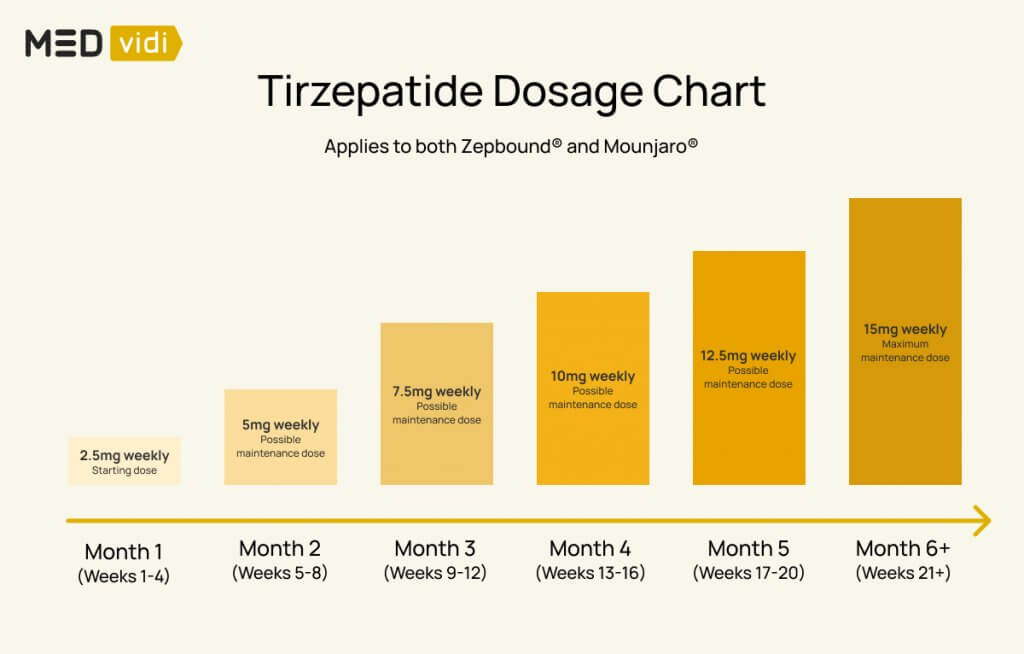
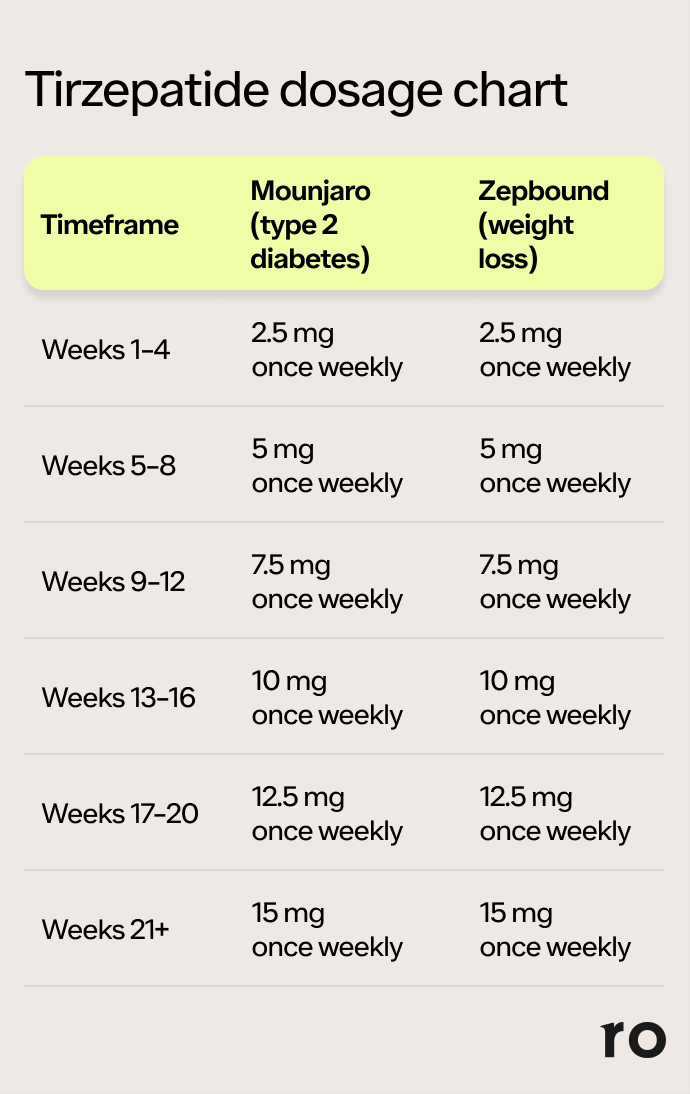
Approved Dosing and Titration Schedules
Currently, Tirzepatide is approved for the treatment of type 2 diabetes and chronic weight management in adults with overweight or obesity, or with weight-related comorbidities. The approved starting dose is typically low, with a gradual titration schedule designed to allow the body to adapt to the medication and reduce the likelihood of gastrointestinal side effects, which are common with GLP-1 receptor agonists.
The titration schedule is a critical aspect of Tirzepatide therapy. It’s akin to a brand’s initial market entry and subsequent expansion. A carefully managed rollout, starting with a foundational presence and gradually scaling up, builds trust and allows for adaptation. In the case of Tirzepatide, the slow increase in dosage allows patients to acclimate to the drug’s effects, and for healthcare providers to monitor for any emerging intolerances. This measured approach is a cornerstone of responsible medical practice, mirroring the strategic planning required for successful product launches and brand growth.
The typical titration schedule involves increasing the dose every four weeks, provided the patient tolerates the current dose and is experiencing clinical benefits. This systematic approach is designed to reach the maximum tolerated and effective dose for each individual.
The Role of Clinical Trials in Defining Maximum Dose
The determination of the maximum dose is heavily reliant on the data generated from extensive clinical trials. These trials, such as the SURMOUNT trials for weight management and SURPASS trials for type 2 diabetes, have been pivotal in establishing the efficacy and safety profiles of Tirzepatide at various dose levels.
These trials are akin to large-scale A/B testing in technology or market research for brands. Researchers meticulously compare outcomes across different dosage groups to identify the dose that provides the greatest clinical benefit without a significant increase in adverse events. The “maximum dose” is essentially the highest dose studied and found to be safe and effective within the parameters of these trials.
For Tirzepatide, clinical trials have investigated doses up to 15 mg once weekly. In some instances, higher doses may be explored in ongoing research, but the approved maximum dose for widespread clinical use is generally understood to be the highest dose demonstrated to be safe and effective in pivotal studies.
Factors Influencing Individual Dosage and Maximum Tolerance
It’s crucial to understand that “maximum dose” is not a one-size-fits-all prescription. Individual responses to medications can vary significantly due to a multitude of factors. This variability is comparable to how different investment strategies yield different returns based on individual risk tolerance and market conditions, or how a technology solution might be optimized differently for distinct user bases.
Patient-Specific Considerations
Several patient-specific factors can influence how an individual tolerates and benefits from Tirzepatide, and consequently, their “maximum” dose. These include:
- Body Weight and Composition: While Tirzepatide is used for weight management, a patient’s baseline weight and body composition can influence how they metabolize the drug and experience its effects.
- Kidney Function: Impaired kidney function can affect drug clearance, potentially requiring dose adjustments.
- Liver Function: Similar to kidney function, liver function plays a role in drug metabolism.
- Age: Age-related changes in physiology can influence drug response.
- Presence of Comorbidities: Other underlying health conditions can interact with Tirzepatide or affect its metabolism.
- Concomitant Medications: Interactions with other medications can influence both the efficacy and safety of Tirzepatide.
- Individual Tolerance to Side Effects: The most common side effects of Tirzepatide are gastrointestinal, such as nausea, vomiting, diarrhea, and constipation. A patient’s ability to tolerate these side effects will ultimately determine how high their dose can be safely and comfortably increased.
This nuanced approach to dosage is akin to personal finance where financial tools are not universally applied. Instead, they are tailored to an individual’s income, expenses, risk tolerance, and financial goals. A wealth management strategy, for example, is highly personalized, recognizing that what works for one person might not be suitable for another.
The Titration Process as an Individualized Approach
The gradual titration schedule is inherently designed to accommodate individual differences. Healthcare providers monitor patients closely during this process, assessing their response to each dose increment. If a patient experiences significant side effects or does not tolerate a particular dose, their physician may decide to slow down the titration, maintain a lower dose, or even discontinue the medication. Conversely, if a patient tolerates a dose well and is experiencing benefits, their physician may proceed with further increases as per the established guidelines.
This iterative process of assessment and adjustment ensures that each patient receives the highest possible dose that is both effective and well-tolerated for their unique physiology and circumstances. It’s a continuous optimization loop, much like a developer refining an app based on user feedback and performance metrics to achieve the best possible user experience.
Balancing Efficacy and Safety: The Ethical Imperative
The pursuit of the “maximum dose” should always be balanced with a commitment to patient safety. This ethical consideration is paramount in medicine and mirrors the responsible innovation principles in technology and the importance of ethical branding.
Understanding Potential Side Effects at Higher Doses
As the dose of Tirzepatide increases, so does the potential for adverse events. While generally well-tolerated, higher doses may be associated with a greater incidence or severity of gastrointestinal side effects. Other potential, though less common, side effects can include pancreatitis, gallbladder disease, and diabetic retinopathy complications (especially in those with pre-existing eye conditions).
Healthcare professionals are trained to identify and manage these potential side effects. They will weigh the potential benefits of a higher dose against the risks for each individual patient. This risk-benefit analysis is a fundamental aspect of medical decision-making.
The Role of the Healthcare Provider
The “maximum dose” of Tirzepatide is not determined by the patient alone. It is a decision made collaboratively between the patient and their prescribing healthcare provider. The physician’s expertise is essential in interpreting clinical trial data, assessing individual patient factors, monitoring for side effects, and ultimately determining the optimal dosage for each person.
This collaborative approach is analogous to the relationship between a financial advisor and their client. The advisor provides expert guidance and recommendations based on market knowledge and the client’s financial situation, but the ultimate decisions are made by the client in consultation with the advisor. Similarly, in branding, a marketing strategist advises a company, but the brand’s direction is guided by the company’s vision and values.
Moving Beyond the Label: Off-Label Use and Ongoing Research
While clinical trials establish approved dosages, scientific inquiry rarely stops there. Ongoing research may explore the efficacy and safety of Tirzepatide at doses beyond those currently approved for specific indications. This might involve studying higher doses for patients who haven’t achieved optimal results at the maximum approved dose, or exploring its use in other patient populations.
However, it is crucial to emphasize that any use of medication outside of its approved indications (off-label use) should be carefully considered, undertaken with extreme caution, and only under the strict supervision of a qualified healthcare professional who is fully informed about the latest research and potential risks.

Conclusion: A Personalized Journey to Optimal Treatment
In summary, when inquiring about “what is the maximum dose of Tirzepatide,” it is essential to understand that this is not a fixed number. It is a guideline, informed by robust clinical trials, that represents the highest dose found to be both safe and effective for the majority of patients within specific approved indications.
The journey to the maximum effective dose for any individual is a personalized one, guided by their unique physiological characteristics, tolerance to side effects, and in close collaboration with their healthcare provider. This approach prioritizes not just the pursuit of maximum therapeutic benefit, but also the fundamental principle of patient safety. As with advancements in technology, strategic brand evolution, and prudent financial planning, understanding the nuances of dosage, the importance of individual variability, and the ethical considerations involved are key to achieving optimal and responsible outcomes with Tirzepatide. The continuous evolution of medical research promises to further refine our understanding of this powerful medication, ensuring its safe and effective use for years to come.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.