The question “what is the max amount of Tylenol per day?” might seem, at first glance, to be a purely medical inquiry, demanding a straightforward numerical answer. However, when viewed through the strategic lens of brand management, this seemingly simple question unlocks a complex and critical dimension of how pharmaceutical companies, particularly those with iconic consumer brands like Tylenol, navigate the delicate balance between product efficacy, consumer safety, regulatory compliance, and the perpetual maintenance of brand trust. For a brand, answering this question isn’t just about providing a number; it’s about crafting a narrative of responsibility, clarity, and authority that underpins its entire market presence and consumer relationship.

In the highly regulated and ethically sensitive pharmaceutical industry, every piece of consumer-facing information, especially concerning dosage and safety, becomes a cornerstone of brand identity. Tylenol, a flagship product of Johnson & Johnson, has spent decades building a formidable brand synonymous with pain relief and reliability. Yet, the active ingredient, acetaminophen, carries known risks when misused or taken in excess, primarily liver damage. This inherent duality—benefit alongside potential harm—necessuates a sophisticated and unwavering commitment to clear, unambiguous, and omnipresent dosage communication. This article delves into the strategic imperatives and nuanced approaches a brand like Tylenol employs to communicate critical safety information, specifically “the max amount per day,” ensuring consumer welfare while simultaneously reinforcing its brand equity.
The Paradox of Product Information: Balancing Accessibility and Responsibility
For any consumer-facing pharmaceutical brand, the communication of product information, especially regarding dosage, presents a profound paradox: it must be simultaneously accessible enough for the average consumer to understand without medical training, yet comprehensive and responsible enough to meet rigorous scientific and regulatory standards. This is not merely an informational exercise but a critical brand-building endeavor. Miscommunication or under-communication of dosage limits can erode trust, invite legal scrutiny, and, most importantly, endanger consumers, leading to catastrophic brand damage.
Simplifying Complex Medical Data for the Consumer
The scientific data behind pharmaceutical dosages is inherently complex, involving pharmacokinetic profiles, clinical trial results, and risk assessments. Translating this intricate medical language into layman’s terms without sacrificing accuracy is a monumental task for brand strategists and medical communicators. Brands like Tylenol must distill vast amounts of scientific information into actionable, easy-to-understand instructions. This often involves:
- Plain Language Summaries: Employing clear, concise language devoid of jargon, ensuring readability for a broad demographic.
- Visual Cues: Using graphics, icons, and color-coding on packaging and inserts to highlight crucial warnings and dosage instructions.
- Repetition and Prominence: Strategically placing maximum dosage information in multiple, highly visible locations on packaging, inserts, and digital platforms to ensure it cannot be easily missed.
- Contextualization: Explaining why a maximum dosage exists (e.g., “to avoid liver damage”) helps consumers understand the gravity and adhere to guidelines more faithfully than a mere number.
This simplification is not about trivializing the science but about optimizing the delivery of critical safety information, transforming complex data into a fundamental element of responsible brand stewardship. The brand’s commitment to clarity becomes a testament to its commitment to consumer well-being, directly impacting perception and loyalty.
The Legal and Ethical Imperatives of Dosage Communication
Beyond the desire for consumer welfare, pharmaceutical brands operate within a formidable framework of legal and ethical obligations. Regulatory bodies like the U.S. Food and Drug Administration (FDA) impose stringent requirements on how drug information, particularly maximum dosage, is presented. Failure to comply can result in severe penalties, product recalls, and irreparable reputational damage.
- Regulatory Compliance: Every word, every numerical limit, and every warning on a Tylenol package is meticulously reviewed to ensure it adheres to current regulatory guidelines. These guidelines are dynamic, evolving with new scientific understanding and public health concerns. A brand’s agility in adapting its communication to these changes is a marker of its operational excellence and commitment to public safety.
- Legal Liability: Clear and unambiguous dosage instructions are a vital defense against product liability lawsuits. In cases of misuse, a brand must demonstrate that it took every reasonable step to inform consumers of safe usage limits. This necessitates not just providing the information but ensuring its prominence and comprehensibility.
- Ethical Responsibility: At its core, a pharmaceutical brand has an ethical duty to “do no harm.” This moral imperative transcends legal requirements, guiding decisions on how aggressively warnings are displayed, how readily information is made available, and how proactively consumer education campaigns are launched. For Tylenol, communicating “the max amount per day” is an exercise in ethical leadership, affirming its commitment to the health of its users.
Tylenol as a Case Study: A Legacy of Trust and Risk Management
Tylenol’s journey is a compelling case study in how a brand builds and maintains trust, especially when confronted with the inherent risks associated with its core product. The brand’s approach to communicating maximum daily dosage is deeply intertwined with its history, its brand promise, and its strategic response to evolving public health concerns.
From Product Efficacy to Consumer Safety: Evolving Brand Messaging
Initially, many pharmaceutical marketing efforts focused heavily on product efficacy – how well a product works to alleviate symptoms. While efficacy remains paramount, brands like Tylenol have progressively evolved their messaging to place consumer safety at an equally prominent, if not leading, position. This shift reflects:
- Increased Consumer Awareness: Modern consumers are more informed and demand transparency about product risks.
- Regulatory Scrutiny: Growing regulatory emphasis on patient safety has pushed brands to be more explicit about potential adverse effects and usage limits.
- Proactive Risk Management: Brands learned that proactively addressing safety concerns fosters greater trust than reacting to crises.
For Tylenol, this evolution meant making maximum daily dosage a central theme, not just an adjunct, to its overall brand communication. Campaigns might highlight the brand’s effectiveness but always with a strong undercurrent of responsible use. Messaging around “take only as directed” or “read the label carefully” isn’t merely disclaimers; they are integral parts of the brand’s commitment to consumer well-being, effectively transforming safety guidelines into a brand attribute. The “max amount per day” is presented not as a limitation, but as a protective measure, delivered by a brand that cares.
Crisis Management and Brand Resilience: Lessons from the Past
Tylenol’s history, particularly the 1982 tampering incidents, stands as a seminal example of brand crisis management. While that crisis was about external malicious acts, it ingrained in the brand’s DNA a profound understanding of the fragility of consumer trust and the absolute necessity of transparent, proactive communication in safeguarding it. In the context of dosage, this translates into:
- Unwavering Clarity: Following scientific updates on acetaminophen toxicity, Tylenol, under Johnson & Johnson’s leadership, was instrumental in reducing the recommended maximum daily dose from 4,000 mg to 3,000 mg for adults and clearly communicated this change across all platforms. This proactive adjustment, though potentially impacting sales, prioritized public safety and reinforced the brand’s integrity.
- Multi-pronged Communication: When dosage recommendations change, or when new safety concerns emerge, Tylenol doesn’t just update its packaging. It leverages extensive public relations campaigns, engages healthcare professionals, and utilizes digital channels to ensure the message reaches as wide an audience as possible.
- Building a Safety Culture: The emphasis on “the max amount per day” for Tylenol isn’t just a marketing directive; it’s a reflection of an organizational culture deeply committed to pharmacovigilance and consumer safety. This internal commitment translates into external credibility and resilience in the face of potential challenges.
This proactive approach to risk management, informed by historical lessons, demonstrates how communication around dosage limits becomes a continuous brand-building exercise, ensuring longevity and unwavering consumer confidence.
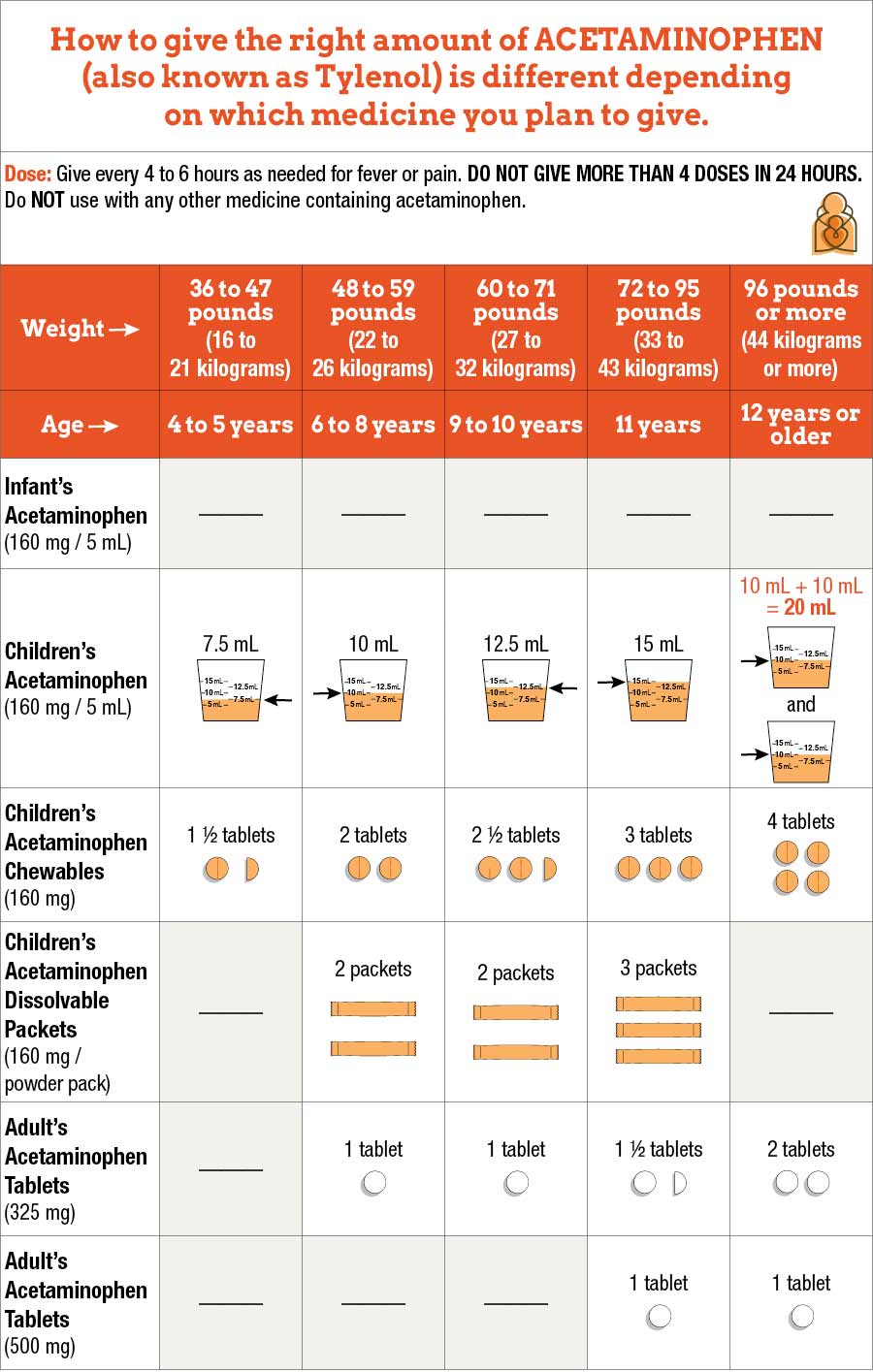
Crafting the Message: Design, Packaging, and Digital Touchpoints
The effective communication of “the max amount of Tylenol per day” goes far beyond simply stating a number. It involves strategic design, meticulous packaging, and comprehensive digital engagement, all orchestrated to ensure clarity, impact, and reach. These elements are not just functional; they are fundamental components of the brand’s identity and its commitment to responsible consumer education.
Packaging as a Critical Communication Tool
In the pharmaceutical world, packaging is often the first, and sometimes only, direct interaction a consumer has with the brand’s safety guidelines. Therefore, Tylenol’s packaging design is a masterclass in making crucial dosage information impossible to ignore:
- Prominent Placement: The “max amount per day” is typically featured prominently on the front, back, or side panels, often in a distinct box or contrasting color to draw the eye.
- Clear Language and Typography: Large, legible fonts and simple, direct language (“Do not exceed X doses in 24 hours”) are used.
- Dosing Devices: Where applicable (e.g., liquid formulations), clear dosing cups or syringes with marked measurements reinforce accurate administration.
- Child-Resistant Features: While not directly about dosage, child-resistant caps are another layer of packaging innovation that underscores a brand’s holistic approach to safety, aligning with the ethos of protecting vulnerable users from accidental overdose.
Every element of the packaging design is carefully considered to minimize confusion and maximize adherence to safety guidelines, positioning the package itself as a trusted guardian of consumer health and a silent spokesperson for the brand’s commitment.
Digital Platforms and Responsible Content Dissemination
In the digital age, a brand’s responsibility to communicate dosage information extends far beyond physical packaging. Websites, social media channels, and health-related apps have become vital touchpoints for informing and educating consumers. Tylenol strategically leverages these platforms to:
- Dedicated Product Pages: Official brand websites feature comprehensive dosage instructions, FAQs, and warnings, often in multiple languages. These pages are designed to be easily navigable and searchable.
- Educational Content: Beyond direct instructions, digital platforms host articles, videos, and infographics explaining why dosage limits are crucial and the risks associated with exceeding them. This content positions the brand as a helpful, authoritative resource.
- Social Media Responsibility: While social media offers opportunities for engagement, pharmaceutical brands must navigate it cautiously. Tylenol’s social media presence focuses on general wellness, brand awareness, and directing users to official sources for specific medical information, reinforcing the importance of consulting packaging or healthcare professionals for dosage specifics.
- Integration with Health Apps: Some brands explore partnerships or integration with health and wellness apps that can provide dosage reminders and tracking, further embedding safe usage practices into daily consumer routines.
By maintaining a consistent, clear, and readily accessible digital footprint for its safety guidelines, Tylenol ensures that “the max amount per day” is not a static piece of information but an actively managed and disseminated message across the consumer’s digital ecosystem.
The Consumer-Brand Relationship: Fostering Informed Decision-Making
Ultimately, the goal of communicating “the max amount of Tylenol per day” is to cultivate a relationship of trust where consumers feel empowered to make informed decisions about their health. This goes beyond mere information dissemination; it’s about education, advocacy, and solidifying the brand’s role as a trusted partner in health management.
Empowering Consumers Through Education, Not Just Information
Simply providing numbers isn’t enough; consumers need to understand the implications. Brands like Tylenol understand that truly informed decision-making comes from education. This involves:
- Understanding the “Why”: Explaining the dangers of exceeding recommended doses (e.g., liver damage) provides a powerful motivator for compliance.
- Addressing Common Misconceptions: Educating consumers about potential pitfalls, such as unknowingly taking multiple products containing acetaminophen (often referred to as “combination product awareness”), is crucial. This proactive guidance demonstrates a deep understanding of consumer behavior and a commitment to preventing harm.
- Promoting Readership: Encouraging consumers to always read the label and patient information leaflet, even for familiar products, is a consistent message. This empowers consumers to take personal responsibility, guided by the brand’s clear directives.
By investing in consumer education, Tylenol elevates its brand from a mere product provider to a health advocate, strengthening the bond of trust and positioning itself as a reliable source of vital health information.
The Role of Healthcare Professionals in Brand Reinforcement
Healthcare professionals (HCPs) play a pivotal role in reinforcing a pharmaceutical brand’s safety messages, including maximum dosage. Brands strategically engage with HCPs through:
- Professional Education: Providing detailed scientific information and safety updates to doctors, pharmacists, and nurses ensures they are well-equipped to counsel patients.
- Trust and Recommendation: When HCPs reiterate dosage instructions and safety warnings, it lends immense credibility to the brand’s messaging, as consumers implicitly trust their medical providers.
- Partnerships: Collaborating with medical associations or patient advocacy groups can amplify safety messages, demonstrating the brand’s commitment to public health beyond commercial interests.
This indirect yet powerful channel of communication reinforces the brand’s authority and integrity. When a doctor or pharmacist echoes the “max amount per day” message found on Tylenol’s packaging, it strengthens the brand’s credibility and its perceived commitment to patient safety.

Conclusion
The seemingly straightforward question, “what is the max amount of Tylenol per day?”, transforms into a microcosm of sophisticated brand strategy when examined through the lens of pharmaceutical brand management. For an iconic brand like Tylenol, providing this information is far more than a regulatory obligation; it is a profound expression of its brand identity, its commitment to consumer trust, and its strategic approach to risk management.
By meticulously balancing accessibility with scientific responsibility, navigating complex legal and ethical landscapes, and consistently evolving its communication strategies across packaging and digital platforms, Tylenol has established itself not just as a pain reliever but as a trusted partner in health. Its rigorous approach to communicating the “max amount per day” underscores a continuous dedication to consumer safety, ensuring that its powerful brand legacy remains synonymous with reliability, transparency, and responsible care. In an industry where trust is paramount, the clarity and consistency of such vital information are the very pillars upon which enduring brand equity is built and sustained.
aViewFromTheCave is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com. Amazon, the Amazon logo, AmazonSupply, and the AmazonSupply logo are trademarks of Amazon.com, Inc. or its affiliates. As an Amazon Associate we earn affiliate commissions from qualifying purchases.